Clear Sky Science · en
“On-water” photosensitization enables redox neutral acylation and alkylation of quinones
Water Helping Green Chemistry
Chemists have long admired how nature uses water as a safe, efficient reaction medium inside living cells. This study takes inspiration from that idea and shows how ordinary water, plus visible light from simple LEDs, can drive useful carbon–carbon bond‑forming reactions. The work reveals that the surface where oil and water meet is not just a boundary; it can act like a tiny, self‑assembling reactor that makes tough transformations cleaner, milder, and more versatile.
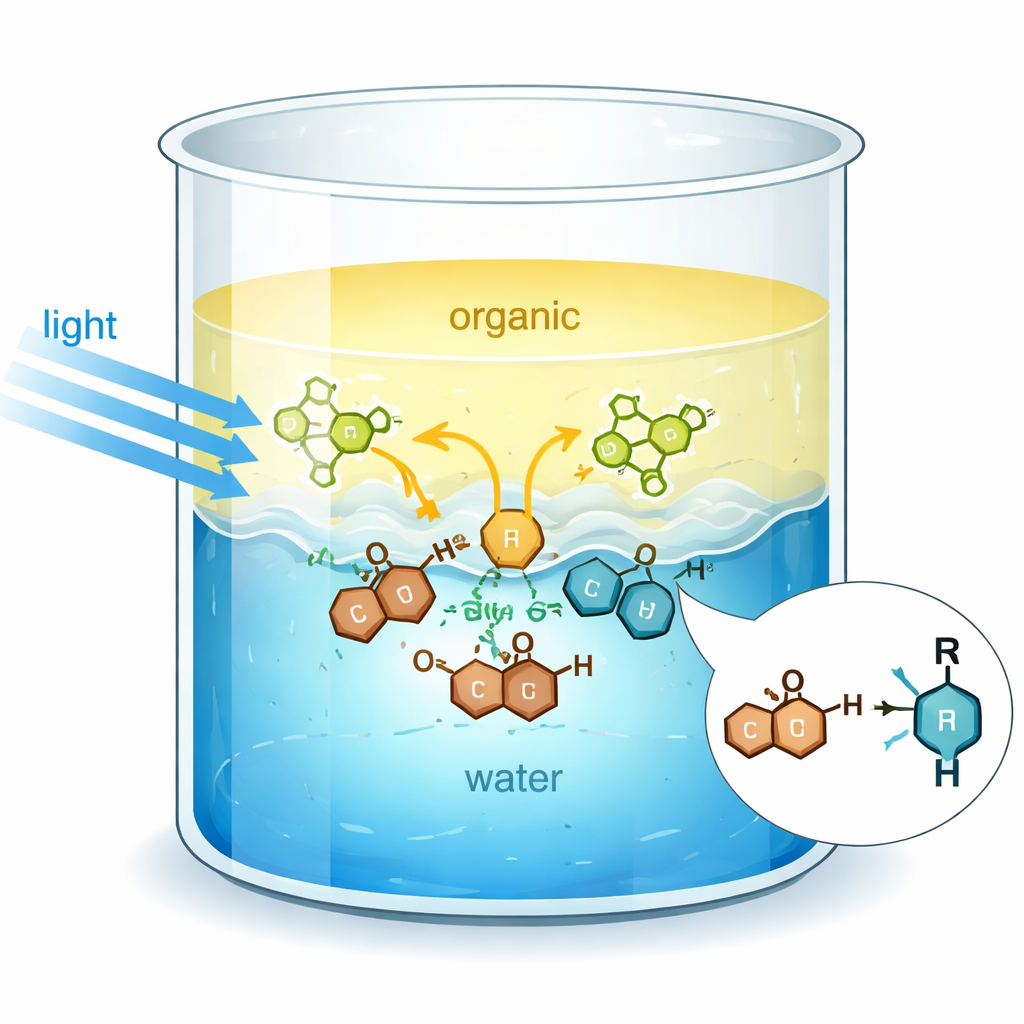
A Busy Border Between Oil and Water
When oil and water are shaken together, they separate into two layers, but the shared surface between them becomes a highly organized environment. Water molecules there form a dense network of hydrogen bonds, which creates a strong, structured interface. The authors show that this interface can assist organic reactions in several ways: it collects otherwise poorly mixed molecules, increases their local concentration, and subtly alters their electronic properties. In particular, key reactants called quinones—ring‑shaped molecules related to many natural products and drugs—interact with water at the surface so that their electronically excited states are stabilized and shifted to lower energy.
Turning Light into Chemical Work
To harvest visible light efficiently, the team uses a common dye, Eosin Y, as a photosensitizer. Under normal conditions, quinones mainly absorb ultraviolet light, which is harsher and can degrade products. At the water–organic interface, however, the energy levels of both the quinone and Eosin Y shift in opposite but complementary ways. Detailed optical measurements show that water moves the quinone’s light‑absorption band toward longer wavelengths while nudging Eosin Y’s band toward shorter ones. This makes energy transfer from excited Eosin Y to the quinone highly favorable under blue or green LED light, activating the quinone without resorting to intense UV sources or elaborate equipment.
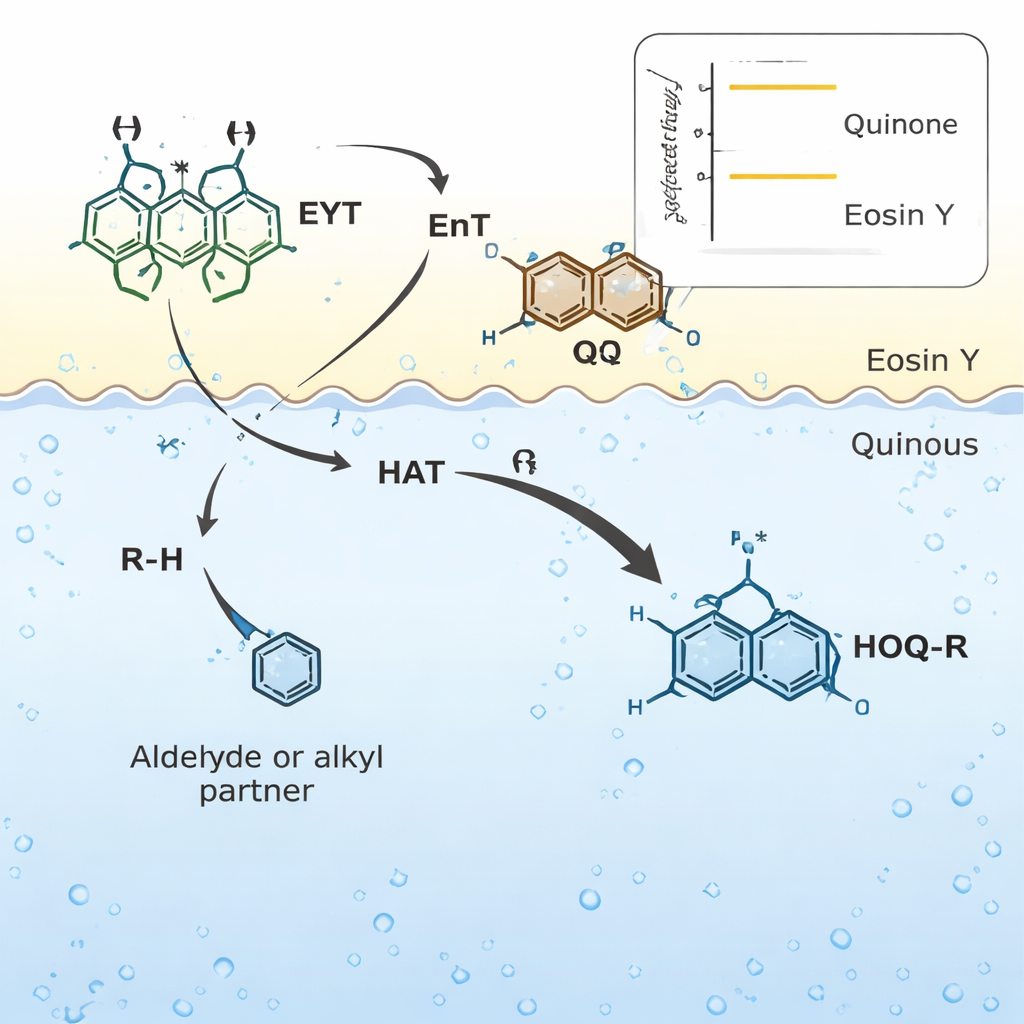
Building Complex Molecules More Gently
Once activated by light at the interface, the quinone can pluck a hydrogen atom from a wide range of partner molecules—such as aromatic and aliphatic aldehydes, ethers, thioethers, alkanes, silanes, and amines—forming short‑lived radicals on both sides. These radicals quickly recombine to forge new carbon–carbon bonds, producing so‑called 2‑functionalized quinols in a single step. Because no net oxidation or reduction occurs, the overall process is redox‑neutral, avoiding extra reagents and waste. The authors demonstrate over a hundred examples, including many fragrance components and building blocks derived from marketed drugs, and show that the method scales from milligram to gram quantities while maintaining good yields.
From Reactive Intermediates to Useful Products
The new quinol products are not just curiosities; they are versatile hubs for further chemistry. Some are readily oxidized back to quinones, reduced to more saturated structures, or folded into more elaborate ring systems reminiscent of natural antibiotics and anticancer agents. The team also applies the method to a variety of quinone‑like cores, including simple benzoquinones, larger polyaromatic systems, and maleimides, highlighting that the underlying concept—light‑driven hydrogen atom transfer from a partner molecule to an excited, water‑stabilized quinone—is broadly applicable. Careful control experiments with radical traps, isotopic labeling, and alternative light‑absorbing catalysts support this mechanistic picture.
Why This Matters for Everyday Chemistry
For a non‑specialist, the impact of this work lies in how it combines three attractive features: water as a benign medium, visible light as a gentle energy source, and widely available organic molecules as partners. By exploiting the natural organization at the oil–water boundary, the method sidesteps harsh reagents, minimizes waste, and opens new routes to bioactive compounds and fine chemicals. In essence, the authors show that the humble surface of water can be engineered into a powerful, green platform for building complex molecules that matter in medicine, materials, and everyday products.
Citation: Mandal, T., Sharma, R., Mendez-Vega, E. et al. “On-water” photosensitization enables redox neutral acylation and alkylation of quinones. Nat Commun 17, 1813 (2026). https://doi.org/10.1038/s41467-026-69343-x
Keywords: photochemistry, green chemistry, quinones, hydrogen atom transfer, on-water reactions