Clear Sky Science · en
A 3D gut-brain-vascular platform for bidirectional crosstalk in gut-neuropathogenesis
Why Your Gut Might Matter to Your Brain
The idea that “you are what you eat” has taken on a new twist: growing evidence suggests that what happens in your gut can influence the health of your brain. This paper introduces a sophisticated laboratory model that links miniature versions of human intestine, blood vessels, and brain tissue on a single chip. By watching how signals move along this tiny highway, the researchers explore how gut troubles might spark brain diseases like Alzheimer’s and Parkinson’s—and how brain problems might, in turn, send distress back toward the gut.
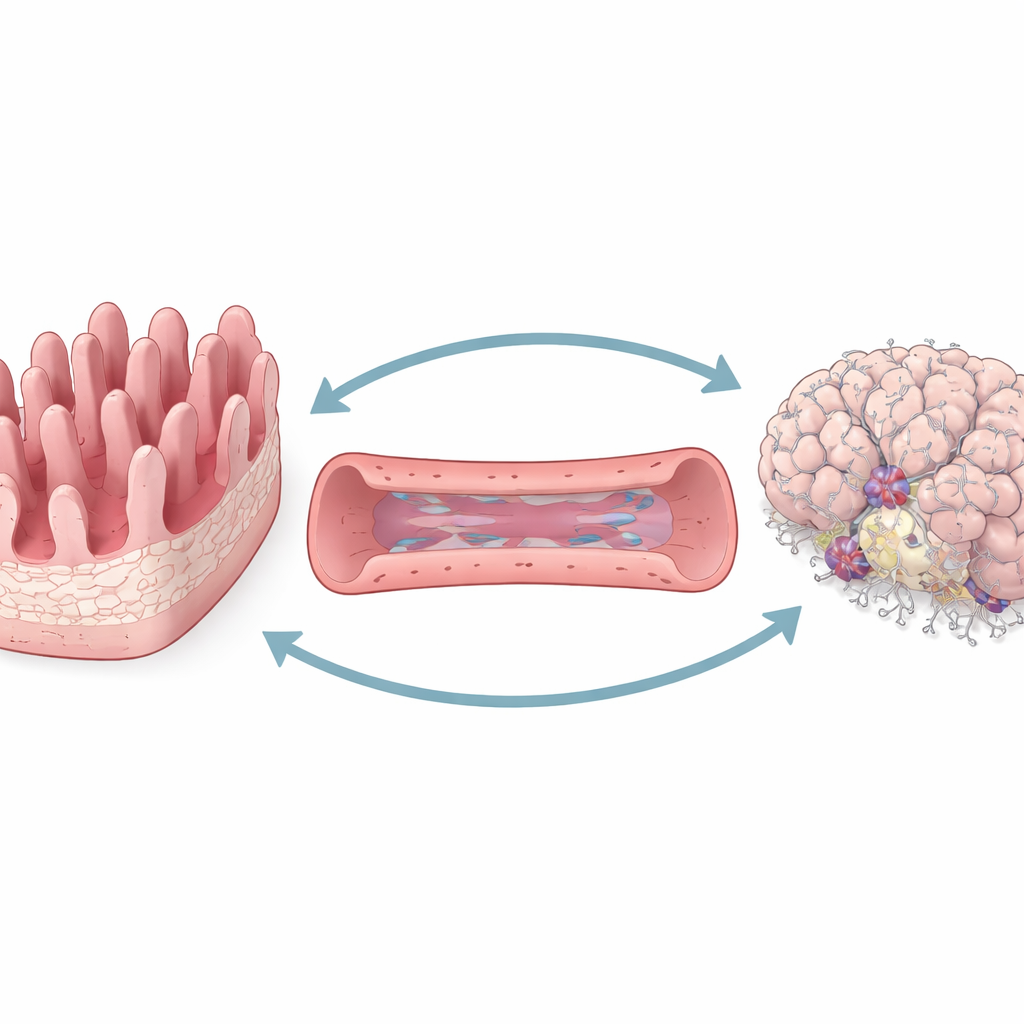
Building a Mini Body on a Chip
To study gut–brain communication in a controlled way, the team engineered a three-part “organ-on-a-chip” platform. One compartment mimics the inner surface of the intestine, complete with finger-like projections that form a barrier between the gut contents and the rest of the body. A second compartment represents blood vessels lined with brain-like vessel cells. The third holds living human neurons and support cells arranged to resemble a small piece of brain. Fluid can flow between these compartments, allowing molecules to travel much as they would through circulation in the body.
Making the Model Act Like Real Tissue
The chip is designed so that cells experience gentle, rocking flow rather than sitting in a static dish. This motion encourages the gut layer to develop thicker, more realistic folds and tighter junctions between cells, making it less leaky. The vessel layer responds by stretching out in the direction of flow, a hallmark of healthy blood vessels, and shows strong barrier behavior against large molecules. In the brain compartment, neurons form active networks that send rhythmic calcium signals, a sign of electrical activity, while support cells wrap around nearby vessels as they do in real brain tissue. Together, these features show that the three regions behave more like they do inside the body than in traditional flat cell cultures.
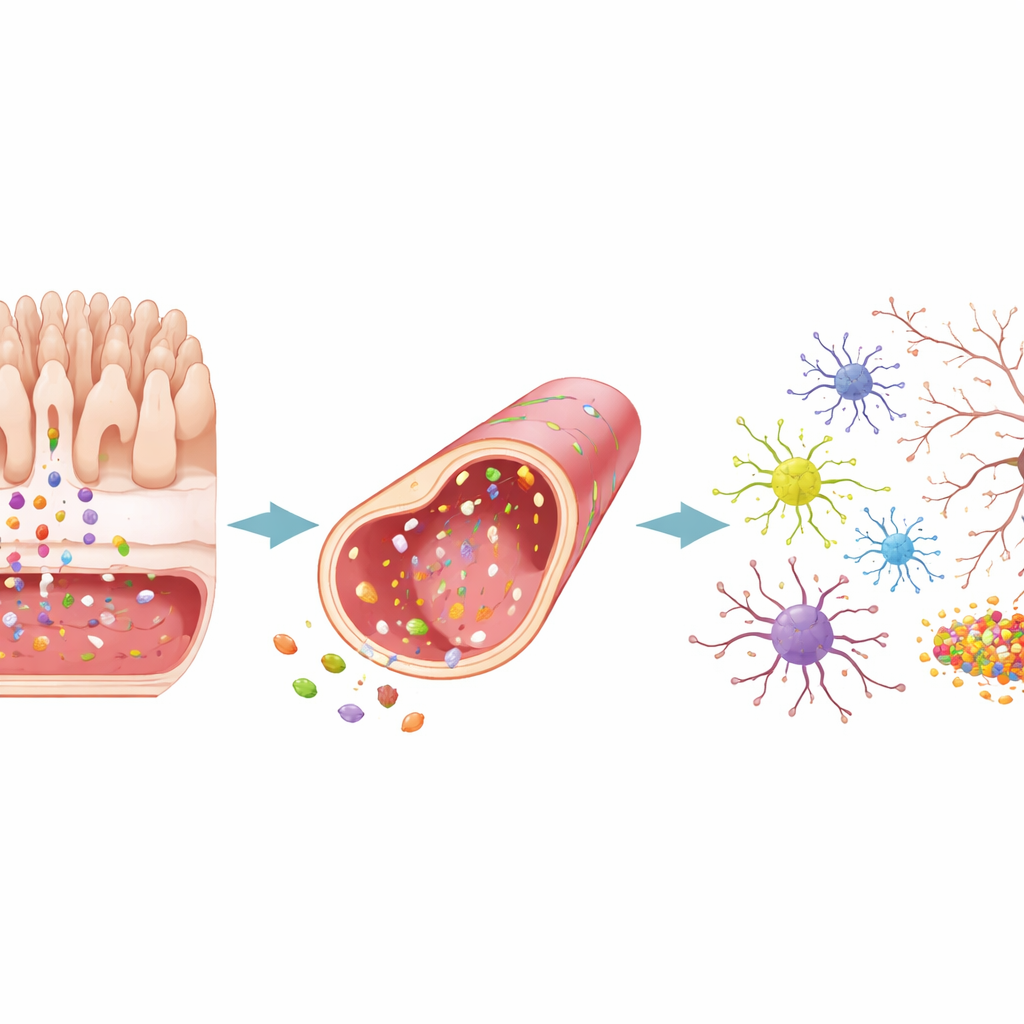
When Gut Irritants Spark Brain Trouble
To model gut-driven disease, the researchers added bacterial products—either material collected from E. coli cultures or a purified toxin called LPS—to the gut side of the chip. These stimuli weakened the gut barrier, allowing more large molecules and bacterial traces to slip through into the vessel compartment and then onward to the brain compartment. As leakage increased, the vessel wall became less tight, and star-shaped brain support cells switched into a reactive, alarmed state. In the brain-like tissue, harmful protein fragments linked to Alzheimer’s and Parkinson’s accumulated, protective connections between neurons declined, and inflammatory signals surged. This sequence, starting with a disturbed gut and ending with stressed brain cells, mirrors patterns seen in animal studies and in patients.
When Brain Disease Sends Signals Back
The team then flipped the direction of the experiment. They created Alzheimer’s-like conditions by engineering brain cells on the chip to overproduce sticky amyloid fragments, and Parkinson’s-like conditions by exposing them to clumps of the α-synuclein protein. In both scenarios, toxic proteins built up first in the brain compartment and then appeared in the vessel and gut regions. The brain tissue released waves of inflammatory molecules, lost synaptic connections, and showed other hallmarks of degeneration. These brain-derived danger signals strongly disturbed the vessel layer, loosening its barrier and activating nearby support cells. The gut layer, however, showed only modest structural changes: its surface became somewhat altered and more “activated,” but its tight junctions largely held, and leakage increased only slightly.
What This Means for Future Treatments
In everyday terms, this chip suggests that a sick gut can drive brain inflammation and damage more directly than a sick brain disrupts the gut. Gut-derived bacterial products and inflammation appear able to weaken both vessels and brain tissue, while brain-origin inflammation mainly targets the blood vessels and only subtly reshapes the intestine. Because the entire system is built from human cells, it offers a promising testbed for therapies aimed at calming gut inflammation, sealing barriers, or blocking harmful protein spread. Ultimately, platforms like this may help doctors pinpoint which patients could benefit most from gut-focused strategies to slow or prevent neurodegenerative diseases.
Citation: Tran, M., Jeong, H.W., An, M. et al. A 3D gut-brain-vascular platform for bidirectional crosstalk in gut-neuropathogenesis. Nat Commun 17, 2504 (2026). https://doi.org/10.1038/s41467-026-69318-y
Keywords: gut brain axis, organ on a chip, neuroinflammation, Alzheimer’s disease, Parkinson’s disease