Clear Sky Science · en
Chemoenzymatic synthesis of pentalenolactones via stereoselective Riley oxidation by engineered P450BM3
Why this matters for future medicines
Many of today’s medicines are inspired by nature, but copying nature’s intricate molecular shapes in the lab is slow, wasteful, and often relies on harsh chemicals. This study shows how reprogrammed enzymes—nature’s own catalysts—can be teamed with classic chemistry to build complex, antibiotic-like molecules more cleanly and efficiently. For readers, it offers a glimpse of how greener chemistry could speed the discovery of new drugs.
Nature’s knotted building blocks
Some promising drug candidates share a compact, knotted carbon framework known as a cis-diquinane core. Molecules with this shape, including the pentalenolactone family of natural antibiotics, can shut down a key enzyme that bacteria need to make energy. Chemists have long wanted simple, scalable ways to make these cores with precise 3D shapes, but traditional methods require many steps and often struggle to control “handedness,” the mirror-image character that can make or break a drug’s activity.
Turning a blunt reaction into a precise tool
One well-known way to add oxygen atoms to molecules is a reaction class called Riley oxidation. In its classic form, it uses toxic selenium-based reagents and gives mixtures of mirror-image products, limiting its usefulness for building complex drugs. The authors set out to turn this blunt chemical tool into a highly selective one by putting an engineered enzyme in charge. They began with an easily made, perfectly symmetrical cis-diquinane starting material and asked whether an enzyme could “desymmetrize” it—attacking only one side to create a single, chiral product.
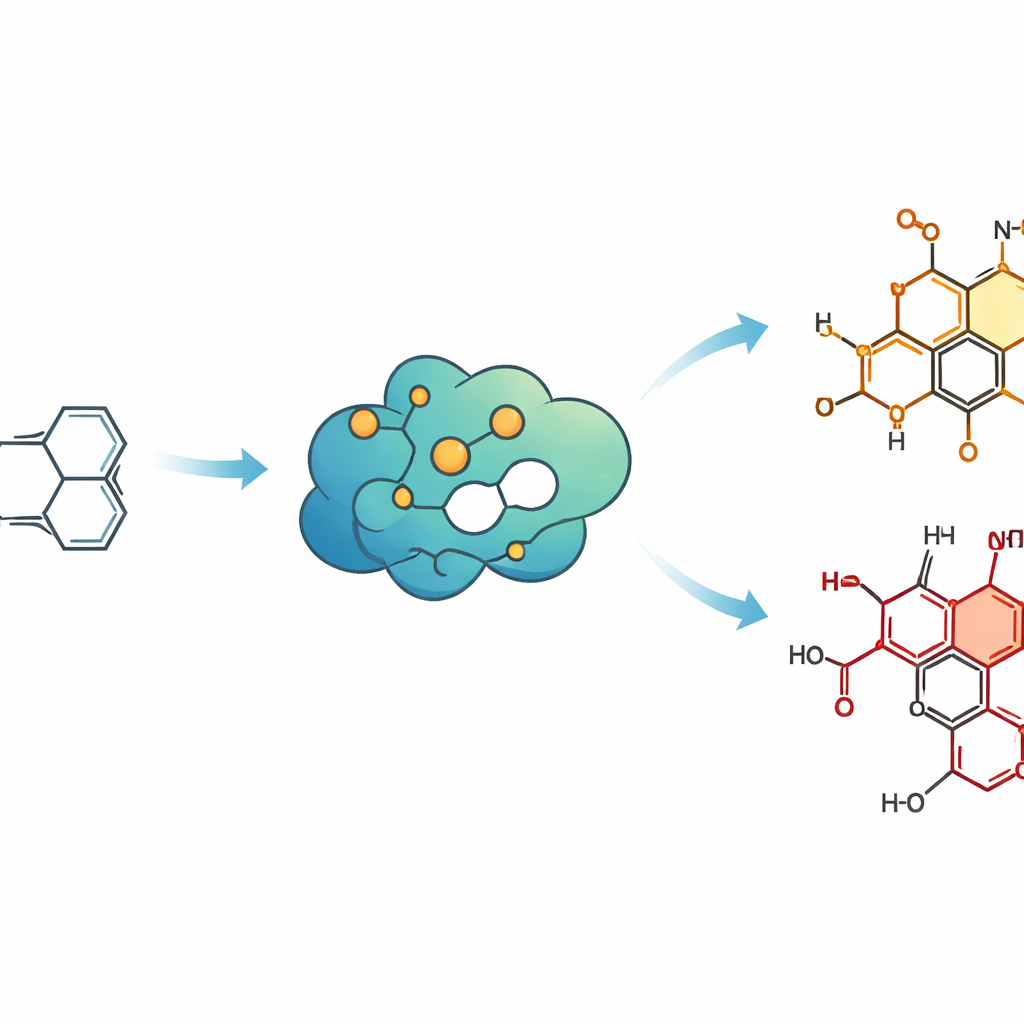
Reprogramming an enzyme as a molecular sculptor
The team screened a library of oxidative enzymes and found that a bacterial enzyme called P450BM3 could perform the desired transformation, but only modestly well. Using protein engineering and directed evolution—cycles of targeted mutation followed by testing—they reshaped the enzyme’s active pocket so it cradled the cis-diquinane in just the right orientation. Step by step, they introduced mutations that sharpened its control over where and how oxygen was added. The final variant, named AAO4, produced the desired oxidized cis-diquinane in gram quantities with excellent control over 3D structure, effectively turning a messy chemical oxidation into a precise, enzyme-guided cut.
Building antibiotics by mixing biology and chemistry
With this chiral building block in hand, the researchers wove together standard organic reactions and additional enzymes to reach two target molecules: pentalenolactone D and neo-pentalenolactone D. Chemical steps stitched the oxidized cis-diquinane into a more elaborate, three-ring framework called pentalenene and then into 1-deoxypentalenic acid, a form that natural biosynthetic enzymes recognize. The team then borrowed enzymes from the microbe that originally makes pentalenolactones. One enzyme carried out a highly selective late-stage hydroxylation, and a second enzyme class performed a Baeyer–Villiger oxidation, gently reshaping a ring to deliver either of the two final antibiotic-like products, depending on which enzyme was used.
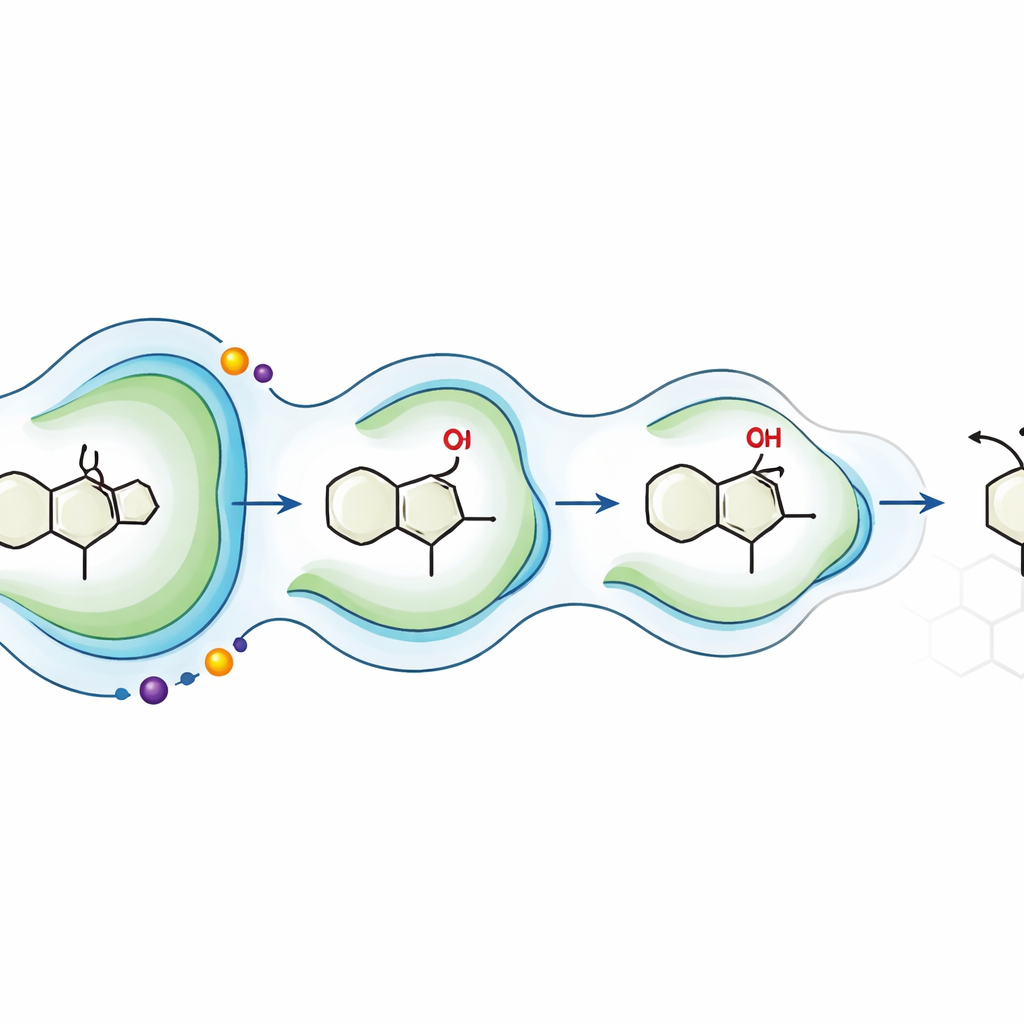
A new playbook for greener complex molecules
This work demonstrates a powerful new strategy: start from a simple, symmetric scaffold; use an engineered enzyme to introduce 3D information in one decisive step; then combine classic chemistry with borrowed biosynthetic enzymes to finish the job. In plain terms, the authors converted a once-harsh, poorly controlled oxidation into a clean, selective, and scalable transformation, enabling streamlined access to complex natural-product-like antibiotics. Their approach suggests that future drug synthesis may rely less on toxic reagents and lengthy routes, and more on carefully tuned enzymes that act as programmable molecular sculptors.
Citation: Xu, Y., Zhang, K., Lv, Q. et al. Chemoenzymatic synthesis of pentalenolactones via stereoselective Riley oxidation by engineered P450BM3. Nat Commun 17, 2569 (2026). https://doi.org/10.1038/s41467-026-69381-5
Keywords: chemoenzymatic synthesis, engineered enzymes, Riley oxidation, pentalenolactone, biocatalysis