Clear Sky Science · en
Microbiota-derived IPA protects against colitis by regulating intestinal HMGCS2-mediated ketogenesis to facilitate mucosal healing
Why Gut Bacteria Matter for a Hurting Intestine
When the lining of the gut is damaged, as in inflammatory bowel disease (IBD) or after harsh medical treatments, the body must rapidly repair this inner “skin” to keep harmful contents from leaking into the bloodstream. This study uncovers how a single molecule made by friendly gut bacteria can help the intestine heal itself, pointing to new ways to treat chronic bowel disorders without broadly shutting down the immune system.
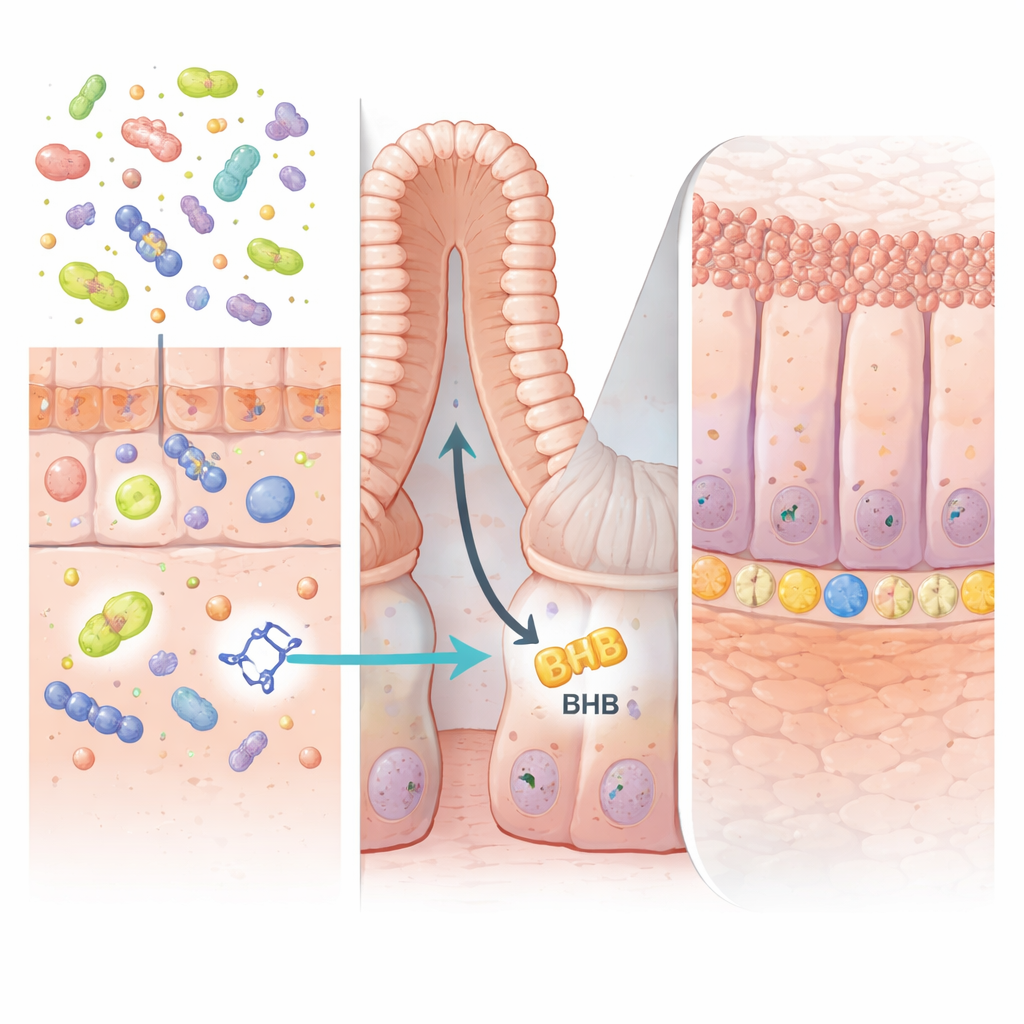
A Helpful Signal from Friendly Microbes
The researchers began by comparing chemical fingerprints from stool samples of people with IBD and healthy volunteers, as well as in several mouse models of colitis. Among hundreds of molecules, one stood out: indole-3-propionic acid (IPA), a breakdown product of the dietary amino acid tryptophan made by certain gut bacteria. IPA levels were consistently lower in people with Crohn’s disease and in mice with colitis than in healthy counterparts. This suggested that losing IPA might be part of what goes wrong when the gut lining becomes inflamed and fails to repair.
Strengthening the Gut’s Protective Wall
To test whether IPA merely tracked disease or actually protected against it, the team gave IPA to mice before inducing several types of intestinal injury, including chemical colitis and radiation damage. Mice receiving adequate doses of IPA had longer, healthier colons, fewer inflammatory changes under the microscope, and a thicker mucus layer with tighter cell junctions. They leaked fewer inflammatory markers and had more goblet cells, which produce the protective mucus coating. Importantly, these benefits were seen even in germ-free mice that lack a resident microbiome, showing that once IPA is present, it can act directly on the gut lining without needing other microbes.
Fueling Stem Cells with a Special Kind of Energy
The key to IPA’s action lay deep in the intestinal “crypts,” the tiny pockets where stem cells sit and constantly renew the lining. The authors showed that IPA switches on a protein called PPARα in intestinal epithelial cells. This, in turn, boosts the production of an enzyme named HMGCS2, which drives ketogenesis—the process of making the energy-rich molecule β‑hydroxybutyrate (BHB). Elevated BHB within the gut wall stimulated LGR5-positive stem cells to expand and regenerate damaged tissue faster. When the HMGCS2 enzyme or PPARα was selectively deleted from intestinal cells in mice, IPA could no longer enhance stem cell activity or protect against colitis, confirming that this ketone-producing pathway is essential.
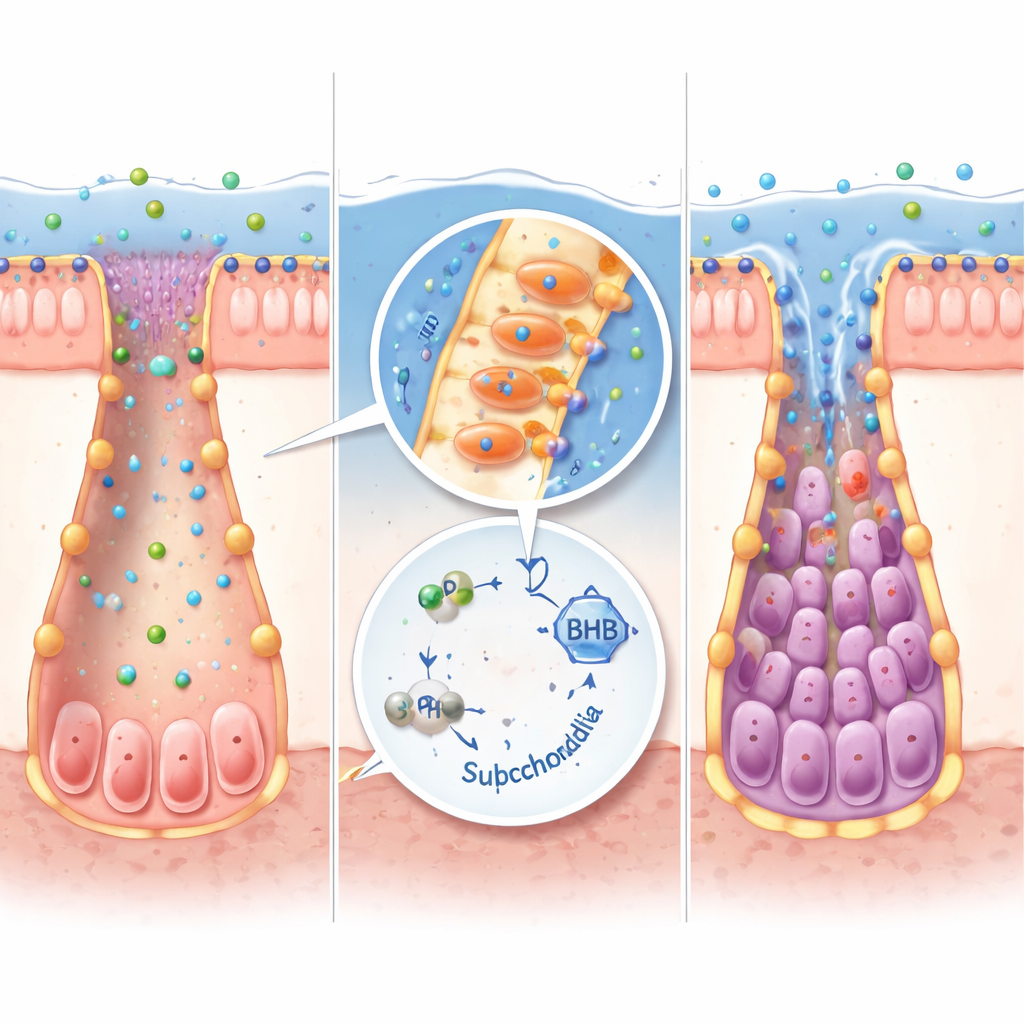
From Bacterial Factories to Human Tissues
Not all gut bacteria can make IPA. By reanalyzing large microbiome datasets, the researchers identified Peptostreptococcus russellii as a key IPA producer that is depleted in several IBD cohorts. In lab cultures, this bacterium converted tryptophan into IPA efficiently. When germ-free mice were colonized with P. russellii, their IPA levels rose, ketone-producing genes turned on, stem cell markers increased, and colitis severity dropped compared with mice receiving a generic microbial transplant. The same IPA–ketone pathway worked in miniature gut organoids grown from mouse and human tissues, where adding IPA or BHB helped stem cells rebound after inflammatory damage while dialing down pro-inflammatory signals.
What This Could Mean for Future Treatment
Taken together, the work outlines a simple but powerful chain of events: certain gut bacteria turn dietary tryptophan into IPA; IPA activates a metabolic switch in intestinal cells; this switch boosts production of BHB; and BHB, in turn, revives stem cells that rebuild the gut lining. For people with IBD or other conditions that injure the intestinal barrier, therapies that restore IPA or safely mimic its effects—through diet, targeted probiotics, or designer drugs—could promote true mucosal healing without broad immune suppression. While more studies are needed in patients, this microbe–metabolite–stem cell axis offers a promising new angle for repairing the gut from the inside out.
Citation: Zhang, Y., Tu, S., Shao, X. et al. Microbiota-derived IPA protects against colitis by regulating intestinal HMGCS2-mediated ketogenesis to facilitate mucosal healing. Nat Commun 17, 2437 (2026). https://doi.org/10.1038/s41467-026-69341-z
Keywords: gut microbiome, intestinal stem cells, inflammatory bowel disease, microbial metabolites, mucosal healing