Clear Sky Science · en
Genetic regulation of methylation across East Asian and European populations
Why our DNA’s chemical tags matter
Our genes do not work alone. They are covered with tiny chemical tags that help switch them on or off, shaping everything from cholesterol levels to risk of heart disease or diabetes. This study asks a simple but crucial question: do these tags—and the genetic switches that control them—behave the same way in people of different ancestries, or are there important differences that affect how we understand disease risk around the world?
Looking at DNA switches in diverse peoples
The researchers focused on a particular chemical tag called DNA methylation, which often acts like a dimmer switch on genes. They studied “methylation quantitative trait loci” (mQTLs)—genetic variants that change how much methylation appears at nearby spots in the genome. Past work had mostly examined people of European ancestry, leaving big gaps in our knowledge of other populations. Here, the team assembled the largest dataset so far in East Asians, analyzing blood samples from 7,619 Han Chinese volunteers and comparing results with 27,750 Europeans.
Finding new control points in the genome
By combining three East Asian studies and using a newer, more comprehensive laboratory array, the scientists mapped over 330,000 DNA sites whose methylation levels are shaped by nearby genetic variants. Nearly 29,000 of these methylation sites had never been linked to genetic control before, and many fell in regions of the genome that help regulate when genes turn on, such as enhancer regions. When the team further merged the East Asian and European data, they discovered even more previously unknown control sites, including ones in specialized regions that manage parent-of-origin effects and in thousands of binding sites for key DNA‑binding proteins.
Shared patterns and population‑specific signals
When the same methylation sites could be measured in both East Asian and European samples, most of the underlying genetic effects looked strikingly similar, suggesting that much of the basic control system is shared across ancestries. However, the study also uncovered many signals that were strong in East Asians but weak or invisible in Europeans. A major reason was frequency: some risk variants are relatively common in East Asians but rare in Europeans, making them far easier to detect in East Asian data even when European studies include more people. These population‑specific switches expand our understanding of how gene regulation can differ across human groups.
Linking chemical tags to real diseases
The authors then asked how much these methylation‑linked variants matter for real‑world traits. Using large genetic studies of dozens of diseases and health measurements from BioBank Japan and other East Asian projects, they found that mQTLs explain more of the inherited risk for many conditions than most other types of functional DNA annotations. Importantly, mQTLs mapped in East Asians did a better job of capturing inherited risk in East Asian disease studies than mQTLs mapped only in Europeans, showing that ancestry‑matched resources sharpen our view of disease biology.
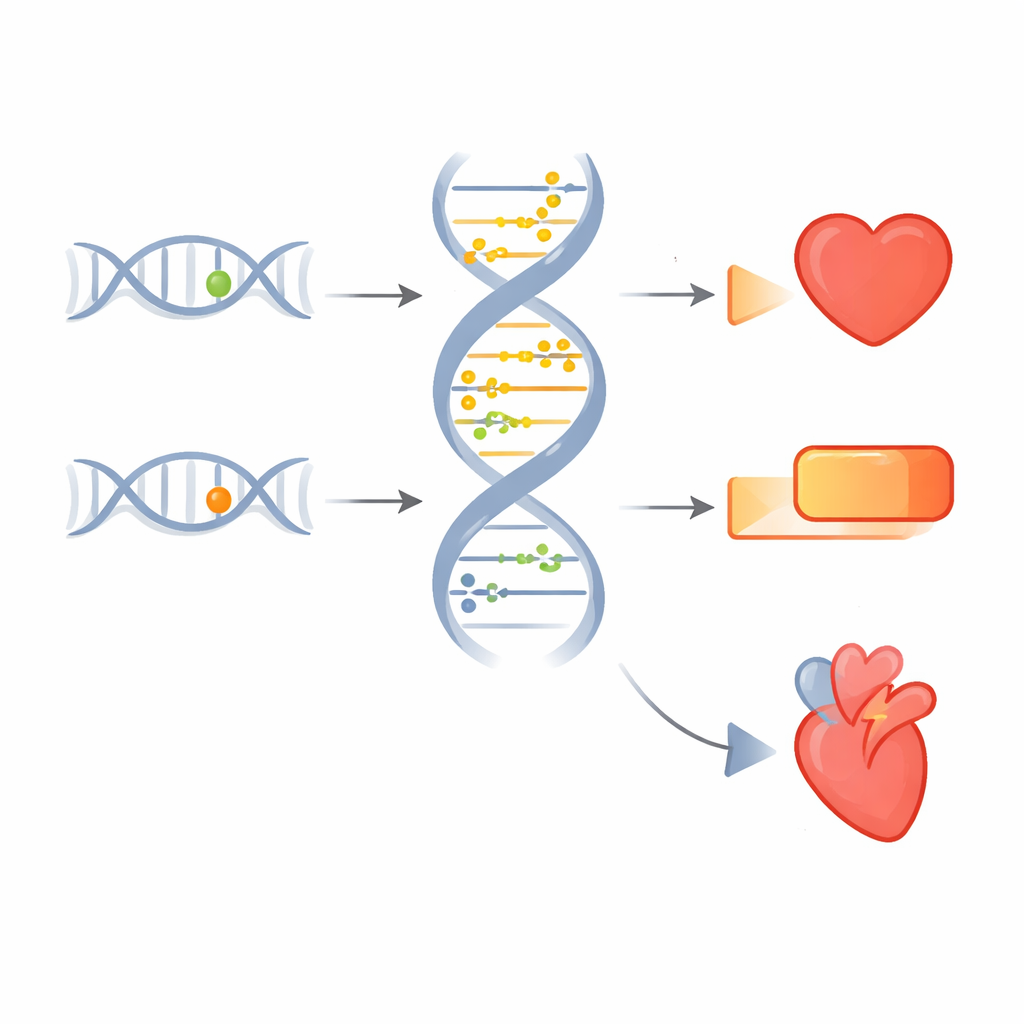
From single variants to heart disease and diabetes
To move from broad patterns to concrete stories, the team zoomed in on individual sites where the same genetic variant seemed to influence both methylation and disease. One example sits near the gene TCF21, which helps shape blood vessel cells. A particular DNA change in this region lowers methylation at a nearby site, boosts TCF21 activity in heart tissue, and is associated with lower risk of several forms of coronary artery disease and with use of heart medications. Another set of sites near the CAMK1D gene showed coordinated effects on blood sugar traits, diabetes risk, and the need for diabetes drugs, suggesting a route by which genetic differences tune metabolic health through methylation changes.
What this work means for health
Together, these findings show that many of the genetic levers controlling our DNA’s chemical tags are shared across populations, but some are much more visible—and impactful—in certain ancestries. Because these levers help connect genetic variation to real diseases, mapping them in diverse groups is essential for fair and accurate medical insight. For everyday readers, the message is that ancestry‑aware genetics and epigenetics can improve how we explain why some people get conditions like heart disease or diabetes and may eventually guide more tailored prevention and treatment strategies.
Citation: Liu, R., Chen, TT., Xia, Y. et al. Genetic regulation of methylation across East Asian and European populations. Nat Commun 17, 2616 (2026). https://doi.org/10.1038/s41467-026-69372-6
Keywords: DNA methylation, genetic regulation, East Asian populations, complex disease risk, epigenetics