Clear Sky Science · en
Fluorescence-free single-molecule microscopy via electronic resonance stimulated Raman scattering
A New Way to See Single Molecules
Being able to watch individual molecules at work has transformed biology and medicine, from tracking how proteins move inside cells to reading DNA sequences. Today this is mostly done with glowing fluorescent tags, but these tags blur together when many different types are present. This study introduces a new kind of microscope contrast that does not rely on glow at all. Instead, it listens to the tiny vibrations of specially designed molecules, opening a path toward clearer, more detailed and more flexible imaging of life at the single-molecule level. 
Why Glowing Dyes Reach Their Limits
Fluorescent dyes have been the workhorse of modern microscopy. They are bright, can be attached to specific molecules and are sensitive enough to reveal individual proteins or strands of DNA. However, each fluorescent dye emits light across a relatively broad range of colors. When many different targets need to be imaged at once, these broad colors overlap, making it hard to tell one molecule from another. To get around this, researchers often run many cycles of staining and washing, which is slow and can disturb delicate samples.
Listening to Molecular Vibrations Instead of Light
Every molecule also has a unique pattern of vibrations, like a fingerprint in how its atoms jiggle and stretch. These vibrations can be probed with techniques such as Raman and infrared spectroscopy, which feel out tiny shifts in light color when it interacts with a vibrating bond. These vibrational fingerprints are extremely narrow compared with fluorescence colors, so in principle dozens of different molecules can be distinguished at once. The catch is that vibrational signals are naturally weak, so past methods either needed metal nanostructures to boost the signal or still relied on fluorescence to read out the effect, bringing back the same background problems.
A Fluorescence-Free Boost for Raman Signals
The authors build on a method called electronic resonance stimulated Raman scattering (ER-SRS), which dramatically amplifies vibrational signals by matching the color of one laser beam to an electronic transition in the molecule and the color difference between two beams to a specific vibration. Earlier versions of ER-SRS struggled because the same conditions that amplified the Raman signal also produced a large unwanted electronic and fluorescent background. To solve this, the team tackled both sides of the problem: they engineered a laser system with two independently tunable beams and created a new family of molecular probes that absorb strongly in the near-infrared but barely fluoresce at all. These "Raman-amplified nonfluorescent molecular probes," or RANMPs, are built around a conjugated core with four vibration-rich nitrile groups that provide strong, sharp Raman fingerprints.
Designing Quiet but Responsive Molecular Probes
The key chemical trick is that RANMP molecules quickly divert energy into a non-glowing triplet state instead of re-emitting it as fluorescence. Heavy atoms such as sulfur inside the structure increase the rate of this diversion, efficiently quenching glow while still allowing the vibrational mode to be driven by the laser beams. Quantum chemical calculations guided the design so that the absorption color and the nitrile vibration lined up with the laser tuning range. By carefully adjusting the molecular structure, the researchers could shift the exact vibrational frequency and strength, creating several related probes with distinct but closely spaced Raman fingerprints. Under optimized ER-SRS conditions, these molecules produced vibrational signals hundreds of times stronger than those from a standard fluorescent dye used in earlier work, yet with far less background.
Seeing Single Particles and Single Molecules
With these ingredients in place, the team demonstrated what the new approach can do. First, they packed RANMP dyes into tiny polymer nanoparticles known as polymer dots, which further concentrate the probes and suppress any residual fluorescence. Using ER-SRS, they imaged individual dots in solution and distinguished two probe types whose nitrile vibrations differed by only a small amount, effectively achieving dual-color single-particle imaging in a single scan. Next, they diluted the probes down to the level of a few molecules embedded in a thin plastic film. By protecting the sample from damage and adjusting laser powers and timing, they recorded sharp, diffraction-limited spots that blinked off in single steps, a hallmark of single-molecule detection. They also showed that these spots vanished when the timing or frequency difference between the two laser beams was shifted off the nitrile vibration and reappeared when it was restored, confirming that the signal truly arises from a specific bond vibration. 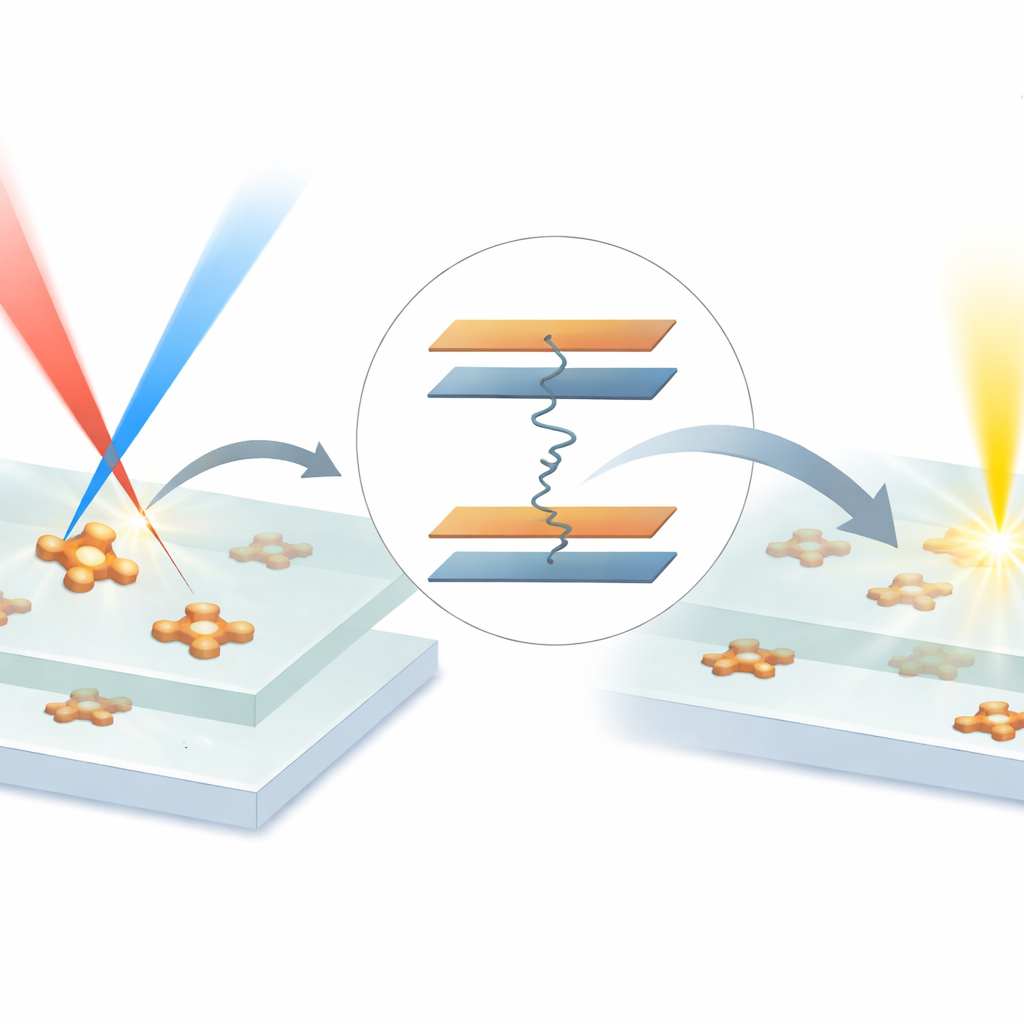
What This Means for Future Imaging
In plain terms, the study proves that it is possible to see and distinguish individual molecules using their vibrational fingerprints alone, without relying on glow. Because vibrational lines are narrow and can be tuned by chemical design, this offers a powerful route to tagging many different targets at once with minimal overlap. The non-glowing nature of the probes also reduces background and should make it easier to look deeper into tissues, where stray fluorescence usually becomes overwhelming. While further work is needed to adapt these probes for live cells and to expand the color palette, ER-SRS with RANMPs points toward a future in which single-molecule maps of complex biological samples can be drawn with unprecedented clarity and multiplexing.
Citation: Oh, S., Eom, Y., Kim, H.Y. et al. Fluorescence-free single-molecule microscopy via electronic resonance stimulated Raman scattering. Nat Commun 17, 2720 (2026). https://doi.org/10.1038/s41467-026-69348-6
Keywords: single-molecule microscopy, stimulated Raman scattering, vibrational imaging, nonfluorescent probes, multiplexed bioimaging