Clear Sky Science · en
Quasi-one-dimensional enaminone-linked covalent organic frameworks for efficient CO₂ photoreduction
Turning Greenhouse Gas into a Useful Fuel
Carbon dioxide from burning fossil fuels is the main driver of climate change, yet it is also a cheap, abundant raw material. Scientists are racing to develop “artificial leaves” that can use sunlight to turn CO₂ and water into useful chemicals, much as plants do. This paper reports a new kind of designer material that does this job far more efficiently than earlier versions, bringing clean solar fuels a step closer to reality.
A New Kind of Tiny Scaffold
At the heart of this work are covalent organic frameworks, or COFs—crystal-like networks built from light elements such as carbon, nitrogen, and oxygen. They are full of tiny, orderly pores and can be chemically tuned almost like Lego. The authors focus on a special subtype called quasi‑one‑dimensional COFs, where the building blocks line up into dual chain–like strands. This architecture exposes many reactive “edge” sites and channels electrons in a preferred direction, both of which are good for harvesting light and driving chemical reactions. However, most previously reported versions relied on a common chemical linkage that is only moderately stable under strong illumination, limiting their usefulness in photocatalysis.
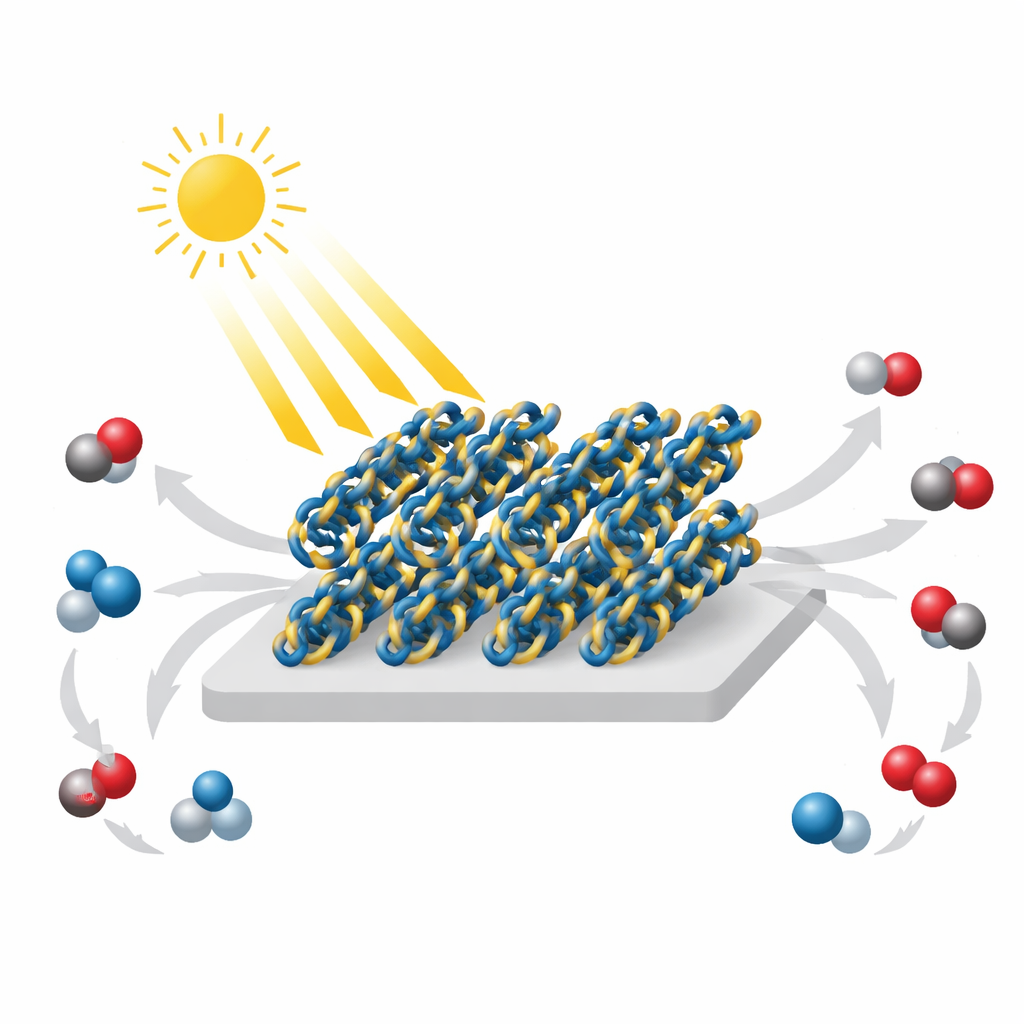
Designing a Better Light-Harvesting Backbone
To overcome this bottleneck, the team swapped the usual linkage for a different one known as an enaminone, which carries a stronger internal electrical polarity. They built three closely related materials: one using only the traditional imine linkage, one mixing both types, and one using only the enaminone connections, dubbed En‑Q1DCOF. Careful structural measurements, including X‑ray diffraction and electron microscopy, showed that all three form well‑ordered, stable frameworks with nanosheet shapes and pores of about 1.5 nanometers across. Optical tests revealed that En‑Q1DCOF absorbs visible light more strongly and has a slightly smaller energy gap between its filled and empty electronic states, giving excited electrons more freedom to move.
From Sunlight, CO₂, and Water to Carbon Monoxide
The researchers then tested how well these materials could drive the conversion of CO₂ and water vapor into carbon monoxide (CO) and oxygen (O₂) under visible light, without adding metals, sacrificial chemicals, or extra dyes. Here, En‑Q1DCOF clearly stood out: over 24 hours it produced 3045 micromoles of CO per gram of catalyst—about seven times more than the mixed-linker COF and twelve times more than the imine-only version—while maintaining nearly 100% selectivity for CO over other carbon products. Isotope-labeling experiments using heavy forms of carbon and oxygen confirmed that the detected CO and O₂ came from the supplied CO₂ and water, not from the breakdown of the material itself. The enaminone-based framework also remained structurally and chemically intact after repeated runs and after soaking in acidic, basic, or solvent-rich environments.
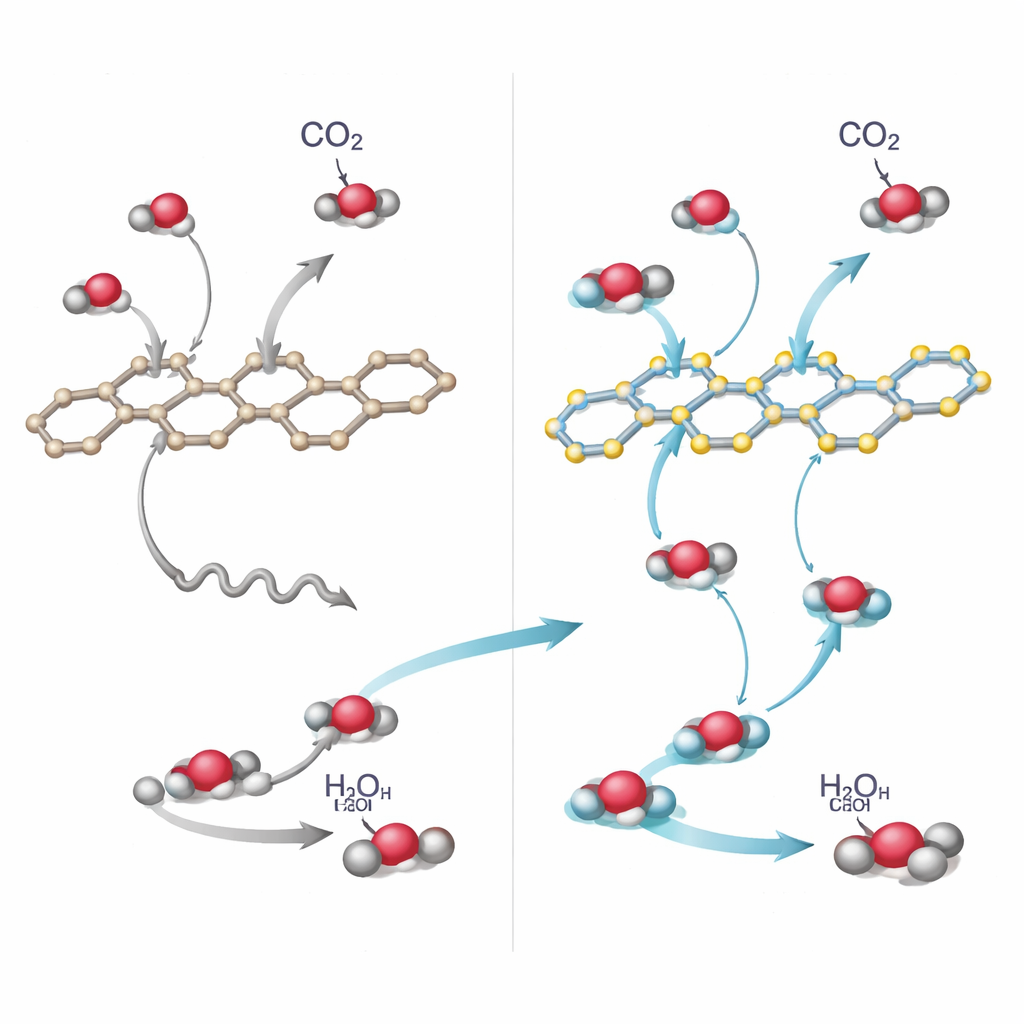
How Polar Bonds and Hidden Hydrogen Help
Why does the enaminone version work so much better? A combination of experiments and quantum-chemical calculations paints a detailed picture. The polar enaminone bonds create small internal electric fields that help pull apart the bound electron–hole pairs formed when the material absorbs light. As a result, charge carriers survive long enough to reach reactive sites instead of simply recombining and wasting the absorbed energy. Electrical measurements show that En‑Q1DCOF conducts photogenerated charges more efficiently and exhibits lower resistance at its interfaces. Subtle photoluminescence and ultrafast spectroscopy studies reveal that excited states in this material decay in ways that favor charge transfer rather than glow, another sign of efficient charge separation.
Guiding CO₂ Along an Easier Path
The chemistry at the surface also changes. Infrared experiments tracking molecules in real time show that CO₂ binds strongly to En‑Q1DCOF and forms key intermediates, such as a bent COOH species, more readily than on the comparison materials. Calculations support this, indicating that the oxygen-rich part of the enaminone unit carries extra negative charge and that the hydrogen attached to its nitrogen can form a stabilizing hydrogen bond with incoming CO₂. This interaction both anchors and weakens the CO₂ molecule, lowering the energy hurdle for the hardest reaction step—converting adsorbed CO₂ into the COOH intermediate on the way to CO. At the same time, the framework helps pull electrons from water to generate oxygen, closing the overall “artificial photosynthesis” cycle.
Bringing Artificial Leaves Closer to Practice
In simple terms, the authors have built a finely tuned, sponge-like crystal that soaks up light, grabs CO₂ molecules, and shuttles charges in just the right way to turn a climate-warming gas into a useful fuel component. By showing that enaminone linkages in quasi‑one‑dimensional frameworks dramatically boost performance without relying on precious metals, this work opens a new design path for solar-powered reactors. With further optimization, such materials could underpin future devices that quietly scrub CO₂ from the air while producing building blocks for cleaner fuels and chemicals.
Citation: Bai, J., Hu, Y., Si, F. et al. Quasi-one-dimensional enaminone-linked covalent organic frameworks for efficient CO₂ photoreduction. Nat Commun 17, 2158 (2026). https://doi.org/10.1038/s41467-026-69361-9
Keywords: CO2 photoreduction, covalent organic frameworks, artificial photosynthesis, solar fuels, porous photocatalysts