Clear Sky Science · en
Fidelity in co-diversified symbiosis
Why beetles and their microbes matter
Many animals rely on hidden microbial partners to eat, grow, and reproduce. This study looks at a striking example: tortoise beetles that depend on a tiny bacterium, Stammera capleta, to digest tough plant leaves. By experimentally swapping these bacteria between beetle species, the researchers show how evolution has fine‑tuned who can partner with whom, and why some alliances stay faithful over tens of millions of years.
A tiny helper that unlocks a leafy diet
Tortoise beetles feed on leaves that are rich in plant cell walls, materials animals generally struggle to break down. Inside specialized chambers at the front of the beetle gut, and in glands near the ovaries, lives S. capleta, a bacterium with an extremely small genome that still retains genes for enzymes that digest plant cell walls. Mother beetles package these bacteria into small caps, or “caplets,” at the front end of each egg. As embryos near hatching, they pierce the caplet and consume bacterial spheres, seeding their own gut organs with the symbiont. If the caplet is removed, larvae hatch without bacteria, cannot properly digest leaves, and rarely survive to adulthood.
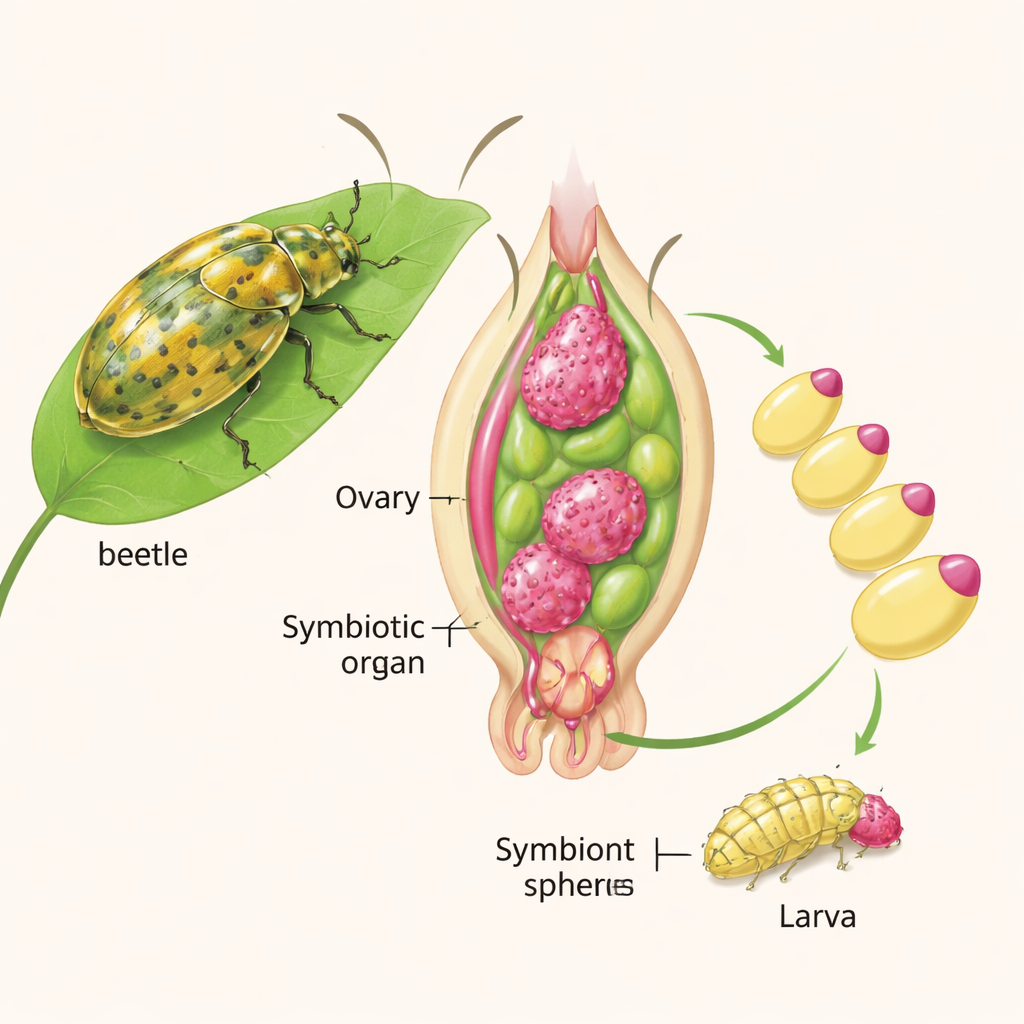
Swapping partners across beetle species
The team took advantage of this external transmission route to ask how specific the partnership really is. Working with six tortoise beetle species, each carrying its own strain of S. capleta, they surgically removed caplets from eggs of a focal species, Chelymorpha alternans, and reapplied bacterial spheres from either the same species or from other beetles. Imaging and DNA tests showed that bacteria from all donor species could successfully colonize the gut chambers of C. alternans larvae. In the short term, then, even a “foreign” symbiont can move into the right place in a new host.
A spectrum from full rescue to partial help
Colonization, however, was not the whole story. The authors measured how many bacteria were present and how well the larvae survived to adulthood. When C. alternans received its own symbiont back, survival matched that of untreated controls. Some closely related bacteria from beetle cousins also grew well and fully restored survival. More distantly related symbionts did colonize, but often reached lower numbers and only partly rescued survival: more larvae made it to adulthood than with no symbiont at all, but fewer than with the native partner. Across strains, host survival rose in step with how genetically similar each symbiont’s genome was to the native one, highlighting a tight link between evolutionary relatedness and current performance.
How the host distinguishes friends from near‑strangers
To see what happens inside the beetle during these mismatched alliances, the researchers examined gene activity in the gut organs. When larvae hosted a foreign symbiont that behaved almost like the native one, their own gene expression was nearly unchanged. In contrast, a genetically distant symbiont that only partly rescued survival triggered a much stronger host response, including the activation of immune‑related genes that in other insects help recognize and attack microbes. This distant symbiont also reached much lower abundance, suggesting that the host’s immune system and metabolism may curb its growth or fail to provide the conditions it needs.
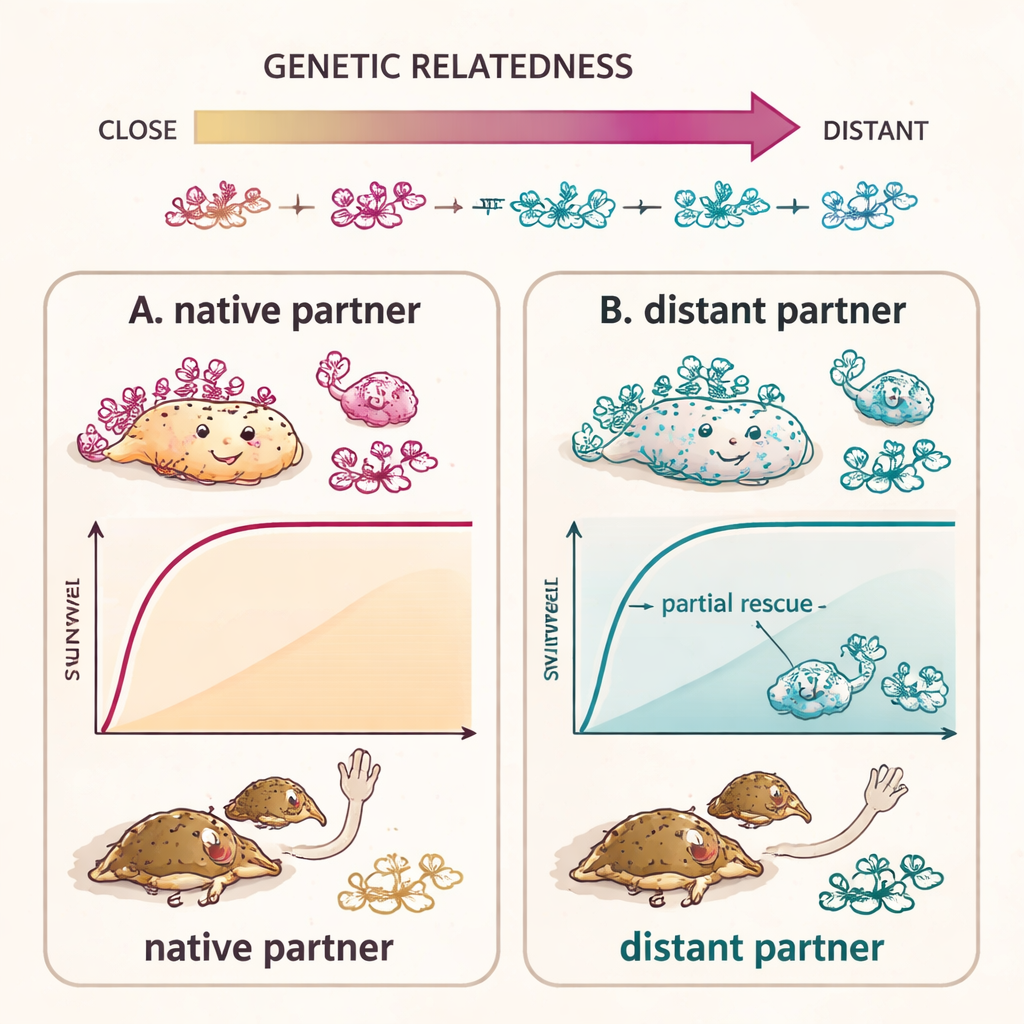
Faithful transmission and quiet competition
The story extends into adulthood and the next generation. Non‑native symbionts that fully rescued larval survival could persist through metamorphosis in adult gut organs and even reach the ovary‑associated glands. Yet they failed at the final step: they were not packaged into the caplets on eggs, leaving the offspring entirely bacteria‑free. In mixed infections where eggs received both native and closely related non‑native bacteria, both strains initially coexisted in larvae, but over time the native symbiont consistently outgrew its rival and alone colonized adult organs and offspring. This shows that, even when foreign strains can function reasonably well, subtle advantages in growth, recognition, and packaging allow the native partner to win.
What this means for long‑term partnerships
To a lay reader, the main message is that long‑term, inherited partnerships between animals and microbes are held together by several reinforcing filters: the host’s ability to recognize familiar partners, the precise way bacteria are passed from mother to offspring, and competition that favors locally adapted strains. In tortoise beetles, these layers ensure that, despite occasional flexibility, the same bacterial partners are kept in the family line, preserving a finely tuned collaboration that began tens of millions of years ago.
Citation: Pons, I., García-Lozano, M., Emmerich, C. et al. Fidelity in co-diversified symbiosis. Nat Commun 17, 1644 (2026). https://doi.org/10.1038/s41467-026-69366-4
Keywords: symbiosis, gut microbiome, insect-bacteria partnership, vertical transmission, host-microbe coevolution