Clear Sky Science · en
Aryl sulfur ligand-modulated silver catalysts with tailored binding affinity for selective nitrate-to-ammonia conversion
Turning Pollution into a Valuable Fertilizer
Modern farming depends heavily on ammonia-based fertilizers, but making ammonia the traditional way devours fossil fuels and releases large amounts of carbon dioxide. At the same time, excess nitrate from fertilizers and industrial waste contaminates rivers and groundwater. This study explores a way to tackle both problems at once: using smartly designed silver-based catalysts that can turn unwanted nitrate in water directly into useful ammonia under mild, electrically driven conditions.
Why Nitrate and Ammonia Matter
Ammonia is the backbone of fertilizer production, and global demand has pushed its manufacture to about 190 million tonnes per year, mostly via the century‑old Haber–Bosch process. That process runs at high temperature and pressure and accounts for a noticeable slice of global energy use and carbon emissions. Meanwhile, runoff from farms and factories loads waterways with nitrate, which can harm ecosystems and drinking water supplies. A technology that converts nitrate pollution into ammonia at room temperature, powered by electricity, could both clean water and provide fertilizer in a more climate‑friendly way.
Shaping the Surface of Silver
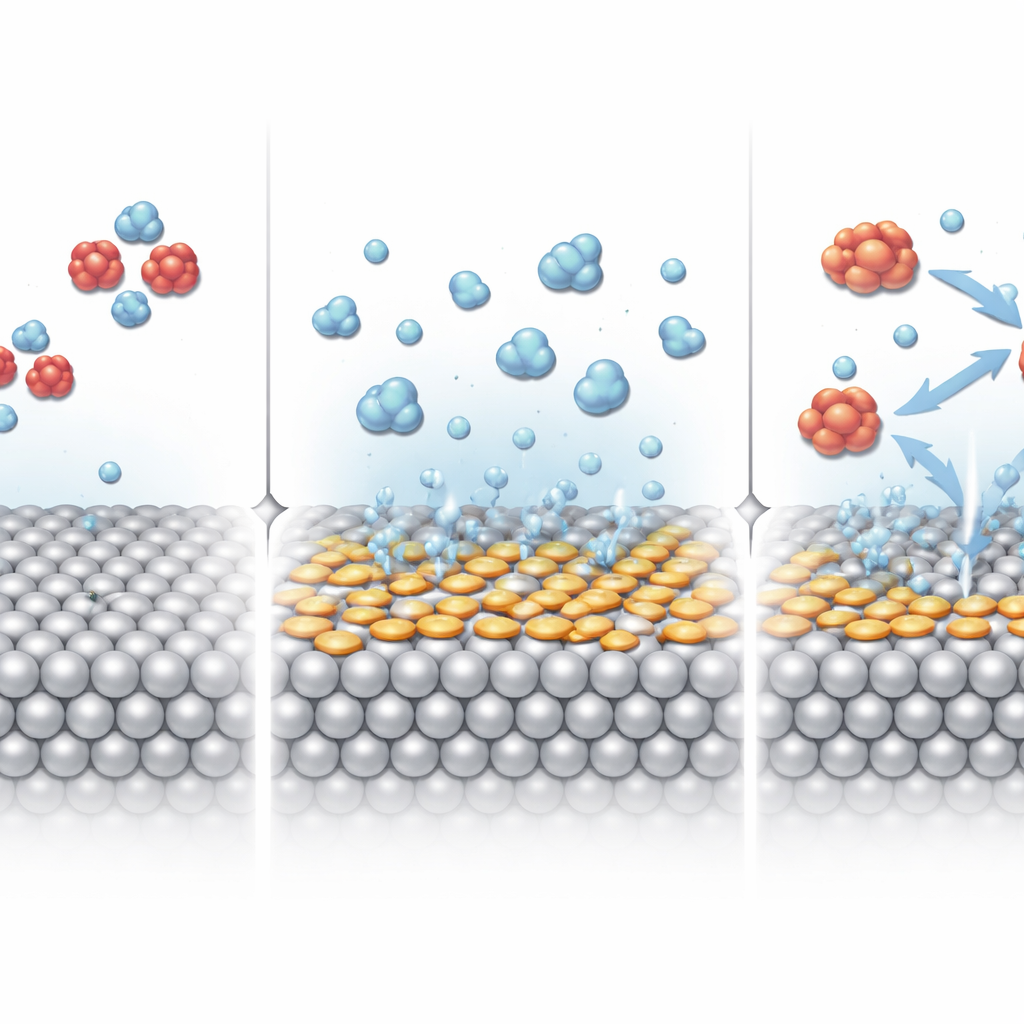
Silver is known to be good at grabbing nitrate and starting its chemical breakdown, but it struggles to carry the reaction all the way to ammonia. The key problem lies in how strongly the silver surface holds onto nitrogen‑containing fragments along the way. The researchers tackled this by “dressing” tiny silver cubes with a family of sulfur‑containing organic molecules that attach firmly to the metal. By carefully changing the electronic character of these aryl sulfur ligands, they could subtly reshape how the silver surface interacts with reaction intermediates without changing the overall size or shape of the nanoparticles.

Finding the Best Molecular Add‑On
Using a combination of computer simulations and electrochemical tests, the team screened five different ligands that either donate or withdraw electron density from the silver surface. Calculations showed that these molecules shift the charge on surface silver atoms and tune how easily nitrate sticks, breaks apart, and reacts with water‑derived hydrogen. One ligand, 4‑(methylthio)benzaldehyde (MTBA), emerged as a standout: it raised the apparent oxidation state of surface silver atoms and created sites that bind key intermediates just strongly enough to speed the reaction, but not so strongly that they become stuck. Experiments confirmed this prediction: MTBA‑modified silver nanocubes almost doubled the fraction of electrical charge that ends up as ammonia, boosting the ammonia yield efficiency from about 51% to nearly 99%, and increasing the production rate by roughly two and a half times.
How Water and Intermediates Work Together
To understand why MTBA works so well, the researchers probed the interface where the solid catalyst, water, and nitrate meet. Advanced Raman spectroscopy revealed that, under working conditions, the MTBA‑decorated surface attracts a population of more weakly hydrogen‑bonded water molecules, which are easier to split into reactive hydrogen species. Electron spin measurements showed that these reactive hydrogen atoms are generated more readily on the modified surface and are quickly consumed in hydrogenation steps instead of making hydrogen gas. Further in situ spectroscopy detected intermediates such as HNO forming at milder voltages and in higher amounts on the MTBA‑treated silver, indicating that the added molecules help funnel both nitrate fragments and hydrogen together efficiently, step by step, toward ammonia rather than side products.
From Lab Cell to Practical Device
Moving beyond small test cells, the team built a membrane‑based electrolyzer using the MTBA‑modified silver nanocubes as the cathode. In nitrate‑containing alkaline water, this device delivered high electrical currents while maintaining ammonia selectivities above 90% for more than 100 hours, and it reduced nitrate and nitrite concentrations in model wastewater below drinking‑water limits within an hour and a half. A simple economic analysis suggests that, if powered by low‑cost electricity, such systems could make ammonia from nitrate‑rich wastewater at costs competitive with current industrial production—while simultaneously offering a service of pollution cleanup.
What This Means Going Forward
This work shows that carefully chosen organic molecules on a metal surface can act like fine‑tuning knobs, steering a complex electrochemical reaction toward a single desired product. By using aryl sulfur ligands such as MTBA to adjust the binding strength of key intermediates and to better activate water, the authors turned silver nanocubes into highly selective machines for converting nitrate to ammonia. The concept of molecular interface engineering demonstrated here could be extended to other metals and reactions, offering a blueprint for cleaner fertilizer production and more sustainable treatment of nitrogen‑laden waste streams.
Citation: Zhang, L., Liu, Y., Li, L. et al. Aryl sulfur ligand-modulated silver catalysts with tailored binding affinity for selective nitrate-to-ammonia conversion. Nat Commun 17, 2553 (2026). https://doi.org/10.1038/s41467-026-69385-1
Keywords: electrocatalytic nitrate reduction, ammonia synthesis, silver nanocatalyst, wastewater valorization, interface engineering