Clear Sky Science · en
Development of a potent monoclonal antibody for treatment of human metapneumovirus infections
Why a hidden winter virus matters
Every cold and flu season, a little-known virus called human metapneumovirus (HMPV) quietly sends millions of young children, older adults, and people with weakened immune systems to clinics and hospitals. Unlike its viral cousin respiratory syncytial virus (RSV), there are no vaccines or targeted treatments for HMPV. This study reports the discovery of a powerful laboratory-made antibody, called 4F11, that can stop HMPV in its tracks in cells and protect infected animals, pointing toward a much-needed new medicine for some of the most vulnerable patients.
The virus that flies under the radar
HMPV is a common respiratory virus worldwide, responsible for an estimated 14 million lower lung infections in children under five each year, and it also exacts a heavy toll in adults over 60. For patients whose immune systems are severely weakened—such as stem cell transplant recipients—the death rate from HMPV lung infections can be alarmingly high. Vaccines are only just emerging for related viruses like RSV, and even if an HMPV vaccine existed, many highly immunocompromised patients would respond poorly. Because of this gap, researchers are turning to monoclonal antibodies—precisely targeted immune proteins that can be given as drugs—to prevent or treat severe infections.
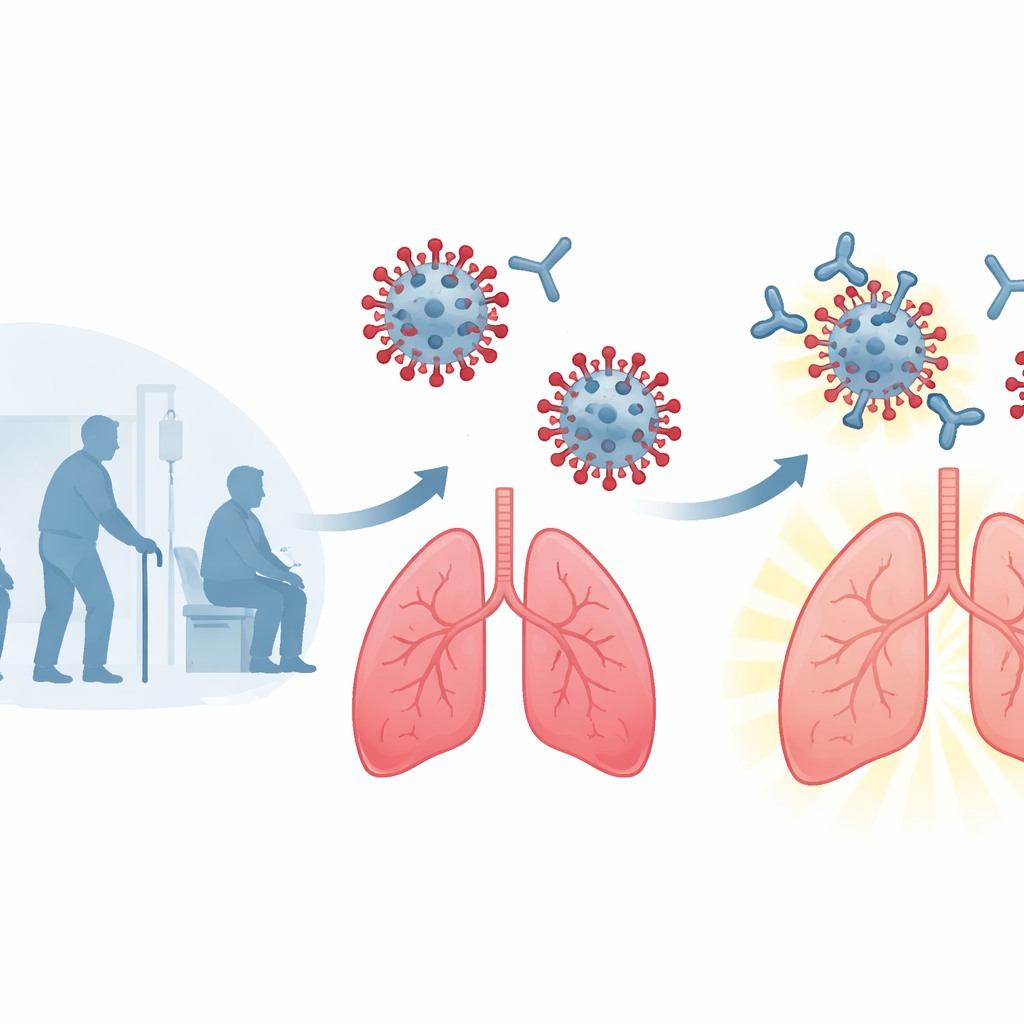
Finding a standout protective antibody
The team set out to find an antibody with three key traits: it had to be extremely potent, work against many different HMPV strains, and leave the virus little room to evolve resistance. They focused on the virus’s fusion protein, a spring-loaded structure on the viral surface that must change shape to allow the virus to merge with and enter lung cells. Using a “bait and switch” strategy, they fished rare human B cells from blood and spleen samples that could recognize one strain of HMPV but neutralize another, enriching for broadly acting antibodies. Out of more than a thousand candidate cells, they isolated three promising antibodies and found that one—4F11—was exceptionally powerful, blocking infection of cells at concentrations nearly 50-fold lower than a previous leading antibody and working against all four major global HMPV subtypes.
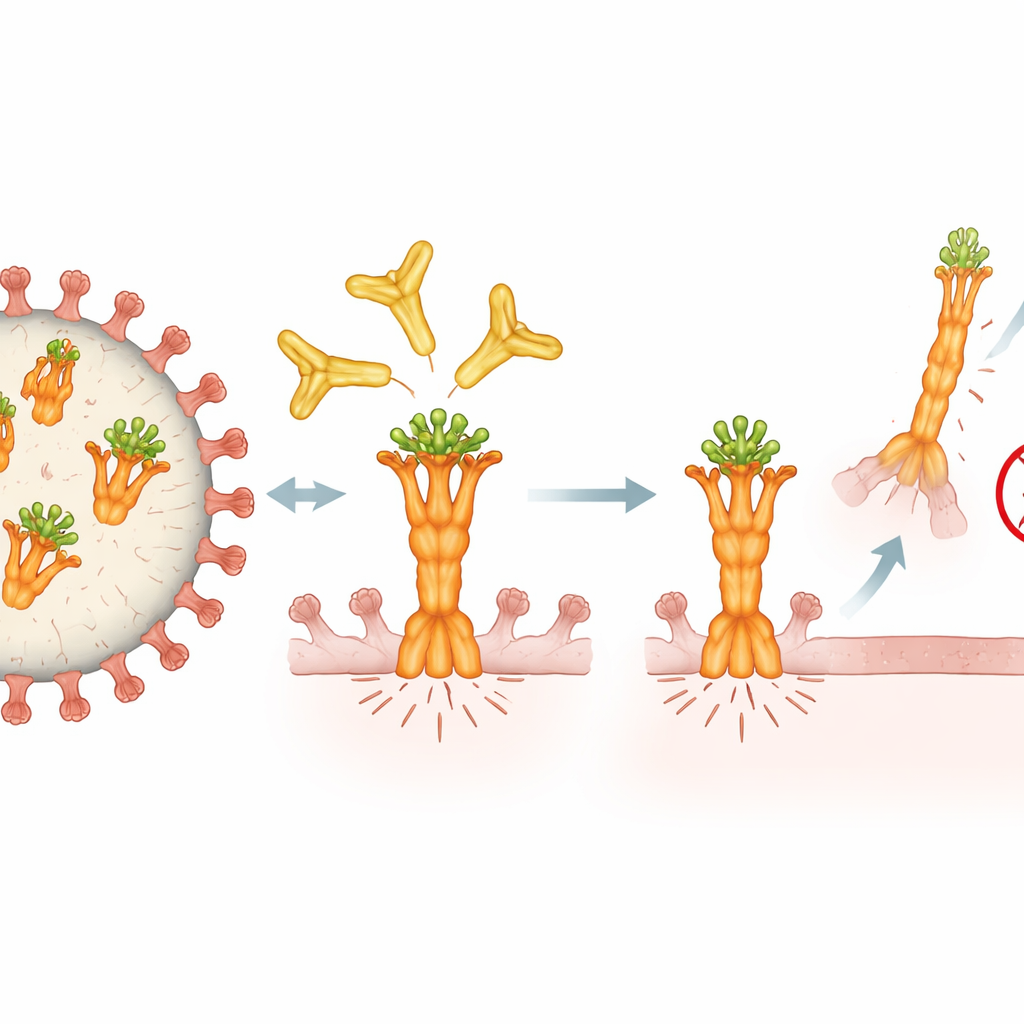
How 4F11 grips the virus’s weak spot
To understand why 4F11 is so effective, the researchers used high-resolution cryo–electron microscopy to visualize how it locks onto the fusion protein. They discovered that 4F11 targets a tiny, conserved patch at the very top of the protein’s prefusion form—the spring before it snaps. Unlike other antibodies that bind three copies per fusion protein, 4F11 sits alone at the apex in a one-to-one arrangement, reaching straight down and even clasping a sugar decoration that shields the surface. This unusual binding angle and reliance on that sugar make 4F11 a unique type of antibody. It recognizes only the prefusion form, which is present on infectious virus, and likely prevents the protein from refolding into the shape needed to pierce and fuse with a host cell.
Can the virus escape—and at what cost?
The team then forced the virus to grow in cell culture in the constant presence of 4F11 to see how easily it could evolve escape. Compared to another antibody control, which quickly drove the emergence of several escape mutations, HMPV had a much harder time slipping past 4F11. Only a single change in the fusion protein, altering one amino acid at position 179, completely disrupted 4F11’s binding. However, viruses carrying this mutation grew far worse than the normal virus, indicating a serious hit to viral fitness. Database searches of hundreds of HMPV sequences showed that this damaging change has never been observed in nature, suggesting that while the virus can, in principle, escape 4F11 in the lab, it pays such a high price in growth that it is unlikely to spread widely in the real world.
Testing protection in an animal model
Laboratory potency still needs to translate into real protection. To test this, the researchers infected golden Syrian hamsters with HMPV and then, a day later, treated them with 4F11—mimicking early therapy rather than prevention. A single low-dose injection of 4F11 sharply reduced virus levels in both the lungs and nasal passages, and at the higher dose it completely eliminated detectable virus from the lungs in most animals. Notably, these doses were lower than those used for several monoclonal antibodies already approved for RSV in humans. By contrast, a previously described antibody that can cross-react with multiple viruses offered little protection at the same dose, highlighting 4F11’s particular strength as a focused HMPV therapy.
What this could mean for patients
Together, these findings position 4F11 as a strong candidate for clinical development. It combines high potency, broad coverage of circulating HMPV strains, a distinctive way of gripping a vulnerable region of the fusion protein, and a resistance landscape in which the virus’s obvious escape route comes with a substantial self-inflicted handicap. For infants, older adults, and immunocompromised patients at high risk of severe HMPV disease, a drug based on 4F11 could one day offer both emergency treatment after infection and added protection alongside or instead of vaccines.
Citation: Harris, E.D., McGovern, M., Pernikoff, S. et al. Development of a potent monoclonal antibody for treatment of human metapneumovirus infections. Nat Commun 17, 2714 (2026). https://doi.org/10.1038/s41467-026-69328-w
Keywords: human metapneumovirus, monoclonal antibody, respiratory infection, fusion protein, antiviral therapy