Clear Sky Science · en
Ultrafast solvent-modulated roaming mechanism in bromoform revealed by femtosecond X-ray solution scattering
Why tiny molecules in water and air matter
Bromoform is a small, naturally produced chemical that carries a big environmental punch. When sunlight hits this molecule in the atmosphere or ocean spray, it can release bromine atoms that help destroy ozone, the shield that protects us from the Sun’s harmful ultraviolet rays. This study asks a deceptively simple question with far‑reaching consequences: does the liquid environment around bromoform change what happens in the first trillionths of a second after sunlight strikes, and therefore change how much ozone‑eating bromine ultimately appears?
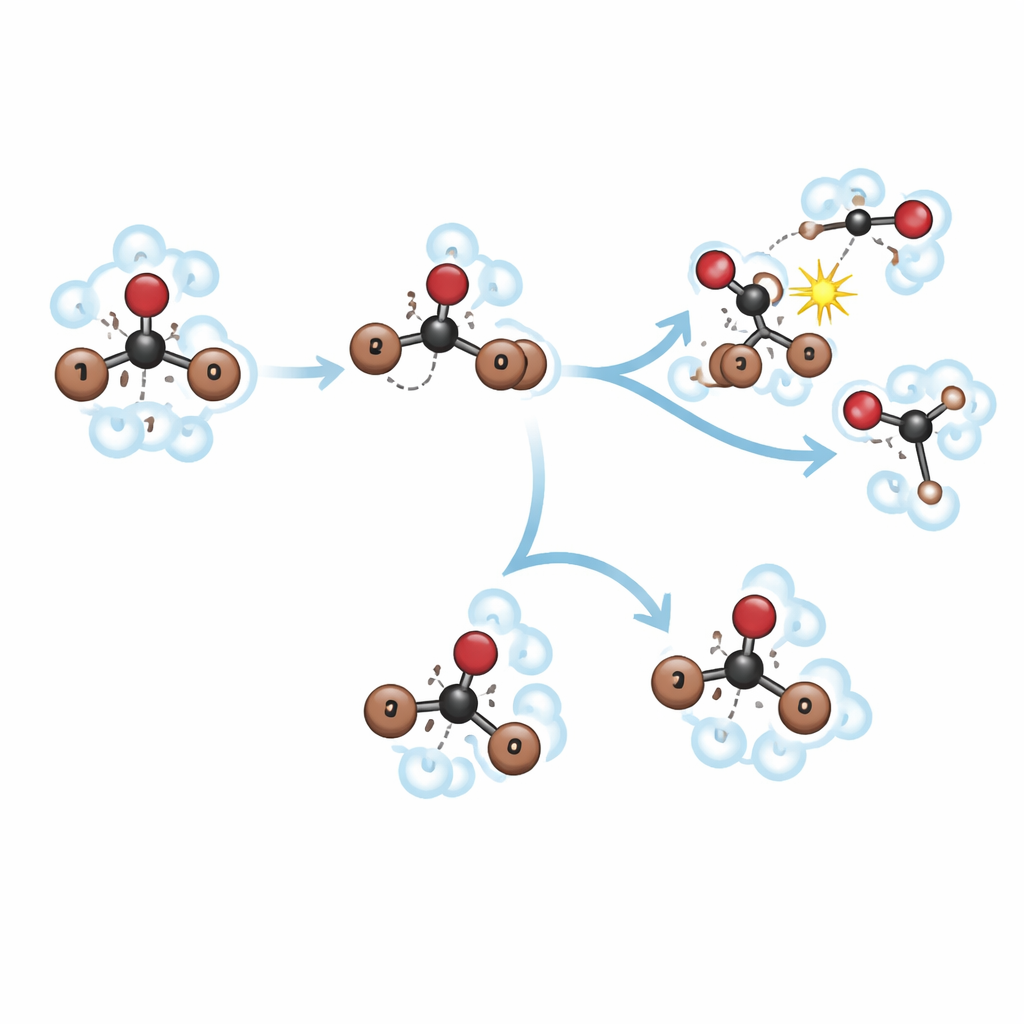
Light that snaps a molecule apart
The researchers start by shining extremely short bursts of ultraviolet light on bromoform dissolved in two different liquids: methanol, which is polar and chemically reactive, and methylcyclohexane, which is non‑polar and relatively inert. The light quickly weakens one of bromoform’s carbon–bromine bonds, causing the molecule to begin to fall apart. But instead of simply flying away, one bromine atom lingers and “roams” around the remaining piece of the molecule, CHBr₂. This roaming motion creates a fleeting, reshaped form called an isomer. The central puzzle is what happens to this roaming intermediate in different liquids, and how that choice controls whether bromoform releases free bromine atoms or is diverted into other products.
Taking molecular movies with X‑rays
To watch these events unfold, the team uses femtosecond time‑resolved X‑ray solution scattering at the European X‑ray Free‑electron Laser. In essence, they record a strobe‑light movie of how inter‑atomic distances change in real time, with both picosecond and sub‑picosecond resolution. By comparing the measured scattering patterns with computer‑generated patterns for many possible structures, they reconstruct how bond lengths and angles within bromoform evolve after excitation. Advanced analysis techniques and machine‑learning‑accelerated simulations help them separate overlapping reaction pathways and assign precise lifetimes to short‑lived species.
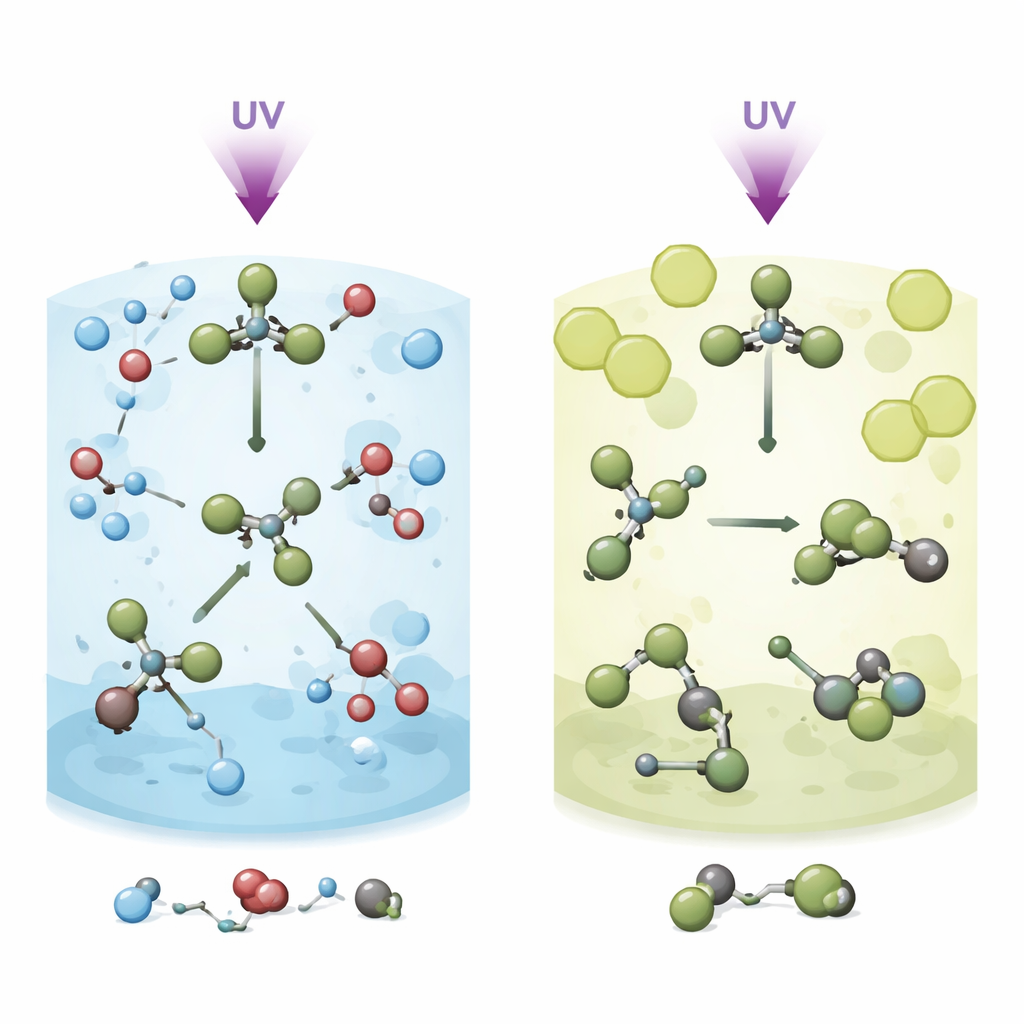
Two liquids, two very different fates
In methanol, the roaming intermediate is present but short‑lived. Within roughly 150 femtoseconds, the initial carbon–bromine bond breaks, forming CHBr₂ and a roaming bromine. Over the next ~400 femtoseconds, this hot, vibrating isomer repeatedly stretches and bends, which brings the fragments into frequent contact with nearby methanol molecules. Instead of relaxing into a stable rearranged form, the roaming structure is quickly intercepted by methanol in a “methanolysis” reaction. This produces new molecules, CH₃OCHBr₂ and HBr, while free CHBr₂ and Br fragments from a parallel, more direct breakup pathway continue to react more slowly over several picoseconds. The solvent’s tight molecular “cage” and its reactive hydroxyl groups work together to steer bromoform down this chemical‑reaction route rather than toward a long‑lived isomer.
When the liquid steps back and lets roaming go on
In methylcyclohexane, the story changes dramatically. The same ultraviolet kick again creates roaming motion within about 150 femtoseconds, but the non‑polar liquid surrounds bromoform more loosely and does not readily react with it. Here, the roaming bromine and CHBr₂ piece have room to reorient and settle into a stable isomeric structure instead of immediately engaging with solvent molecules. Direct breakup into separate CHBr₂ and Br fragments still occurs and competes with isomer formation, but now the rearranged isomer survives for much longer times. Simulations show that the larger, softer solvent cage in methylcyclohexane encourages this gas‑like roaming behavior, while the cramped, strongly interacting cage in methanol channels the same intermediate straight into chemical reaction.
What this means for ozone and beyond
Together, these experiments and calculations reveal that roaming is a universal early step when bromoform absorbs ultraviolet light in liquid environments, but the surrounding solvent decides what happens next. In methanol‑like settings, roaming mainly serves to drive very fast reactions with the liquid, limiting the formation of long‑lived isomers and shaping how and when bromine‑bearing products appear. In methylcyclohexane‑like environments, roaming instead feeds a long‑lived rearranged form that can later release bromine on different timescales. By directly filming these ultrafast motions, the work shows that the microscopic character of droplets, aerosols, and other condensed‑phase environments can strongly control the chemical pathways of ozone‑relevant molecules, helping scientists build more accurate models of atmospheric and solution‑phase photochemistry.
Citation: Su, P., Zhang, J., Wang, H. et al. Ultrafast solvent-modulated roaming mechanism in bromoform revealed by femtosecond X-ray solution scattering. Nat Commun 17, 2514 (2026). https://doi.org/10.1038/s41467-026-69374-4
Keywords: bromoform, roaming mechanism, ultrafast photochemistry, solvent effects, ozone depletion