Clear Sky Science · en
Preoptic kisspeptin-nNOS-GnRH (KiNG) neuronal network regulates LH rhythmicity through activation-inhibition in mice
Why this brain story matters for fertility
Ovulation and fertility depend on a tiny group of brain cells that release a master hormone in pulses and occasional surges. When this timing goes wrong, reproduction can fail. This study reveals how two kinds of signaling molecules in the mouse brain, one that turns activity on and another that quietly turns it off again, work together to shape these hormone rhythms. Understanding this hidden timing circuit may eventually help explain certain forms of infertility and suggest new ways to treat reproductive disorders.
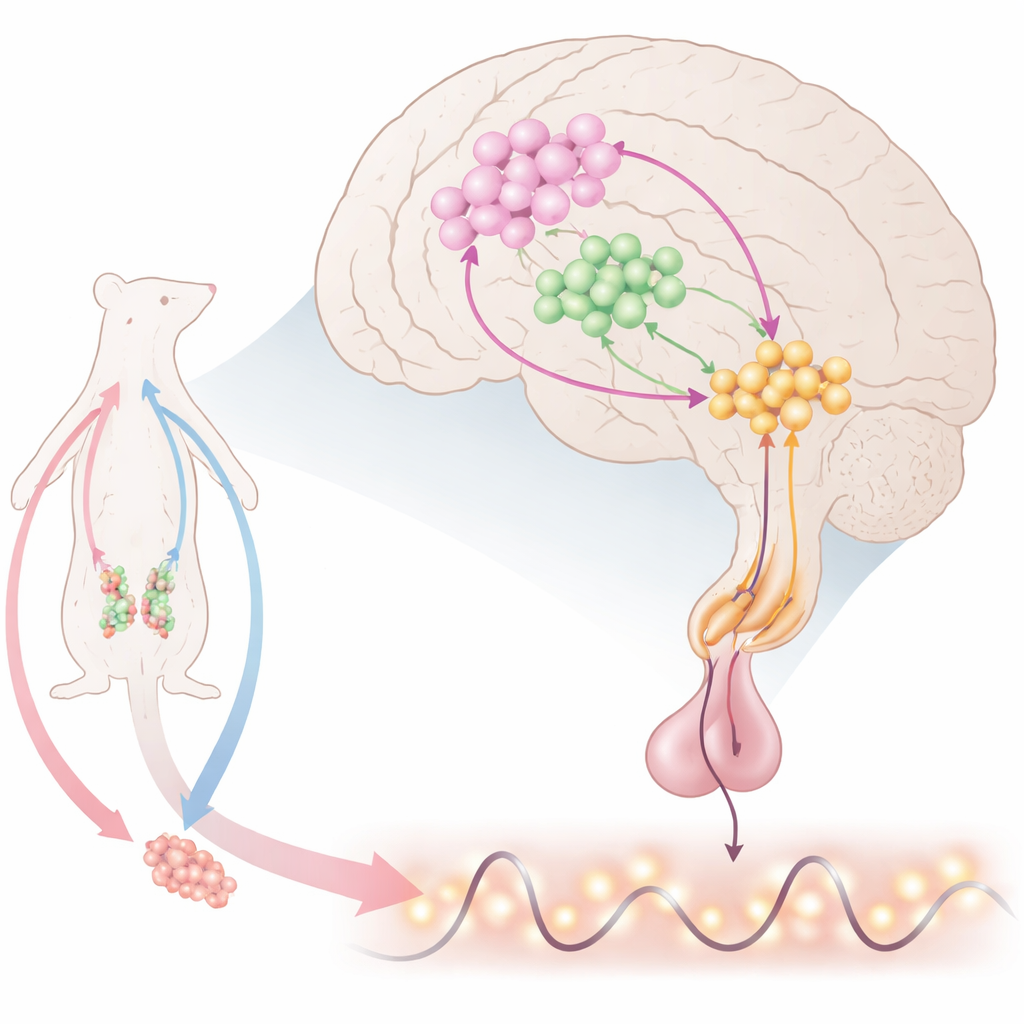
The brain’s timekeeper for reproduction
Deep within the brain, special nerve cells release gonadotropin-releasing hormone (GnRH), which tells the pituitary gland to send out luteinizing hormone (LH) and follicle-stimulating hormone. These, in turn, control the ovaries and testes. GnRH does not trickle out steadily; instead, it appears as regular pulses and, in females, a large pre-ovulatory surge. Scientists know that many signals feed into GnRH cells, but the exact combination that produces both pulses and surges has remained unclear. This work focuses on a small region at the front of the hypothalamus, where GnRH cells mingle with two other important players: kisspeptin-producing neurons that excite GnRH, and neurons that make nitric oxide (NO), a gaseous signal that can diffuse over short distances.
An on–off partnership between two signals
The researchers propose a three-part “KiNG” network, made of kisspeptin, NO-producing (nNOS) neurons, and GnRH cells. Kisspeptin is a powerful activator that can drive GnRH and LH release, but constant stimulation actually shuts the system down, suggesting that an internal brake is needed. Using highly sensitive molecular probes, genetic tools, and brain-slice recordings in mice, the team shows that kisspeptin does more than excite GnRH neurons directly. It also activates nearby nNOS neurons in the preoptic area, especially in a region called the OV/MePO. When kisspeptin levels rise, these nNOS cells produce bursts of NO, which trigger production of a second messenger, cGMP, in surrounding cells, including GnRH neurons themselves. This NO–cGMP signal dampens GnRH firing, providing an inbuilt off-switch to the kisspeptin drive.
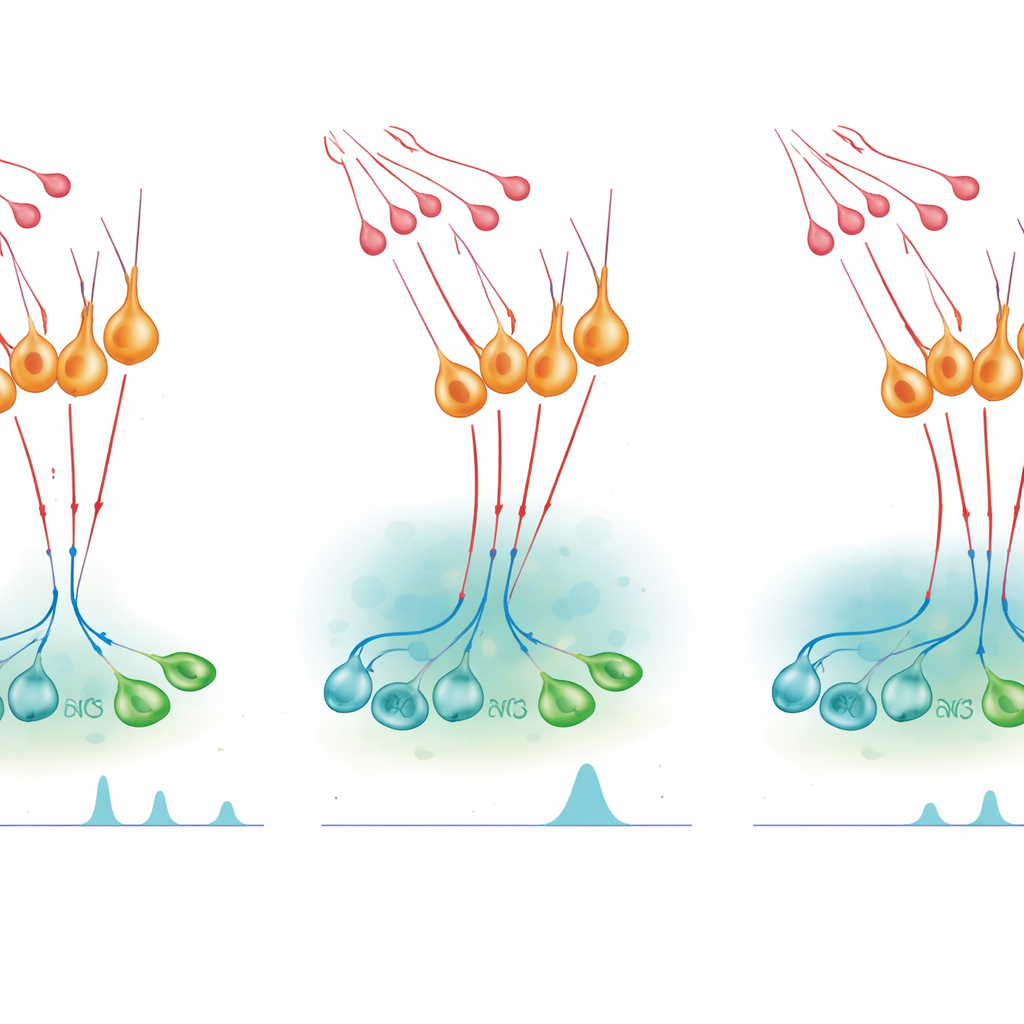
Turning hormone surges on and trimming them back
To test how this microcircuit affects hormone release, the team used chemogenetic switches—engineered receptors activated by an otherwise inert drug—to turn nNOS neurons on or off in living mice. Artificially activating nNOS cells during a normally quiet phase of the female cycle was enough to produce a surge-like rise in blood LH, and the same was true in males. Blocking NO production erased this effect, showing that NO from these neurons is necessary for the surge. Yet, when the scientists inhibited nNOS neurons or reduced their kisspeptin sensitivity, a shot of kisspeptin produced an exaggerated and prolonged LH rise. Pharmacological drugs that block NO synthesis had a similar amplifying effect, whereas a drug that prolongs cGMP signaling shortened the kisspeptin response and reduced later LH output. Together, these experiments indicate that NO both helps synchronize GnRH neurons to create surges and limits how long kisspeptin can keep them firing.
A flexible rhythm across the ovarian cycle
The study also reveals that this on–off balance changes across the female cycle. Using a sensitive RNA detection method, the authors found that more nNOS neurons express the kisspeptin receptor on the day before ovulation, when estrogen levels are high and the LH surge occurs. During the quieter stages, fewer nNOS neurons respond to kisspeptin, producing only small, local amounts of NO that gently restrain GnRH pulses without stopping them. On the surge day, stronger kisspeptin input and heightened receptor expression massively boost NO output, allowing NO to spread further and briefly silence many GnRH neurons at once. As the NO signal fades, these cells rebound together, generating the large, coordinated LH surge needed for ovulation.
What this means for human health
In plain terms, this work shows that the same signal that starts GnRH neurons—kisspeptin—also recruits a delayed braking system through NO, ensuring that hormonal bursts are strong but not runaway. The KiNG network therefore acts as both the accelerator and the brake for reproductive timing, tuning GnRH pulses during most of the cycle and shaping the surge that triggers ovulation. Because genetic changes in kisspeptin and NO pathways have been found in people with reproductive problems, understanding this finely balanced circuit could inform new approaches to diagnose and treat infertility and related conditions.
Citation: Delli, V., Moulinier, M., Lazaridou, AM. et al. Preoptic kisspeptin-nNOS-GnRH (KiNG) neuronal network regulates LH rhythmicity through activation-inhibition in mice. Nat Commun 17, 2558 (2026). https://doi.org/10.1038/s41467-026-69316-0
Keywords: reproductive hormones, hypothalamus, kisspeptin, nitric oxide, ovulation timing