Clear Sky Science · en
Dual spillover of carbon monoxide and hydrogen initiates tandem urea electrosynthesis
Turning Waste into Plant Food
Modern farming depends heavily on urea fertilizer, but making it the usual way burns a lot of fossil fuel and releases large amounts of carbon dioxide. This study explores a cleaner route: using electricity from potentially green sources to turn two waste streams—carbon dioxide from exhaust gases and nitrate pollution from water—directly into urea. Along the way, the system also produces formate, a useful chemical, showing how future factories might clean up emissions while manufacturing essential products.
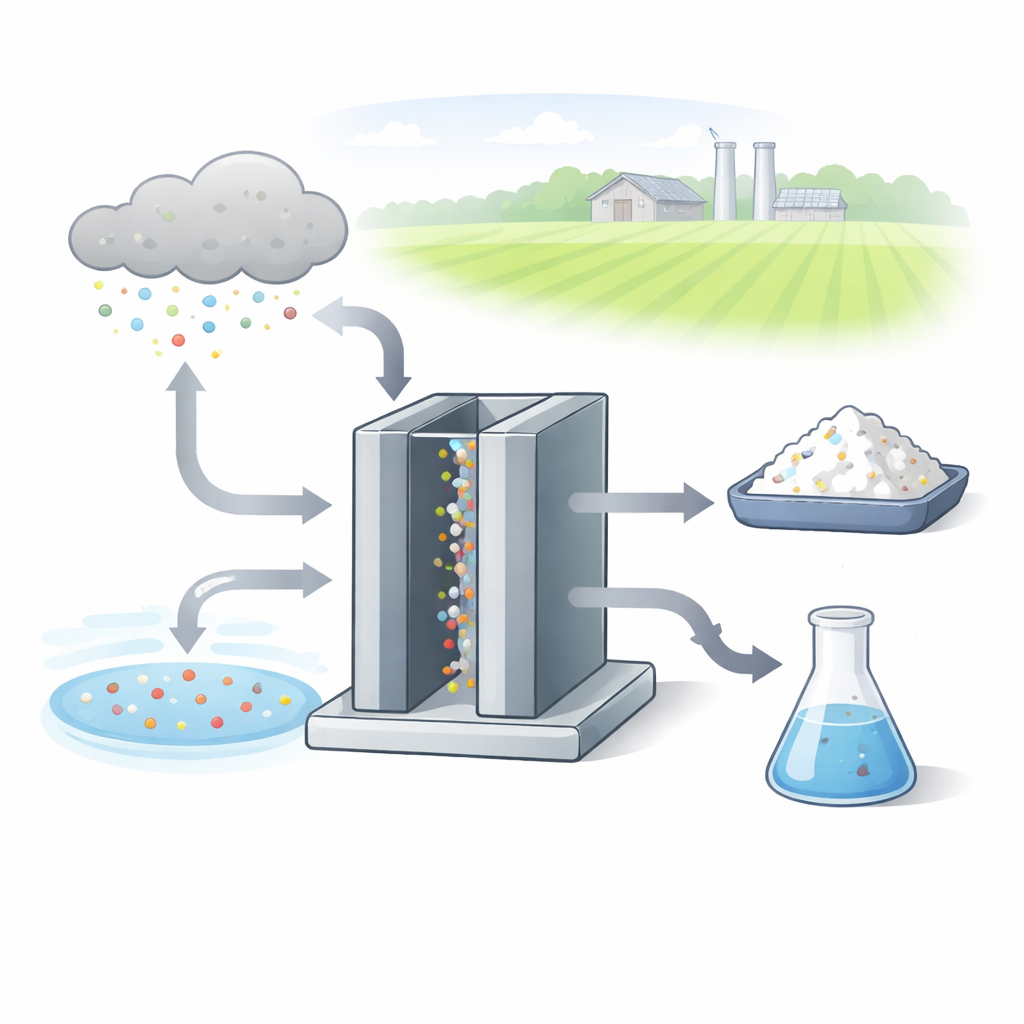
A New Kind of Fertilizer Factory
Today’s urea plants rely on the century‑old Haber–Bosch process, which first makes ammonia and then reacts it with carbon dioxide. Both steps are energy‑hungry and carbon‑intensive. The researchers instead build an “electrochemical” factory: an enclosed device where electricity drives chemical changes in water. One inlet supplies carbon dioxide, the other supplies nitrate, which can come from polluted waterways or sustainably produced nitrogen compounds. Inside, specially designed metal particles on an electrode surface coax these simple molecules to rearrange and join carbon and nitrogen atoms into urea at room temperature and modest pressures.
A Team Effort Between Two Metals
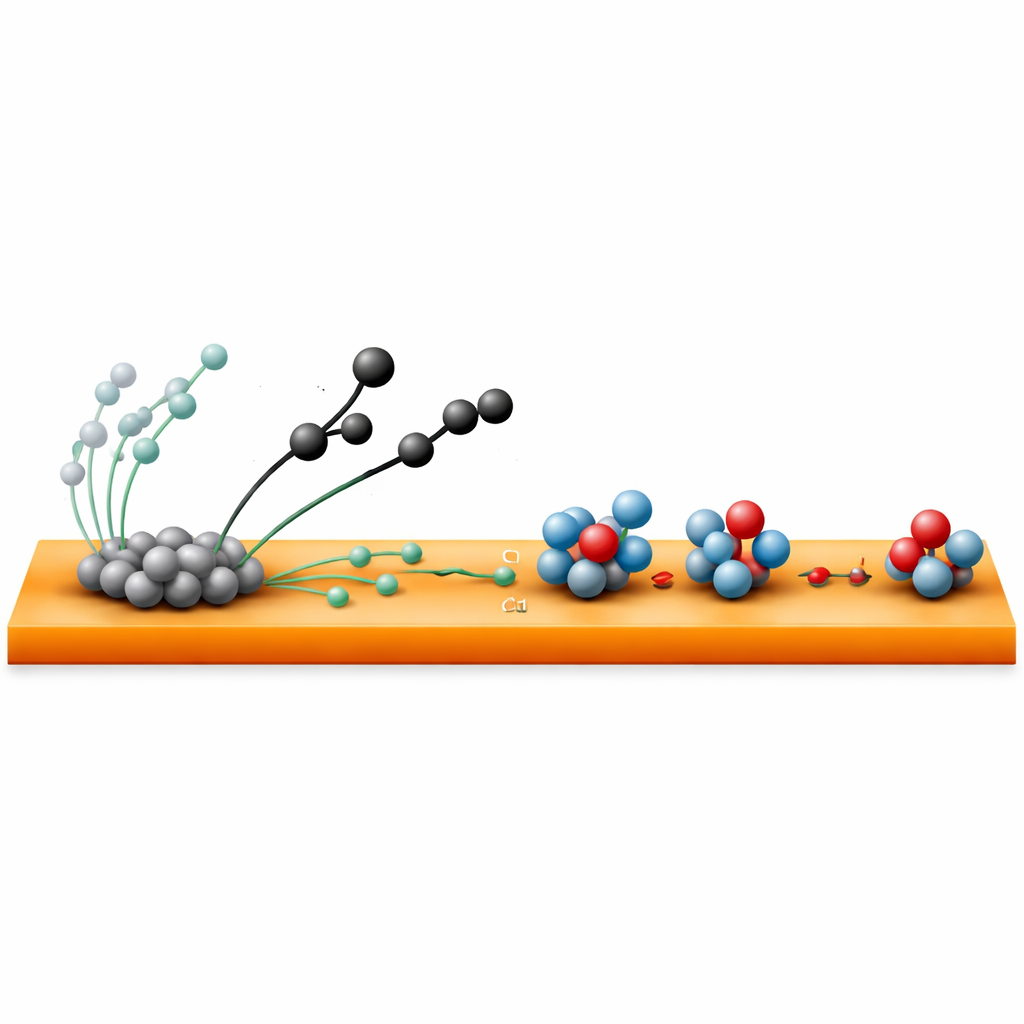
The heart of the work is a tandem catalyst made from copper sheets decorated with tiny particles of palladium hydride, a palladium metal that stores hydrogen in its crystal lattice. Each metal plays a different role. Palladium hydride is very good at turning carbon dioxide into a reactive carbon‑containing fragment, while copper excels at turning nitrate into nitrogen‑containing fragments. Normally these fragments struggle to find each other and to receive the right number of hydrogen atoms, so the desired urea molecule forms slowly and many side products appear. Here, the two components are placed so close together that they can continuously exchange reactive species across their shared surface.
Spillover: Handing Off the Reactive Pieces
The key innovation is a “dual spillover” effect. First, the palladium hydride particles create carbon‑based fragments that migrate, or spill over, from the palladium surface onto the nearby copper. Second, hydrogen stored inside the palladium hydride seeps out and travels to the copper as well. On the copper, nitrogen‑bearing fragments from nitrate are already in place. The incoming carbon fragments join with these nitrogen species to create an early carbon–nitrogen building block, while the spilled‑over hydrogen helps gently finish and stabilize the newly formed molecule. Careful experiments and computer simulations show that this hand‑off dramatically lowers the energy hurdles that used to slow down the critical bond‑forming and finishing steps.
Performance, Durability, and Climate Impact
Because the tandem catalyst can manage each step efficiently, it produces urea at high rates and with over 60% of the incoming electrical charge going into the desired product—among the best values reported so far. The team then scales up the concept into a larger flow cell that runs continuously for more than a week. In this device, the same palladium‑copper material is used at both electrodes: on one side to make urea from carbon dioxide and nitrate, and on the other side to turn methanol into formate, another valuable chemical. Economic modeling suggests that, especially when low‑cost electricity is available, the income from formate can offset most of the cost of urea production. A life‑cycle analysis further indicates that this route could roughly halve the carbon footprint of urea compared with today’s industrial process.
Why This Matters for a Greener Future
This work shows that smartly pairing materials so they can share reactive fragments across their interface can unlock cleaner ways to make bulk chemicals. By turning carbon dioxide and nitrate—two major pollutants—into urea and formate using electricity, the system points toward fertilizer production that could be powered by renewable energy and integrated with pollution control. While more improvements are needed before such devices become standard in industry, the dual‑spillover strategy provides a promising blueprint for designing future catalytic systems that are both efficient and climate‑friendly.
Citation: Li, Y., Han, B., Liu, Y. et al. Dual spillover of carbon monoxide and hydrogen initiates tandem urea electrosynthesis. Nat Commun 17, 2506 (2026). https://doi.org/10.1038/s41467-026-69307-1
Keywords: electrochemical urea synthesis, tandem catalysis, carbon dioxide utilization, nitrate valorization, palladium copper catalyst