Clear Sky Science · en
STING activation induces cytotoxic and immune responses in meningiomas via inflammatory cell death pathways
Turning the Body’s Alarms Against Brain Tumors
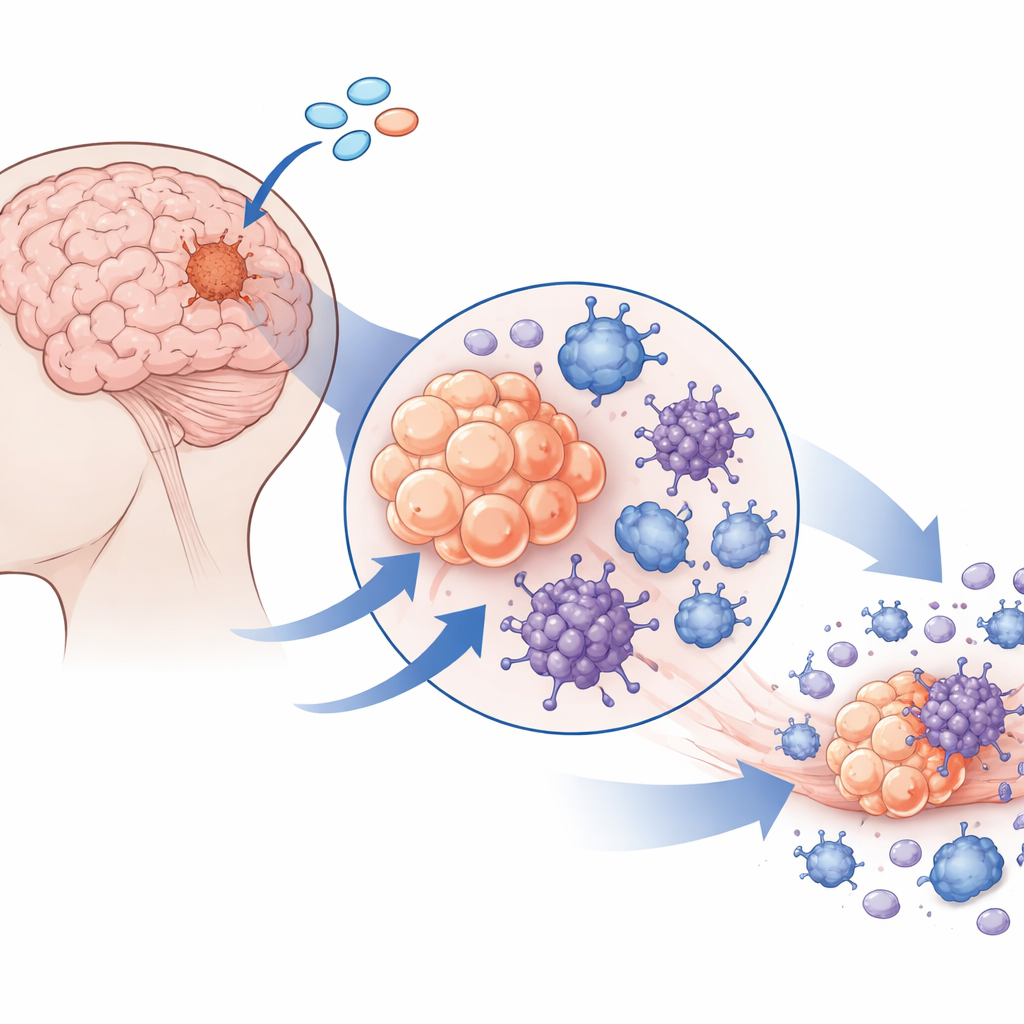
Meningiomas are among the most common brain tumors, and while surgery and radiation often help, some tumors keep coming back. These stubborn growths can cause seizures, headaches, and disability, yet there are no widely accepted drug treatments. This study explores a way to make the tumor’s own cells and nearby immune cells sound a powerful internal alarm, using a molecule that switches on an innate danger-sensing system called STING. By doing so, the researchers aim not only to kill tumor cells directly, but also to awaken the immune system and soften the tumor’s tough scaffolding.
A Tumor Hidden in a Quiet Immune Neighborhood
The authors first mapped the cellular “neighborhood” inside human meningiomas using single-cell sequencing and spatial imaging. They found that these tumors are packed with innate immune cells, especially macrophages, while classic tumor-fighting T cells and natural killer (NK) cells are sparse and often exhausted. Surrounding the tumor cells is a dense shell of collagen and other matrix proteins that form an armored, layered structure. Many macrophages in these regions carry inhibitory receptors that are switched on by collagen, creating a heavily suppressed environment where immune cells are present but held back rather than unleashed against the cancer.
Discovering a Switch That Tumor Cells Still Carry
Next, the team searched for pathways that might flip this suppressive setting into an inflamed, tumor-hostile one. They focused on STING, a sensor that normally responds to misplaced DNA inside cells and triggers antiviral and inflammatory responses. Unlike many other brain tumors, meningioma cells showed unusually high STING levels. This was traced to open chromatin and low DNA methylation at the STING gene, meaning the gene is epigenetically primed to be active. STING was not only abundant in tumor cells but also in nearby blood vessel cells and macrophages, suggesting that a single drug could simultaneously affect many key players in the tumor microenvironment.
Forcing Tumor Cells Into Fiery Self-Destruction
The researchers treated freshly removed patient meningioma samples with a potent STING agonist called 8803. Even when immune cells were stripped away, tumor cells died, proving a direct effect. By adding drugs that selectively block different cell death routes, and by reading out gene activity, they showed that 8803 did not simply cause quiet apoptosis. Instead, it pushed meningioma cells into several inflammatory forms of death—pyroptosis, necroptosis, and ferroptosis—which burst cells open rather than tidily dismantling them. A key step was the activation and cleavage of a protein called gasdermin D, which forms pores in the cell membrane. Electron microscopy revealed tumor cells riddled with membrane holes and damaged mitochondria, and blocking reactive oxygen species reduced the killing, tying mitochondrial stress to this fiery demise.
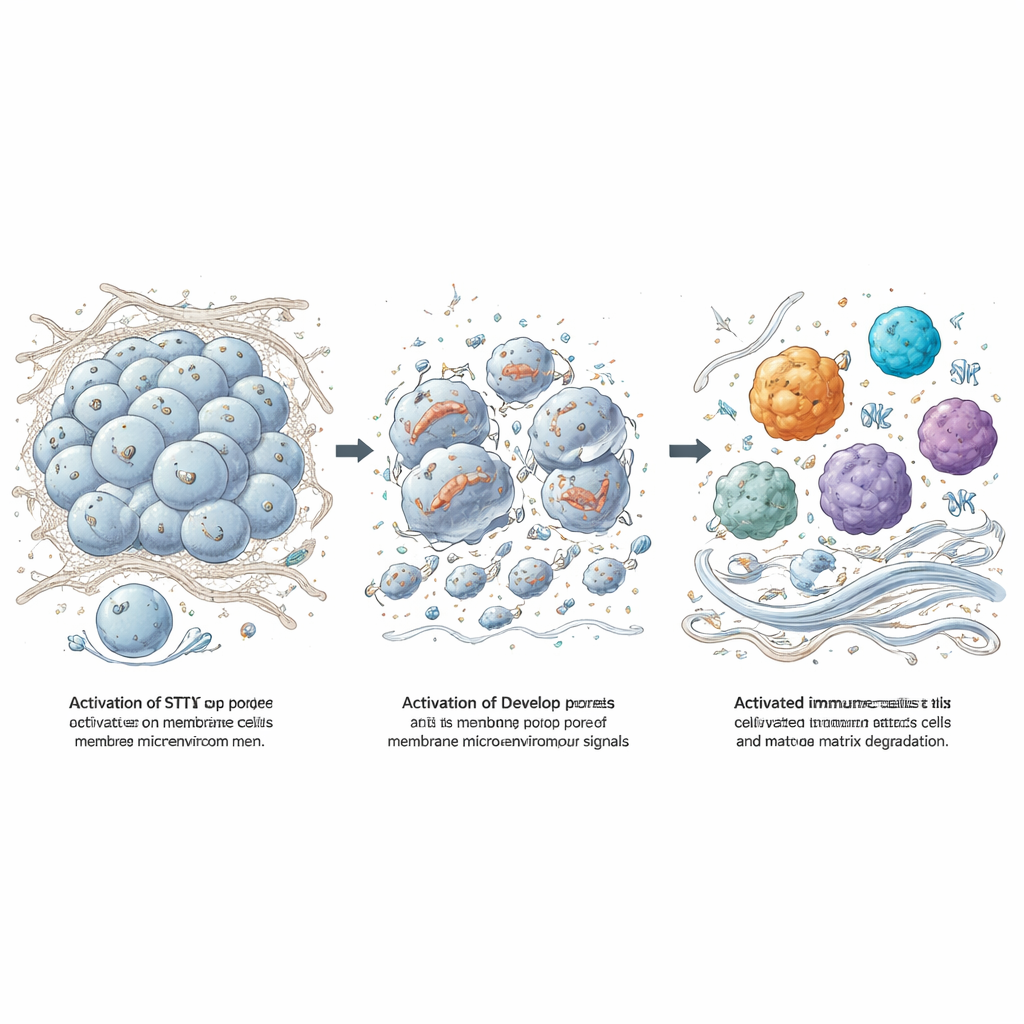
From Tumor Debris to Immune Awakening and Matrix Erosion
When tumor cells die in this dramatic way, they spill “danger” molecules that can rouse immune cells. The team exposed mouse macrophages to lysed meningioma cells and saw a surge in inflammatory factors and enzymes that degrade collagen, especially matrix metalloproteinase-3 (MMP-3). By working with mice lacking specific signaling adaptors, they showed that toll-like receptors, another danger-sensing system, are crucial for this response, while STING activation by 8803 adds its own layer of immune stimulation. In mouse meningioma models, direct injection of 8803 into tumors shrank or stabilized growth, extended survival, and converted the tumor site from a quiet, collagen-rich mass into one packed with active macrophages, NK cells, T cells, and markedly reduced collagen staining.
Why This Approach Could Change Treatment for Patients
Overall, the study shows that meningiomas harbor a built-in vulnerability: their tumor cells retain an accessible STING switch that, when flipped by 8803, drives them into explosive, inflammatory cell death. This process both kills cancer cells and floods the area with alarm signals that recruit and energize immune cells, while also helping chew away the collagen scaffold that contributes to mass effect. Because meningiomas are typically localized rather than widely metastatic, and because standard options are limited for recurrent disease, a locally delivered STING agonist like 8803 could offer a new, targeted way to both debulk tumors and reprogram their immune environment for lasting control.
Citation: Youngblood, M.W., Tripathi, S., Najem, H. et al. STING activation induces cytotoxic and immune responses in meningiomas via inflammatory cell death pathways. Nat Commun 17, 2685 (2026). https://doi.org/10.1038/s41467-026-69296-1
Keywords: meningioma, STING pathway, brain tumor immunotherapy, inflammatory cell death, tumor microenvironment