Clear Sky Science · en
A medium-entropy oxygen electrode enables high-performance and contaminant-tolerant reversible solid oxide cells
Cleaner Power from a Tougher Electrode
Modern societies need ways to store and deliver clean energy around the clock, not just when the wind blows or the sun shines. Reversible solid oxide cells (Re-SOCs) are promising devices that can both generate electricity and store energy, but one of their most important parts—the oxygen electrode—tends to weaken in real-world, contaminated air. This study introduces a new "medium-entropy" oxygen electrode material that keeps working efficiently even in harsh, chromium-laden conditions, moving Re-SOCs a step closer to practical, large-scale use.
Why Flexible Ceramic Batteries Matter
Re-SOCs are high-temperature ceramic devices that can switch roles. In fuel cell mode, they turn fuels such as hydrogen into electricity; in electrolysis mode, they use electricity to split water or other molecules, storing energy in chemical form. This dual capability makes them attractive for stabilizing power grids fed by renewable sources, shaving peak demand and filling valleys when generation is low. However, the oxygen electrode, which has to breathe air and handle fast oxygen reactions, often becomes the weak link—especially at the intermediate temperatures favored for efficiency and durability.
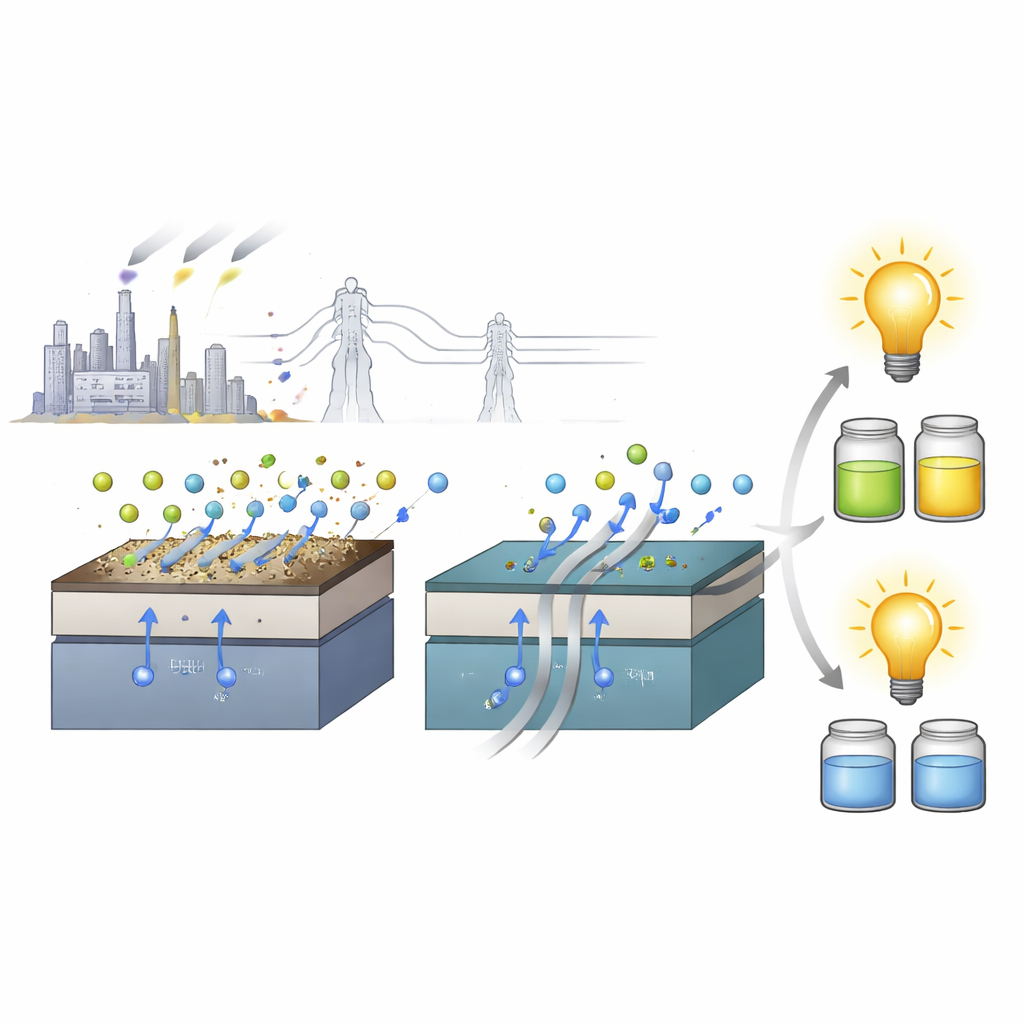
How Air Impurities Quietly Poison Cells
Inside a Re-SOC stack, metallic connectors slowly release gaseous chromium compounds when heated. These chromium species drift through the air channels and react with ingredients that naturally migrate to the surface of many oxygen electrodes, forming electrically sluggish crusts. Over time, these crusts blanket the electrode surface and even let chromium seep into its crystal framework. The result is simple but damaging: electrical pathways are blocked, oxygen has a harder time getting in and out, and the device’s ability to generate or store energy fades much faster than designers would like.
A New Mix of Metals for a More Robust Electrode
The researchers tackled this problem by designing a complex oxide with several different metal elements mixed at the atomic level: praseodymium, barium, strontium, calcium, and cobalt, arranged in a perovskite structure. This "medium-entropy" composition is engineered so that the disorder among the different metal ions actually stabilizes the structure at high temperatures and discourages harmful segregation of surface species that attract chromium. Detailed microscopic and spectroscopic tests show that the material has abundant surface sites where oxygen can quickly come and go, high electrical conductivity, and fast oxygen transport through its bulk—key ingredients for strong performance in both electricity generation and electrolysis.
Putting the New Electrode to the Test
When the team built full Re-SOC devices using their new oxygen electrode, they found that it delivered very high power outputs in fuel cell mode, even matching or exceeding many of the best reported materials tested under cleaner conditions. Crucially, the performance remained impressive when the air was deliberately contaminated with chromium vapor and water, mimicking realistic operating environments. In electrolysis mode, the same cells pushed large currents while splitting steam, again under chromium exposure, and they continued to run stably over many hours. The researchers even cycled the devices repeatedly between fuel cell and electrolysis operation for 100 hours and 25 full cycles, with the new electrode maintaining its function despite continuous contamination.
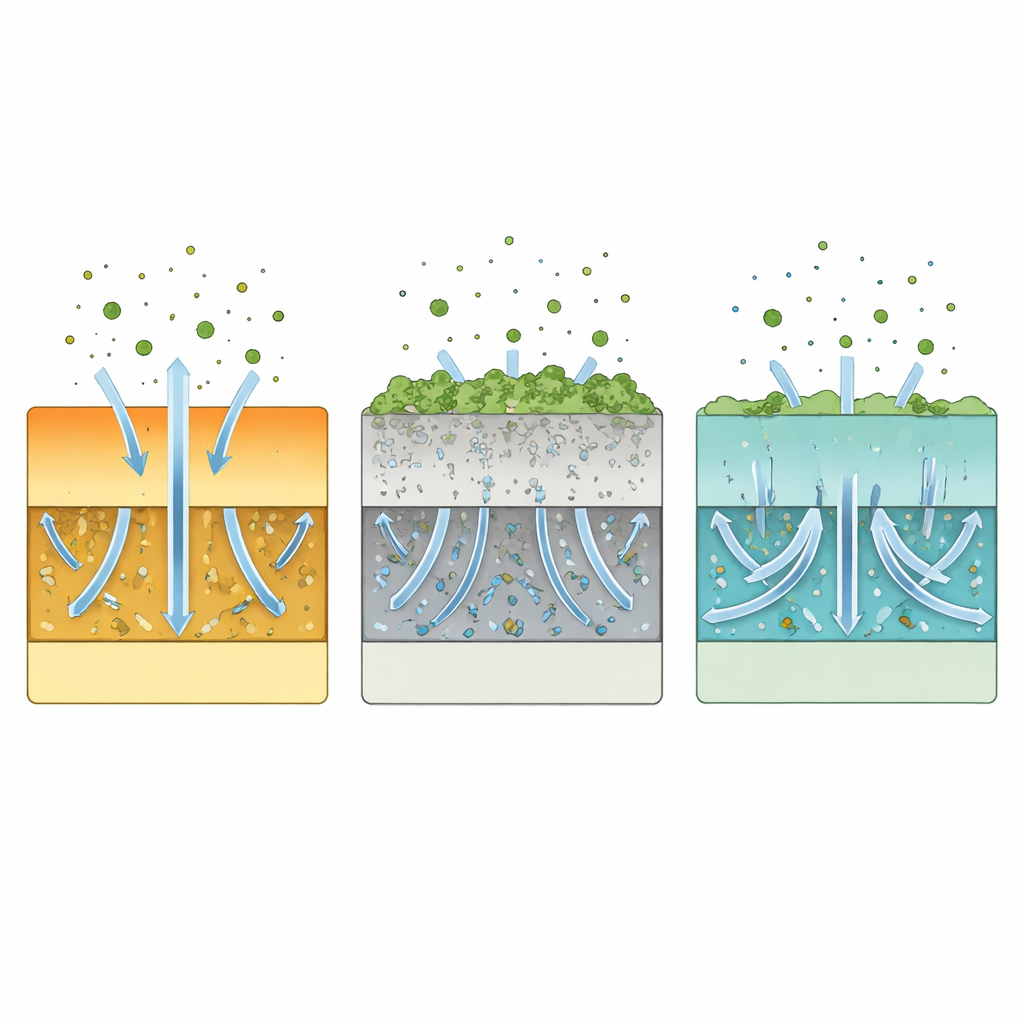
Why This Electrode Resists Poisoning
To understand why the new material is so tolerant, the authors compared it with a closely related but less complex electrode. They found that on the conventional material, chromium-rich compounds accumulated more heavily on the surface and penetrated deeper into the porous network, clogging pathways needed for oxygen movement. By contrast, the medium-entropy electrode showed far fewer chromium deposits and much shallower penetration, preserving open channels for gas flow and charge transport. Measurements of oxygen exchange rates and electrical conductivity over time confirmed that the new material degraded more slowly, tying its chromium resistance directly to its physical and chemical resilience.
What This Means for Future Energy Systems
In everyday terms, the study shows that by carefully mixing several elements into a single, slightly disordered crystal, it is possible to build an oxygen electrode that keeps working in dirty, high-temperature air where ordinary materials fail. This tougher, high-performance electrode helps Re-SOCs deliver both strong power and reliable long-term operation in the presence of chromium impurities that are difficult to avoid in practical devices. As energy systems increasingly rely on flexible, high-efficiency technologies to balance renewable power, such contaminant-tolerant materials could play a central role in making ceramic energy converters reliable enough for widespread commercial use.
Citation: Zhu, F., Xu, K., Liao, Y. et al. A medium-entropy oxygen electrode enables high-performance and contaminant-tolerant reversible solid oxide cells. Nat Commun 17, 2617 (2026). https://doi.org/10.1038/s41467-026-69338-8
Keywords: reversible solid oxide cells, oxygen electrode, chromium poisoning, high entropy oxides, clean energy storage