Clear Sky Science · en
Engineered exosome nanovesicles for delivery of antibodies to treat inflammatory bowel disease
New hope for a stubborn gut disease
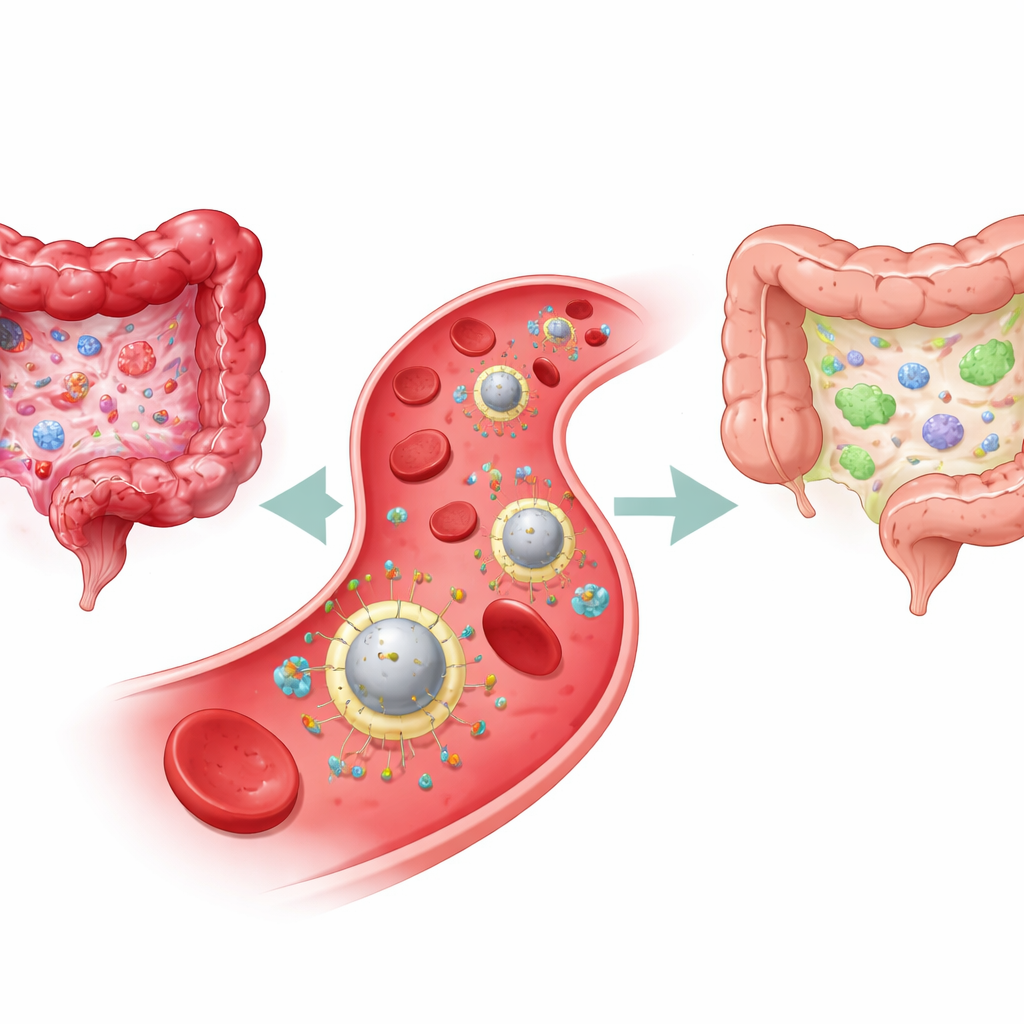
Inflammatory bowel disease (IBD) can turn everyday life into a constant battle with pain, diarrhea, and fatigue, and it also raises the long-term risk of colon cancer. Many current drugs calm flare‑ups but do not truly reset the faulty immune reactions driving the illness, so symptoms often return. This study introduces a cleverly engineered “smart” nanoparticle that travels through the bloodstream, finds inflamed areas of the intestine, and delivers a two‑part treatment designed both to dial down harmful inflammation and to rebuild lasting immune balance.
Why current treatments fall short
IBD, which includes Crohn’s disease and ulcerative colitis, stems from a chronic tug‑of‑war in the gut’s immune system. On one side are aggressive immune cells and inflammatory molecules that damage the intestinal lining; on the other are regulatory cells that normally keep this response in check. Standard therapies—such as steroids, immunosuppressants, and biologic drugs—mainly try to blunt inflammation everywhere in the body. They can ease symptoms, but they often do little to restore the underlying immune balance, and broad immune suppression can raise infection risks. Even newer antibody drugs that block a key inflammatory signal called interleukin‑23 (IL‑23) help only part of the patient population and still circulate widely, not just where they are needed.
Building a homing, shape‑shifting medicine
The researchers set out to design a treatment that would behave more like a living cell than a simple drug. They started with tiny bubbles naturally released by regulatory T cells—immune cells that promote tolerance and peacekeeping. These bubbles, called exosomes, carry proteins and genetic material that can nudge other immune cells into a calming mode. The team fused these exosomes with fragments of platelet membrane, borrowing platelets’ knack for sticking to damaged blood vessels in inflamed tissue. Onto the surface of this hybrid shell they attached IL‑23‑blocking antibodies using a molecular “string” that can be cut by enzymes (matrix metalloproteinases) that are abundant only in inflamed gut tissue. The result, named PrEXO‑a23, is a nanovesicle that circulates in the blood, latches onto injured intestinal vessels, and then sheds its antibody payload only where disease is active.
Re‑educating the gut’s immune neighborhood
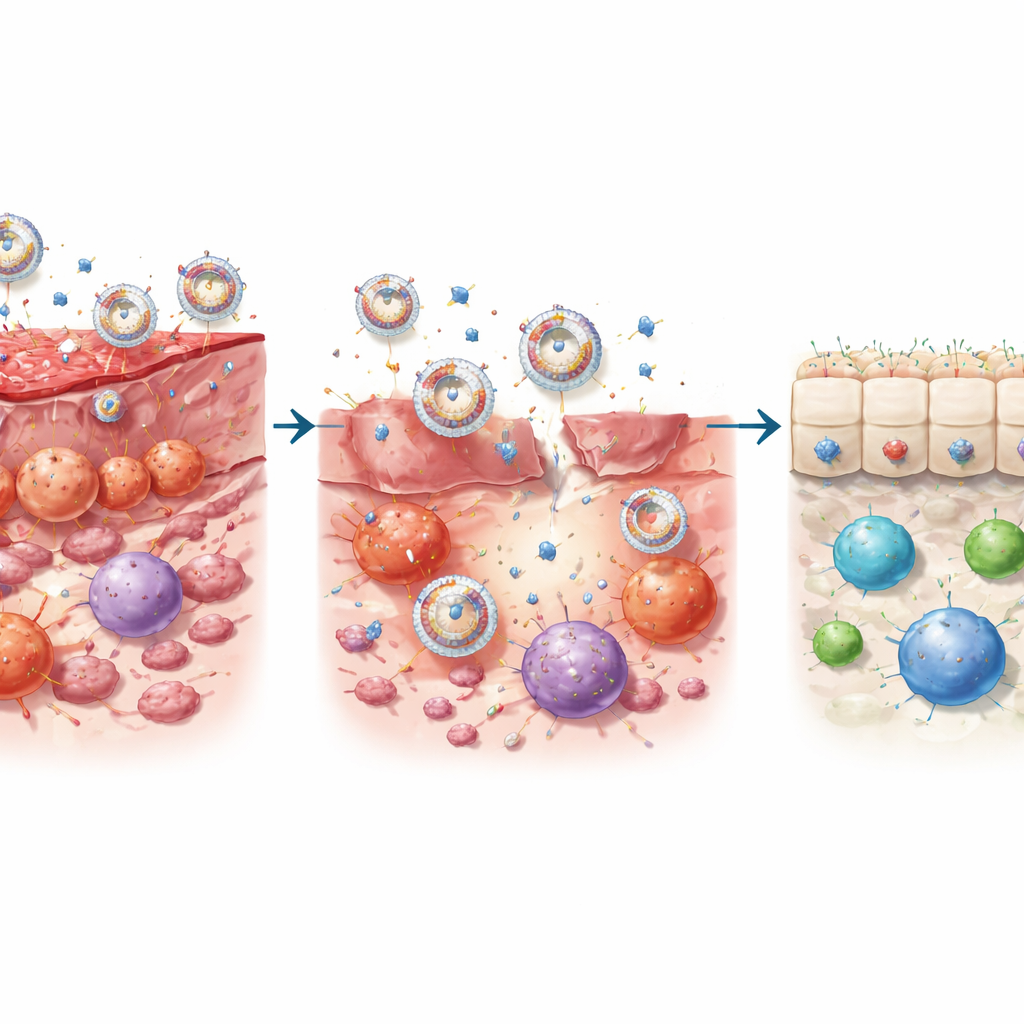
In cell studies, PrEXO‑a23 outperformed either the antibody or the exosomes alone. It strongly reduced the formation of inflammatory T‑helper 17 (Th17) cells and the release of damaging cytokines that injure the intestinal lining, while protecting human gut epithelial cells from death. At the same time, it pushed dendritic cells—gatekeepers that instruct T cells—away from a hostile, activating state toward a “tolerant” state that encourages the growth of regulatory T cells. In mouse models of colitis, the nanovesicles homed efficiently to inflamed colon tissue, were taken up by multiple immune cell types, and released antibodies in response to local enzyme activity. This dual action—blocking IL‑23–driven inflammation and boosting tolerance‑promoting signals—brought inflammatory markers close to normal and restored a healthier mix of T‑cell types.
Repairing tissue and preventing long‑term damage
Mice with chemically induced colitis that received PrEXO‑a23 lost less weight, had longer, healthier colons, and showed less spleen enlargement, a sign of systemic inflammation. Microscopic examination revealed preserved intestinal architecture and tighter junctions between epithelial cells, indicating a repaired barrier. The treatment also helped rebalance gut bacteria, increasing families associated with gut health and reducing those linked to disease. In a more severe model that mimics the progression from chronic colitis to colon cancer, PrEXO‑a23 not only eased ongoing inflammation but completely prevented the development of tumors and curbed the thickening and scarring of the intestinal wall that characterize fibrosis. Molecular analyses pointed to restoration of protective p53 signaling—a central guardian against cancer—which proved important for the anti‑cancer effect.
Toward smarter, longer‑lasting IBD care
To a non‑specialist, the key message is that this work moves beyond simply “turning down” inflammation. By combining targeted delivery, on‑site antibody release, and immune‑soothing exosome cargo in one tiny package, PrEXO‑a23 both calms active disease and helps retrain the immune system toward lasting tolerance. In mice, this led not only to symptom relief but also to protection from scarring and colon cancer, two of the most feared complications of long‑standing IBD. While much testing remains before such a therapy could reach patients, the study showcases how biologically inspired nanomedicines might one day offer more precise, durable control over complex immune disorders like IBD.
Citation: Cao, J., Luo, R., Miao, R. et al. Engineered exosome nanovesicles for delivery of antibodies to treat inflammatory bowel disease. Nat Commun 17, 2737 (2026). https://doi.org/10.1038/s41467-026-69382-4
Keywords: inflammatory bowel disease, nanomedicine, immune tolerance, exosomes, colorectal cancer prevention