Clear Sky Science · en
Loss-of-function variants in the CAPN1 activator CD99L2 cause X-linked spastic ataxia
Why this matters for families with unexplained movement problems
Many people live for years with unexplained walking difficulties, muscle stiffness, or problems with balance and speech, never learning the real cause. This study shows how modern DNA testing can finally give some of these families answers. The researchers not only compared different genetic tests for rare movement disorders, they also uncovered a previously unknown cause of a condition called X-linked spastic ataxia, pointing toward biological pathways that may also be important in more common brain diseases.
Finding the genetic needles in a haystack of rare diseases
Rare movement disorders, such as ataxia (unsteady movements) and spastic paraplegia (stiff, weak legs), are often suspected to be genetic, but for most patients standard tests come back negative. The team followed 2,811 people in Germany and across Europe who were referred for suspected rare movement disorders over six years. First, they looked at traditional targeted tests that search for known repeat expansions in a handful of genes; these delivered answers in about 11% of cases. Next, they used exome sequencing, which reads just the protein-coding parts of the genome, and found definite genetic explanations in about 19% of patients, especially in those with spasticity.
Looking beyond standard tests with whole-genome sequencing
To push further, the scientists used whole-genome sequencing, which reads nearly all of a person’s DNA, including regions that standard tests and exomes may miss. Among 486 individuals who had this more comprehensive test, the diagnostic rate increased by roughly 7.5 percentage points, largely because genome sequencing is better at spotting complex changes such as structural rearrangements and repeat expansions. The study also showed that carefully recorded clinical information—especially specific symptom descriptions, younger age at testing, and having a combination of spasticity with other movement problems—helped predict who was most likely to receive a clear genetic diagnosis.
Uncovering a new X-linked cause of spastic ataxia
Even after these extensive tests, many patients remained without a diagnosis. The researchers pooled genetic data from more than 13,000 individuals and used a “gene-burden” approach, asking which genes carried suspicious variants more often in patients than in unaffected controls. This analysis pointed not only to known disease genes, but also strongly highlighted a previously overlooked gene on the X chromosome called CD99L2. By combining results from several families around Europe, they identified 25 affected males from 20 families who carried damaging variants in this gene. These men typically developed walking problems, leg stiffness, slurred speech, and sometimes balance difficulties in mid to later adulthood, while female carriers were mostly unaffected—patterns that fit an X-linked disorder. The variants mainly destroyed the normal protein or removed crucial parts of it, strongly suggesting that losing its function causes disease.
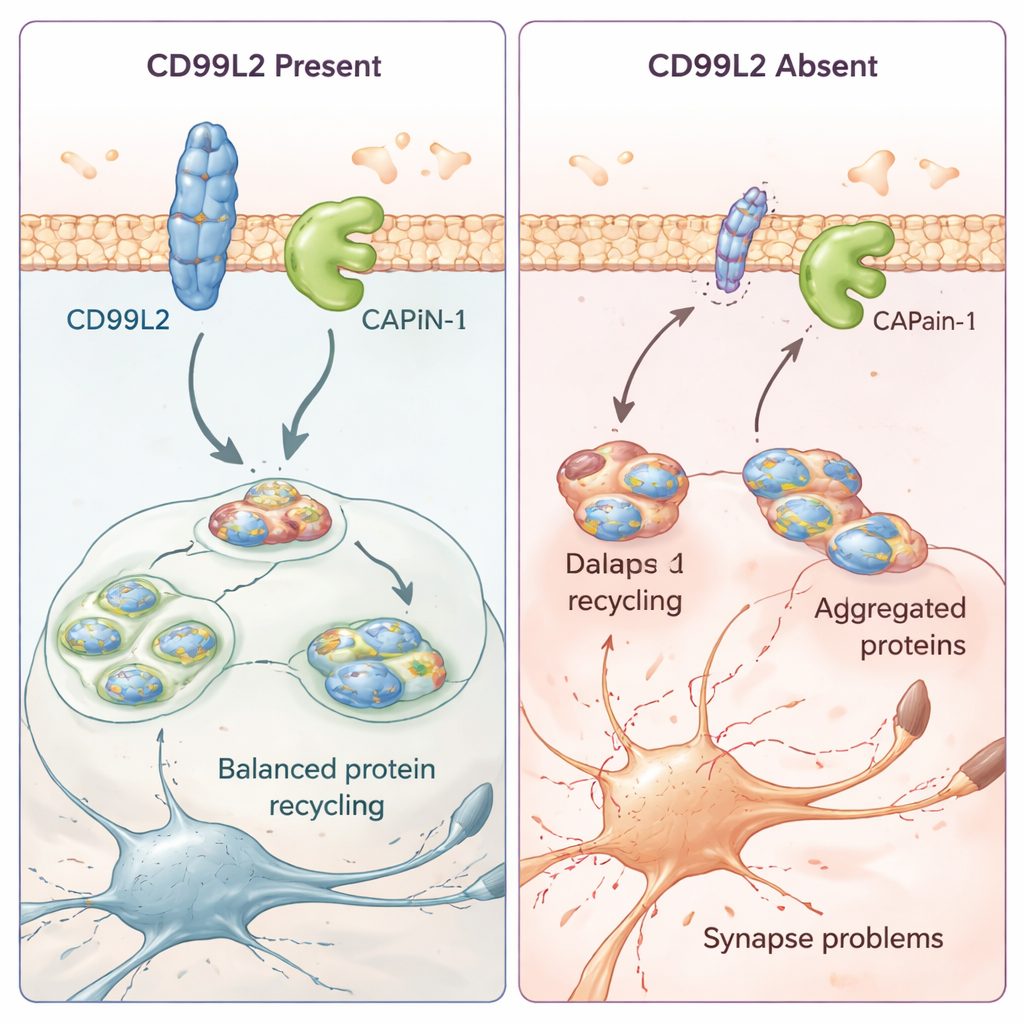
How a small membrane protein helps protect brain cells
To understand what CD99L2 actually does in cells, the team turned to cell models and patient-derived skin cells. They found that the CD99L2 protein sits in the cell membrane and is usually tagged with small “ubiquitin” markers that control how long it survives before being broken down. CD99L2 physically binds to calpain-1 (CAPN1), a calcium-activated enzyme that trims other proteins and helps keep synapses—the contact points between nerve cells—healthy. When CD99L2 is present and intact, it helps turn calpain-1 on and off in a controlled way, and then is itself clipped and recycled. When CD99L2 is missing or structurally altered, calpain-1 activation is impaired. In patient cells, this goes hand in hand with disturbed activity of many genes related to synapses and nerve-cell communication, suggesting that subtle but widespread changes in brain circuitry may underlie the gradual movement problems.
What this means for patients today and tomorrow
For families with unexplained spastic ataxia or spastic paraplegia, this work offers two kinds of progress. First, it shows that using whole-genome sequencing early, together with careful clinical description, can noticeably increase the chances of a firm genetic diagnosis. Second, it adds CD99L2 to the list of genes that control calpain activity, a pathway already implicated in other rare ataxias and in common conditions like Alzheimer’s and Parkinson’s disease. In everyday terms, the study reveals a new “on–off” switch that helps keep brain-cell maintenance in balance; when that switch is broken, nerve cells slowly deteriorate, leading to stiffness and poor coordination. Understanding this switch may eventually open the door to treatments that fine-tune calpain activity and protect brain cells across a range of neurological diseases.
Citation: Menden, B., Incebacak Eltemur, R.D., Demidov, G. et al. Loss-of-function variants in the CAPN1 activator CD99L2 cause X-linked spastic ataxia. Nat Commun 17, 1698 (2026). https://doi.org/10.1038/s41467-026-69337-9
Keywords: spastic ataxia, rare movement disorders, genome sequencing, CD99L2, calpain-1