Clear Sky Science · en
Proprioceptive limit detectors contribute to sensorimotor control of the Drosophila leg
How Flies Keep Their Steps Safe
Every time a leg swings forward, there’s a risk of overreaching and stumbling. From house cats to fruit flies, animals rely on hidden sense organs that tell the nervous system when a joint is near its limit. This study reveals how tiny sensors in the front legs of the fruit fly act as built‑in “stop guards,” helping the animal shift its weight, avoid collisions, and keep a stable posture—even while walking fast or grooming.
Hidden Sensors at the Edge of Motion
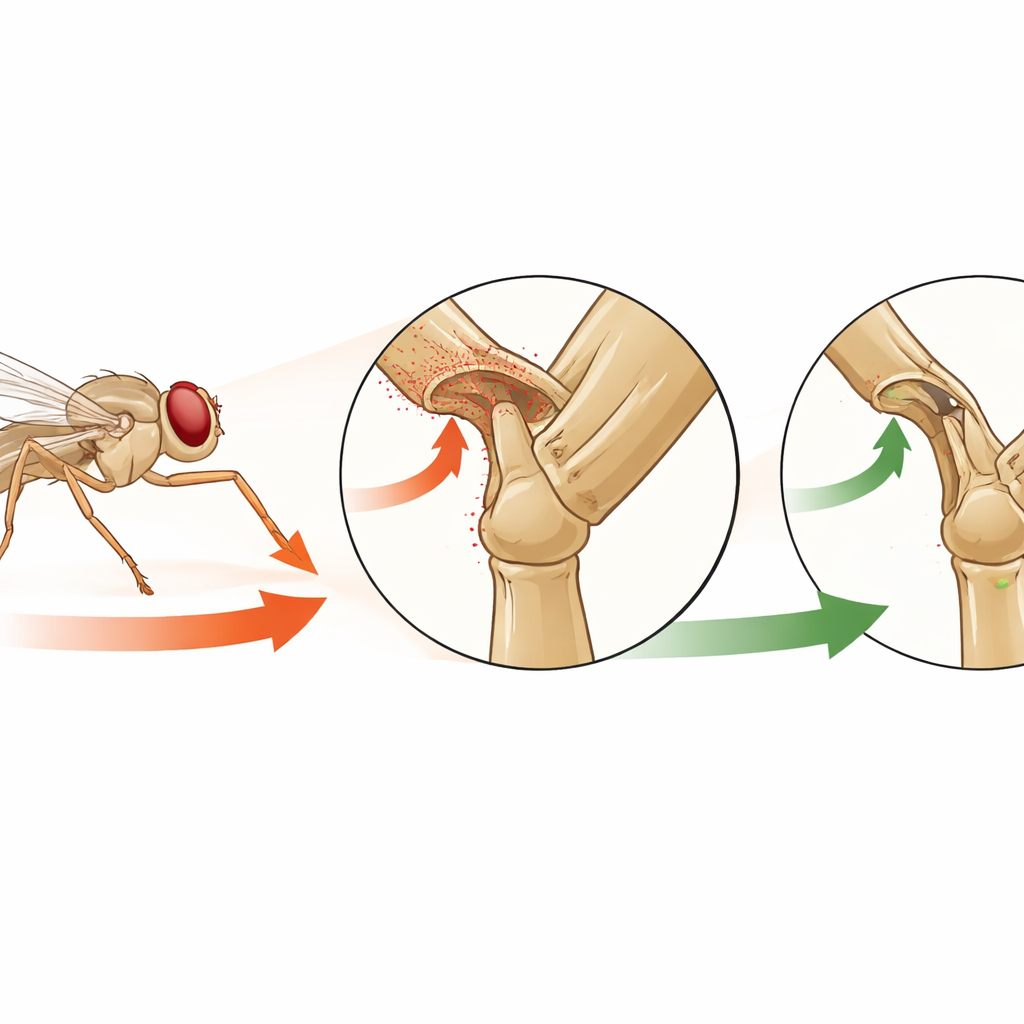
The authors focus on a special set of touch‑like sensors on the fly’s leg called hair plates. These are small clusters of stiff hairs sitting in folds of the leg joints, positioned so that they bend only when the joint is pushed toward the edge of its normal range. Using high‑resolution imaging and genetic tools, the team mapped all such hair plates on the fly leg and then zoomed in on one particular group on the front leg, named CxHP8. Their goal was to understand what angles of leg movement these sensors detect, how they are wired inside the nervous system, and what they actually do during natural behaviors.
Watching Nerve Activity While Moving the Leg
To see when CxHP8 turns on, the researchers used a two‑photon microscope to measure calcium signals inside its nerve fibers, a standard way to track activity. At the same time, they precisely controlled and tracked the position of a front leg in three dimensions. As they slowly moved the leg, they found that CxHP8 fired strongest when the upper leg segment rotated inward and translated toward the fly’s body—positions that correspond to the front leg reaching far forward and toward the midline. When the leg was held in these extreme positions, the activity stayed high rather than pulsing briefly, showing that these sensors continuously signal that the joint is at its limit. During natural behaviors like walking and grooming on a small ball, the same angles were reached, and CxHP8 stayed active whenever the leg swung to these extremes.
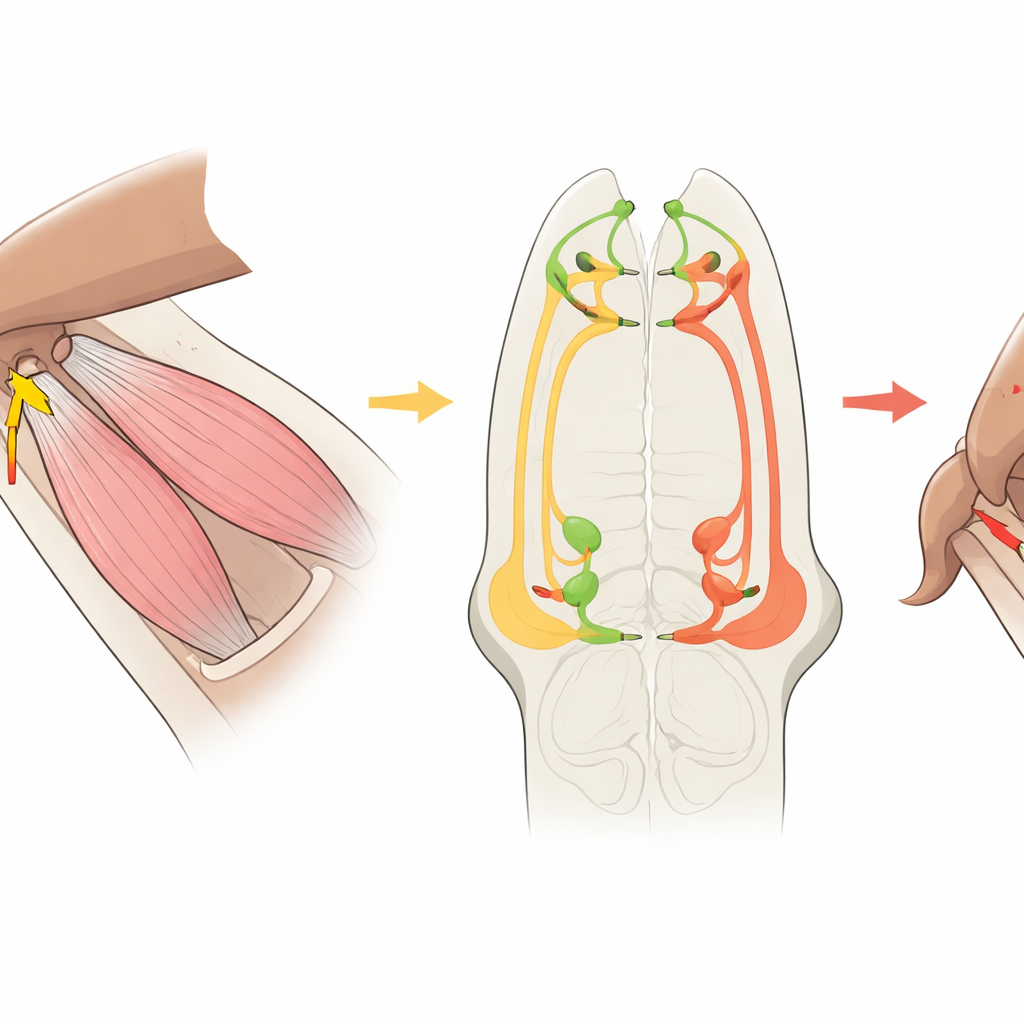
Tracing the Wiring from Sensor to Muscle
The team then turned to a detailed wiring map of the fly’s ventral nerve cord, reconstructed from electron microscopy of thousands of neurons. They traced the CxHP8 fibers from the leg into the nervous system and identified all their connections. Most of CxHP8’s outputs went directly or indirectly to leg motor neurons, especially those that pull the leg backward, as well as to a smaller group that push it forward. In this circuit, CxHP8 strongly excites motor neurons that move the leg rearward and, through inhibitory middleman cells, suppresses motor neurons that drive forward motion. This layout suggested a simple rule: when the leg reaches its forward limit and CxHP8 fires, the circuit should flip the leg from the forward‑swinging phase into a backward, weight‑bearing phase.
Forcing and Removing the Signal During Real Behavior
To test this prediction, the authors used light‑sensitive proteins to either activate or silence CxHP8 in behaving flies. When they flashed red light onto the joint of tethered flies engineered to activate CxHP8, the stimulated leg quickly moved backward, rotated outward, and flexed, with little effect on the other legs. Conversely, when they silenced CxHP8 either briefly with a light‑gated channel or chronically with a potassium channel, the front leg tended to overshoot its usual forward reach during walking. On both a floating ball and a motorized treadmill, steps ended with the foot placed slightly farther forward and closer to the midline, and the swing phase traveled a longer distance, even though the timing of steps and coordination between legs stayed largely intact. During rest, flies with silenced CxHP8 also spread their legs farther apart, forming a wider base of support.
Many Small Sensors, Each with a Job
Having confirmed the function of one hair plate, the researchers used the same wiring map to predict the roles of other hair plates at nearby joints. They reconstructed their nerve fibers and counted synapses onto different motor neuron groups. This analysis suggested that each hair plate is tuned to a particular direction of joint limit—some favoring forward motion, some backward, and others stabilizing between the two—by preferentially targeting the muscles that move the associated leg segment. In effect, the fly’s leg is ringed with small, specialized limit detectors, each pre‑wired to push the leg away from the specific extreme it monitors.
Why This Matters for Movement
This work shows how a tiny insect leg can serve as a model for understanding joint‑limit sensing in all animals. For a lay reader, the key idea is that the nervous system does not just track how fast or how far a limb is moving; it also has dedicated “trip wires” that fire when a joint gets too close to the edge. In the fruit fly, one such trip wire, CxHP8, continuously signals when the front leg swings too far forward and, through a simple but well‑defined circuit, triggers a shift into a backward, supporting step and helps set stable resting poses. Because the wiring diagram is now known in detail, this study also demonstrates that, at least in this small nervous system, it is possible to predict specific reflexes—such as when a leg will switch from swing to stance—directly from the layout of connections between sensors and muscles.
Citation: Pratt, B.G., Dallmann, C.J., Chou, G.M. et al. Proprioceptive limit detectors contribute to sensorimotor control of the Drosophila leg. Nat Commun 17, 2664 (2026). https://doi.org/10.1038/s41467-026-69333-z
Keywords: proprioception, Drosophila, locomotion, sensorimotor circuits, hair plate neurons