Clear Sky Science · en
Competing hydrogen-bond orders drive water’s anomalous surface tension
Why the Surface of Water Is Stranger Than It Looks
Water’s surface lets insects walk on ponds, shapes raindrops, and controls how bubbles form and burst. Yet when scientists measure how “tight” that surface is—the surface tension—they find that water behaves in a surprisingly odd way as it gets colder, especially when it is chilled below its normal freezing point. This article uses advanced computer simulations to reveal how the hidden organization of water molecules at the surface explains this long-standing mystery.
Two Hidden Types of Liquid Water
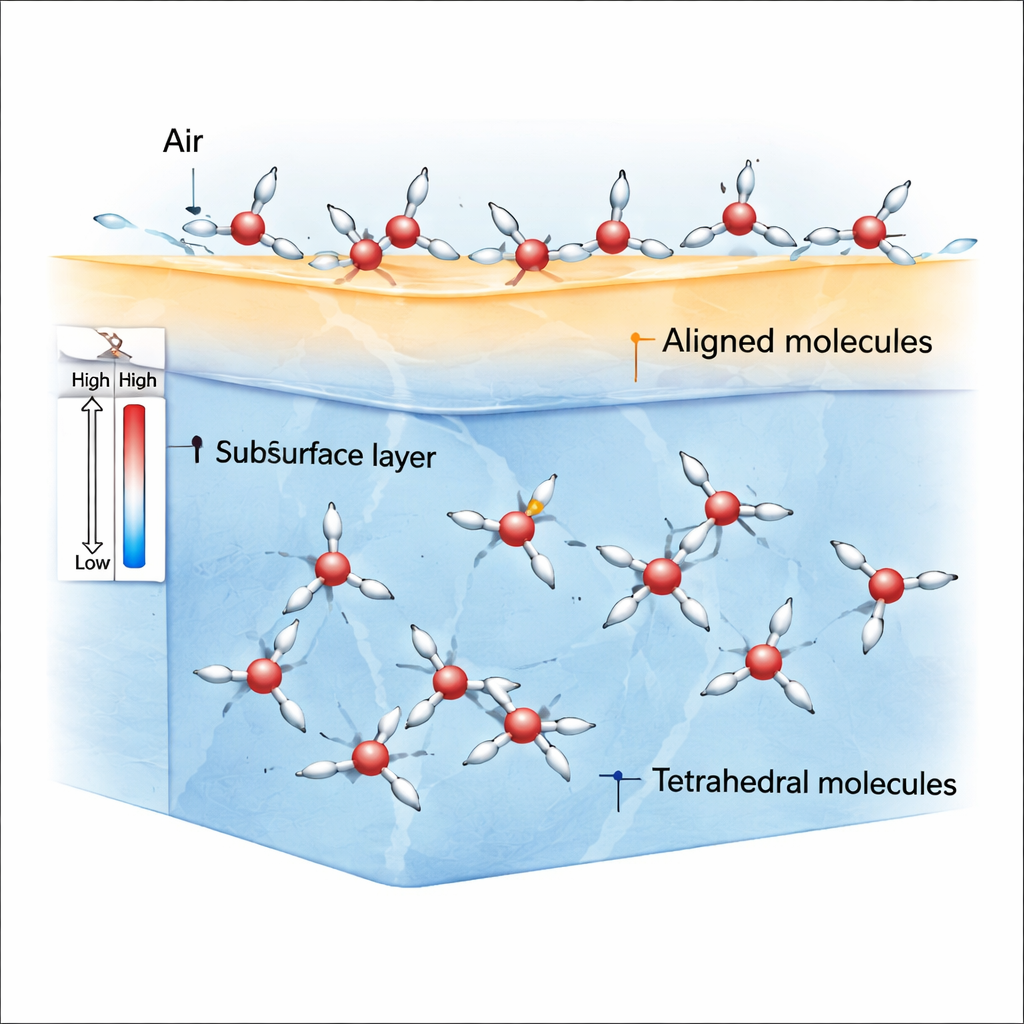
Although we experience water as a simple, uniform liquid, its molecules can locally arrange themselves in two contrasting ways. One arrangement is more crowded and disordered; the authors call this the ρ-state. The other is more open and symmetrical, with each molecule surrounded in a roughly four-way, tetrahedral pattern; this is called the S-state. Previous work suggested that the balance between these two local patterns helps explain many of water’s odd bulk properties. Here, the authors ask how this same two-state picture plays out at the air–water interface, where surface tension is born.
How the Surface Pulls Molecules into Line
At the boundary between air and water, molecules no longer feel equal pulls in all directions. This broken symmetry favors ρ-state molecules whose hydrogen atoms and molecular dipoles can easily lean in a preferred direction. Simulations show that near room temperature and somewhat below, the very top layer of water becomes packed with these aligned ρ-state molecules, while S-state molecules are more common slightly beneath the surface and in the bulk. Because ρ-state molecules line up strongly, they create an imbalance of forces—stress anisotropy—that generates a relatively large surface tension compared with ordinary liquids such as benzene.
Why Cooling First Slows, Then Speeds Up Surface Tightening
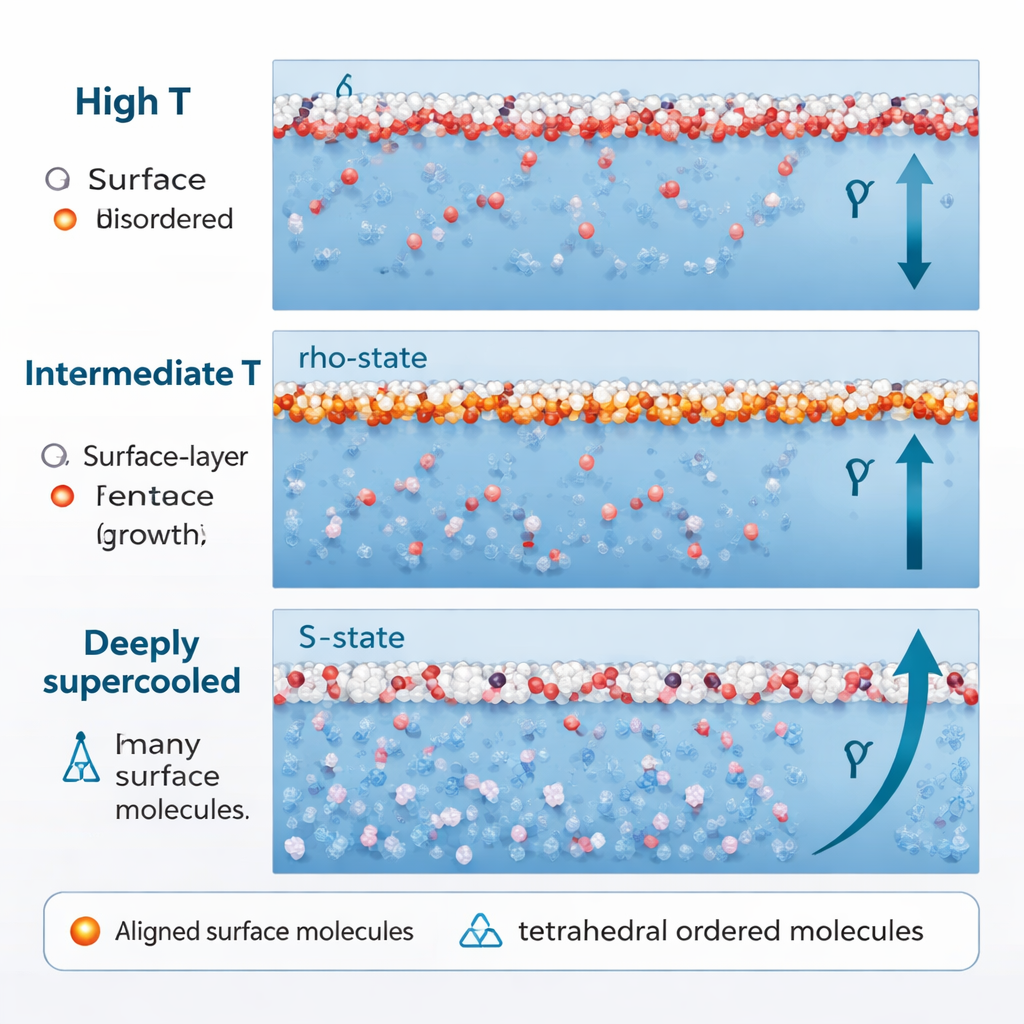
In most simple liquids, cooling steadily tightens the surface, making surface tension rise almost linearly. Water does this only at higher temperatures. As it cools toward about –0.15 °C (275 K), the rise in surface tension begins to slow. The simulations show that this slowdown happens because the ρ-state molecules at the surface have already become as aligned as they can; further cooling barely changes their contribution. At the same time, S-state molecules in the subsurface remain mostly randomly oriented, so they do little to increase the surface stress. The net result is a kind of plateau: temperature keeps dropping, but the surface tension grows only sluggishly.
Deep Supercooling Triggers a Second Tightening
When water is cooled much further, well below its normal freezing point into the deeply supercooled regime (around 250 K and below), its behavior changes again. The fraction of S-state, tetrahedral structures rises sharply, even near the surface. Crucially, these S-state molecules no longer point in random directions. Their dipoles start to align along the direction perpendicular to the surface, driven by interactions between neighboring molecular dipoles and by a region of negative pressure just under the surface. Once that happens, S-state water, which earlier softened the surface tension, now begins to reinforce it. This added, ordered contribution produces a renewed, faster rise—the “reentrant” increase—in surface tension at low temperatures.
From Surface Puzzles to Ice and Beyond
The same tetrahedral S-state clusters that stiffen the surface at low temperatures also resemble early building blocks of certain forms of ice, and the simulations find them enriched near the interface. That means the air–water boundary can act as a cradle for ice formation, helping explain why freezing often starts at surfaces. More broadly, the work provides a concrete, molecule-level link between how water molecules arrange and orient themselves and how strongly the surface pulls inward. This structural–mechanical picture not only resolves the strange temperature curve of water’s surface tension, but also offers a roadmap for understanding and controlling interfacial phenomena in other network-forming liquids, from supercooled water in clouds to materials used in technology and biology.
Citation: Yuan, J., Qiu, K., Sun, G. et al. Competing hydrogen-bond orders drive water’s anomalous surface tension. Nat Commun 17, 1498 (2026). https://doi.org/10.1038/s41467-026-69356-6
Keywords: water surface tension, hydrogen bonding, supercooled water, liquid structure, ice nucleation