Clear Sky Science · en
In situ-formed C-N anchors embedded into Sn-based negative electrodes for long-life Na-ion batteries
Why tougher batteries matter
Rechargeable batteries quietly power our phones, laptops and, increasingly, electric cars and grid-scale storage. To store more energy in the same space, engineers are exploring new, higher-capacity materials for the negative electrode (the anode). But these promising materials tend to swell and crack as batteries charge and discharge, causing them to fail long before they should. This study describes a clever way to turn that destructive swelling into an advantage, creating sodium-ion batteries that survive thousands of fast charge–discharge cycles.
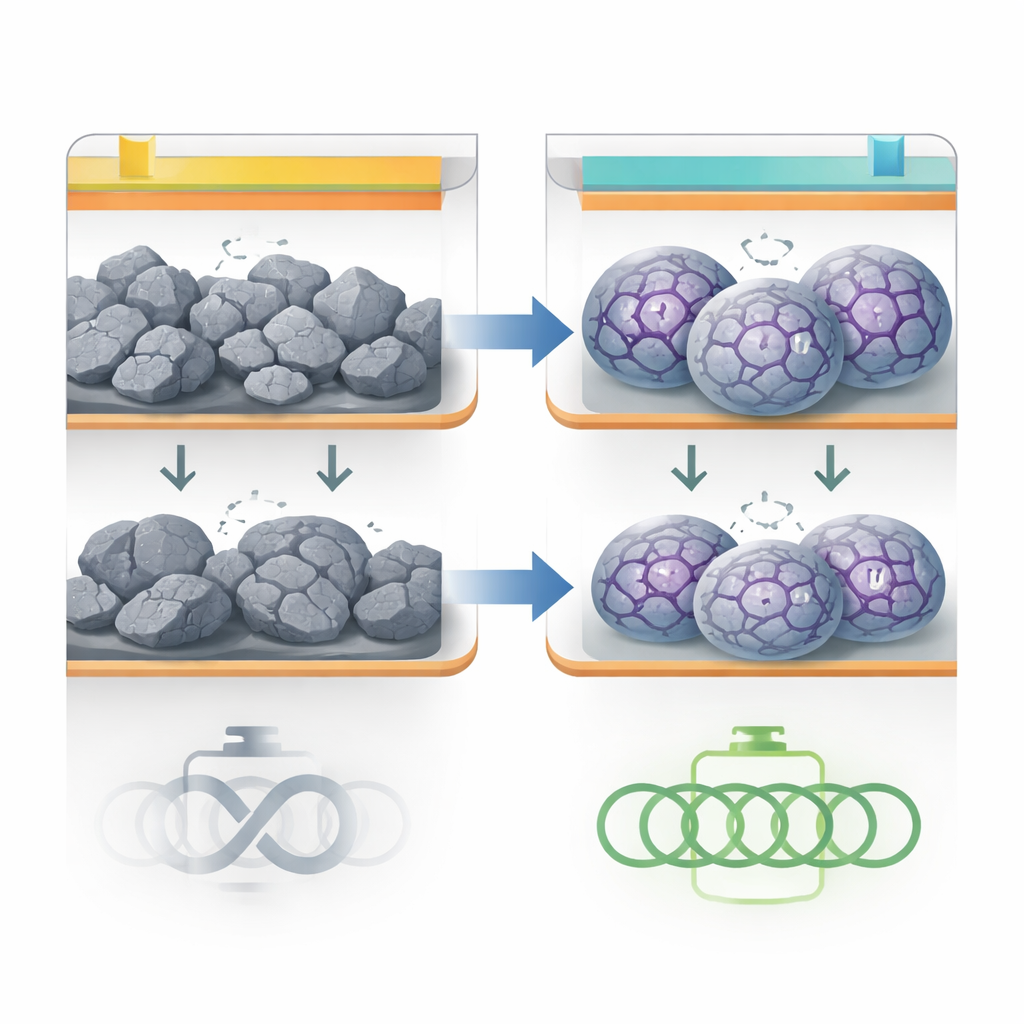
The problem with swelling anodes
Many next-generation batteries rely on metals that react strongly with incoming ions, storing far more charge than today’s common carbon anodes. Tin is one of these metals for sodium-ion batteries. It can theoretically hold several times more charge than graphite, and it is relatively abundant and inexpensive. The catch is that when tin absorbs sodium, it can expand by more than 400 percent in volume. Repeating this expansion and contraction quickly shatters the particles, breaks electrical connections and repeatedly damages the fragile boundary layer where the solid electrode meets the liquid electrolyte. The result is rapid capacity loss and short battery life, which has so far blocked the commercial use of such alloy-type anodes.
A built-in supporting skeleton
The researchers tackled this challenge by building a microscopic skeleton directly inside tin-based particles. They start from tiny tin oxide spheres mixed with an organic molecule called tyrosine. When heated in a controlled way, the tin oxide is reduced to metallic tin, while tyrosine is converted into a carbon-and-nitrogen-rich framework. This framework forms a continuous, nanoscale network that threads through and around the tin, creating what the authors call C–N anchors. Advanced 3D X-ray imaging and electron microscopy show that the final particles contain a uniform distribution of tin intertwined with this C–N network, plus an unusual pattern of alternating crystalline and disordered tin domains that help the material better tolerate stress.
Letting the structure rebuild itself
Beyond simply holding tin in place, the C–N anchors change how the material reacts with sodium. Using in situ X-ray diffraction and solid-state NMR, the team tracked which atomic phases form as the battery charges and discharges. In conventional tin particles, the reaction races all the way to a fully packed final phase, producing huge, damaging volume changes. In the anchored particles, phase changes are slowed and partially “stalled,” so that a mixture of intermediate and final phases remains. This phase hysteresis, imposed by the nanoscale framework, limits abrupt swelling. At the same time, repeated cycling gradually transforms the initially dense tin core into a stable, coral-like porous network that is still supported by the C–N skeleton. Three-dimensional X-ray images over hundreds of cycles reveal that this self-constructed architecture preserves particle integrity despite large, reversible volume swings.
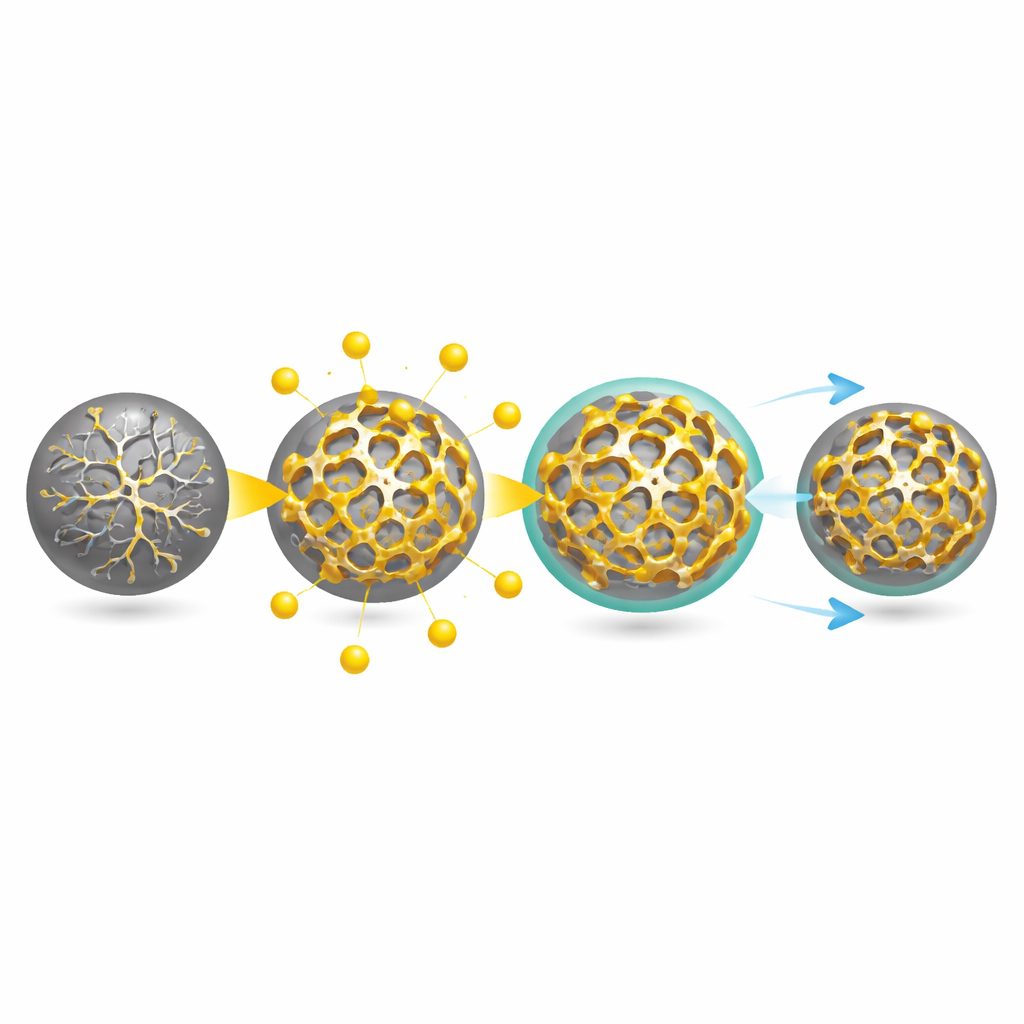
A flexible skin that doesn’t crack
The interface between the electrode and the electrolyte—the so-called solid-electrolyte interphase—is another weak point in swelling anodes. Here, the C–N anchors again play a central role. Chemical analysis shows that nitrogen-containing groups from the C–N network become incorporated into this interphase, chemically tying it to the underlying particle. The layer also develops a mix of organic components, which provide flexibility, and inorganic salt, which adds strength and ion transport. Mechanical tests with atomic force microscopy reveal that this interphase behaves like a viscoelastic skin: it can stretch and relax rather than snap when the particle expands and contracts. In contrast, the interphase on ordinary nano-tin is stiffer, more brittle and prone to repeated rupture and repair, which wastes electrolyte and degrades performance.
From lab concept to durable cells
When tested in half-cells against sodium metal, the engineered tin/C–N anodes delivered high capacities close to theoretical values even at high charge–discharge rates, and maintained most of their capacity after 7,000 cycles at twice the normal current. They also performed strongly in full sodium-ion cells paired with a commercial-type positive electrode and in prototype pouch cells, keeping high capacity over thousands of cycles. In simple terms, by weaving a microscopic scaffold and flexible skin into tin particles, the authors turn the material’s once-fatal tendency to swell into a controlled, self-adjusting breathing motion. This strategy points toward longer-lasting, higher-energy sodium-ion batteries that could one day help store renewable electricity at large scale.
Citation: Li, Y., Fan, X., Wang, L. et al. In situ-formed C-N anchors embedded into Sn-based negative electrodes for long-life Na-ion batteries. Nat Commun 17, 2476 (2026). https://doi.org/10.1038/s41467-026-69319-x
Keywords: sodium-ion batteries, tin anode, energy storage, battery lifetime, electrode design