Clear Sky Science · en
Orientation-dependent mutual crystalline and amorphous order in a single phase solid
When Order and Disorder Live Side by Side
Most of the materials around us fall into two neat categories: crystals, where atoms line up in repeating patterns like tiles on a floor, and glasses, where atoms are jumbled like frozen liquid. This study reveals something surprising in between: a solid that is glass-like in two directions but crystal-like in the third. That unusual mix of order and disorder could change how we think about everyday materials, from batteries to computer chips, and how we design them from the atomic level up. 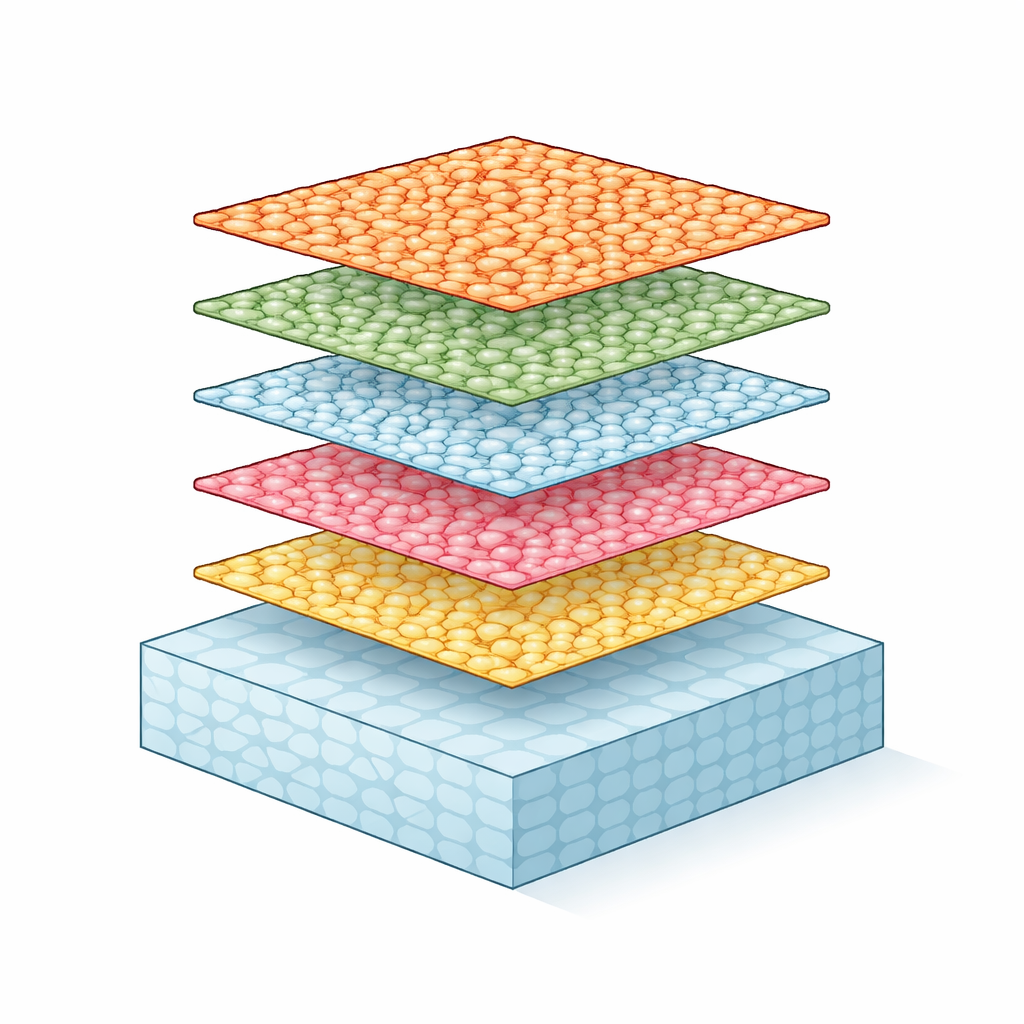
A New Kind of Atomic Jigsaw
Crystals are defined by their long-range order: if you know where a few atoms sit, you can predict where many more will be. Amorphous materials, like window glass, lack this repeating order, even though atoms still keep some regular spacing with their closest neighbors. For decades, scientists have debated how to describe the “in-between” zone of medium-range order, where the pattern extends a few atoms out but not to infinity. The authors of this paper take a different angle: instead of asking whether an entire material is ordered or disordered, they ask whether different directions inside the same solid can behave differently.
Layered Rods with a Hidden Pattern
The team created thin films made of tiny rods containing niobium, tungsten, and oxygen (Nb–W–O) using pulsed laser deposition, a technique that fires short bursts of energy at a ceramic target to build up material on a crystal surface. By choosing a well-known crystalline base material, strontium titanate, cut along different faces, they could control how the Nb–W–O rods grew. Electron microscope images showed that inside each rod, atoms within a single layer lie in a disordered, glass-like arrangement across the plane. Yet when the researchers looked along the perpendicular direction, they saw that these disordered layers were stacked at nearly perfect, regular spacings over hundreds of atomic layers: a crystal built from glassy sheets. 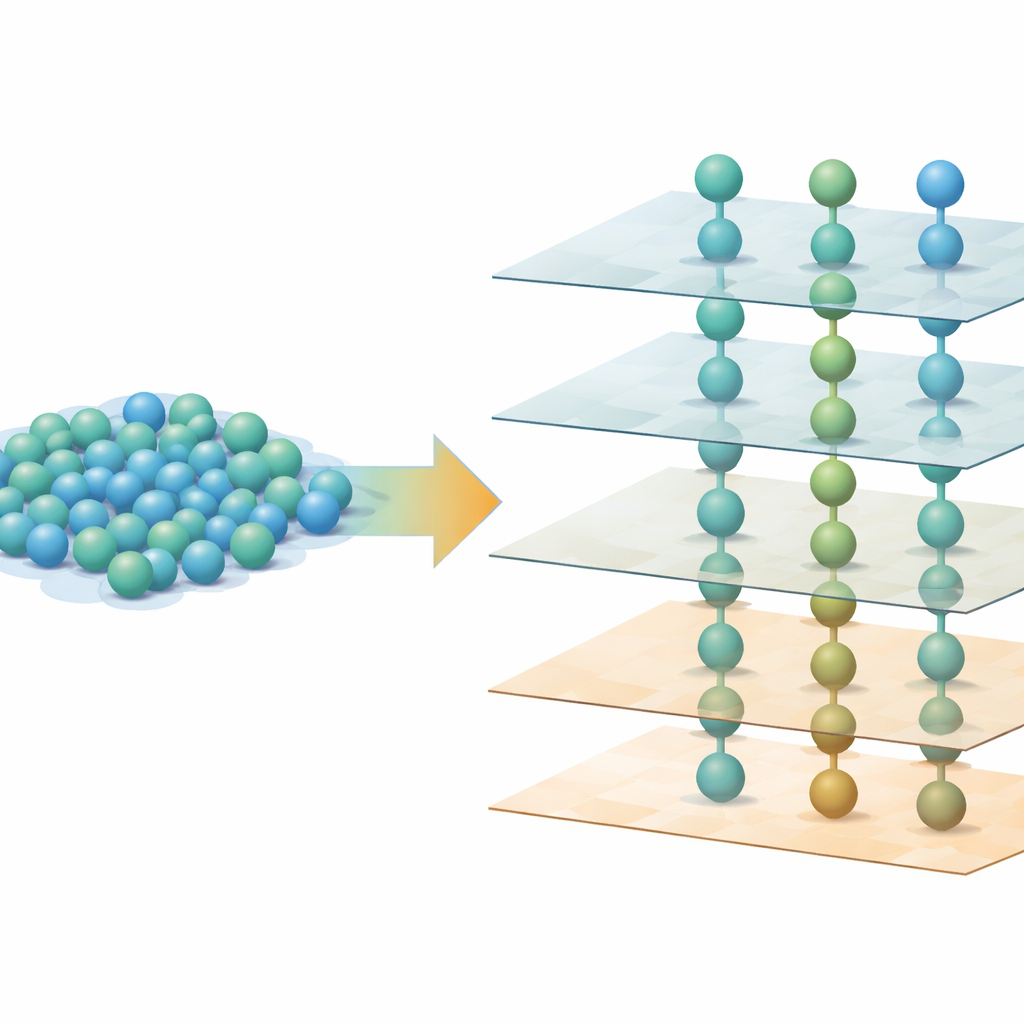
Seeing Randomness up Close
To test how random the layers really were, the researchers combined several powerful probes. High-resolution electron microscopy images, along with their Fourier transforms, showed no repeating pattern in the plane of each layer, confirming the lack of long-range order. Pair distribution function measurements, which chart typical distances between atoms, revealed sharp peaks only at very short distances, indicating that atoms still form basic building blocks—octahedral units where a metal atom is surrounded by oxygen atoms—but that any larger repeating pattern quickly fades away. Advanced X-ray absorption techniques confirmed that niobium and tungsten sit in these distorted octahedra, while chemical maps showed that niobium and tungsten atoms are mixed without a regular pattern across the layer.
Stacked Glass Sheets that Behave Like a Crystal
Although each layer is structurally disordered in its own plane, their vertical stacking is anything but random. Three-dimensional reciprocal space mapping with synchrotron X-rays, a method that turns scattering patterns into a kind of fingerprint of atomic order, revealed sheet-like features that match simulations of periodically stacked amorphous layers. Depending on how the underlying strontium titanate is oriented, the rods grow in one, two, or three preferred directions, but in all cases the spacing between the layers is nearly the same and strongly linked to the spacing of the substrate crystal. In other words, the base crystal acts as a rigid ruler, forcing the glass-like layers to pile up with crystalline regularity along one principal axis, even though they remain disordered sideways.
Why This Borderland Matters
This unusual material shows that the usual dividing line between crystal and glass is not just about how far order extends, but also about which directions you look along. Within a single solid, atoms can form a continuous random network in two dimensions while still arranging themselves in a perfect rhythm in the third. That insight gives scientists a new playground for tuning properties: one can imagine materials where electrical conduction, ion movement, or mechanical strength are highly directional because order and disorder coexist in a controlled way. Beyond this particular niobium–tungsten–oxide, the work offers a platform for exploring and modeling stacks of two-dimensional amorphous matter, helping refine how we describe, measure, and ultimately engineer solids that sit between the familiar worlds of crystals and glasses.
Citation: Xia, R., Li, J., Birkhölzer, Y.A. et al. Orientation-dependent mutual crystalline and amorphous order in a single phase solid. Nat Commun 17, 2646 (2026). https://doi.org/10.1038/s41467-026-69359-3
Keywords: amorphous materials, crystalline order, niobium tungsten oxide, thin film nanorods, atomic structure