Clear Sky Science · en
Explainable AI-based analysis of human pancreas sections identifies traits of type 2 diabetes
Why this research matters for everyday health
Type 2 diabetes affects hundreds of millions of people, yet doctors still struggle to see clear, physical signs of the disease inside the pancreas, the organ that makes insulin. This study uses powerful artificial intelligence, paired with advanced microscopy, to read subtle patterns in human pancreatic tissue that are invisible to the naked eye. By doing so, it reveals new structural traits linked to type 2 diabetes and points to fresh ideas for diagnosis, prevention, and treatment.
Looking inside the pancreas in unprecedented detail
Traditionally, scientists have examined pancreas tissue from deceased donors to look for changes associated with type 2 diabetes, such as scarring, fat deposits, or loss of insulin-producing cells. These studies have produced a long list of possible abnormalities, but none has been reliable enough for pathologists to look at a microscope slide and confidently say whether a person had diabetes. In this work, the researchers assembled a unique collection of ultra–high-resolution images from pancreas samples taken during surgery from 100 living donors, 35 with type 2 diabetes and 65 without. Each sample was stained in multiple ways to highlight different cell types: hormone-producing cells in the islets, blood vessels, fat cells, and nerve fibers. This rich visual dataset, far too complex for a human to process fully, is exactly where artificial intelligence excels. 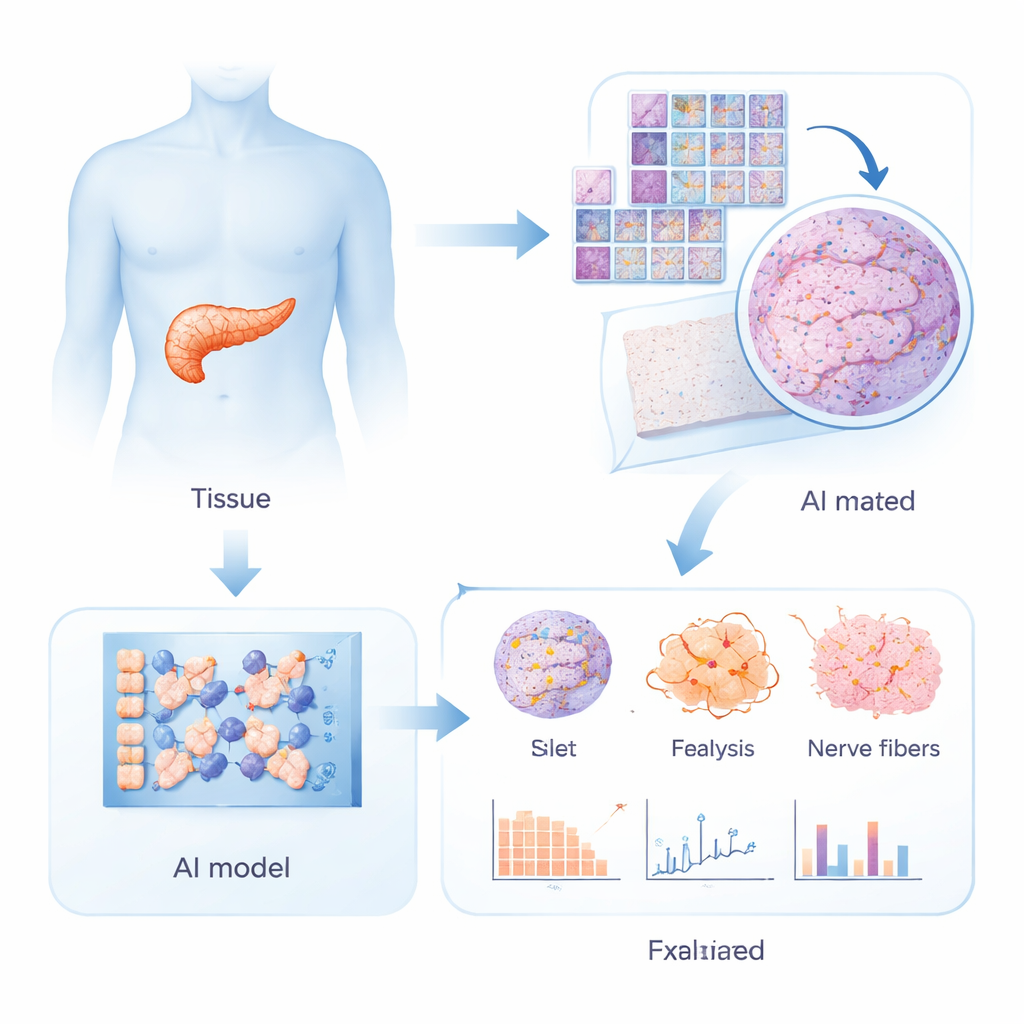
Training AI to recognize the diabetic pancreas
The team trained deep learning models to distinguish between tissue from people with and without type 2 diabetes. Because each whole-slide image contains billions of pixels, they first divided the slides into many small patches. A pretrained vision transformer extracted features from each patch, and a specialized "multiple instance learning" classifier then combined information across all patches from a slide to decide whether it came from a diabetic or non-diabetic donor. To avoid overfitting, they used repeated cross-validation and then averaged the predictions of 15 separate models. The best performance came from multiplex fluorescence images that simultaneously highlighted alpha cells, delta cells, and nerve fibers, reaching an area-under-the-curve of 0.956—strong evidence that the AI could reliably "see" diabetes-related patterns that pathologists have not been able to formalize.
Making the black box explain itself
However, prediction alone was not the goal; the authors wanted biological insight. They therefore applied explainable AI techniques that show which regions and pixels the model relied on when making its decisions. Attention maps highlighted the most influential areas across entire slides, while attribution methods zoomed down to the level of individual cells and structures. To turn these colorful heatmaps into numbers, the team trained separate segmentation networks that could automatically outline islets, fat cell clusters, and connective (fibrotic) tissue. They then quantified features such as islet size, total fat area, number of fat clusters, distance between islets and nearby fat, and extent of fibrotic tissue, and analyzed how these "histologic biomarkers" related to diabetes status and insulin secretion, while accounting for age, sex, body mass index, and other clinical factors. 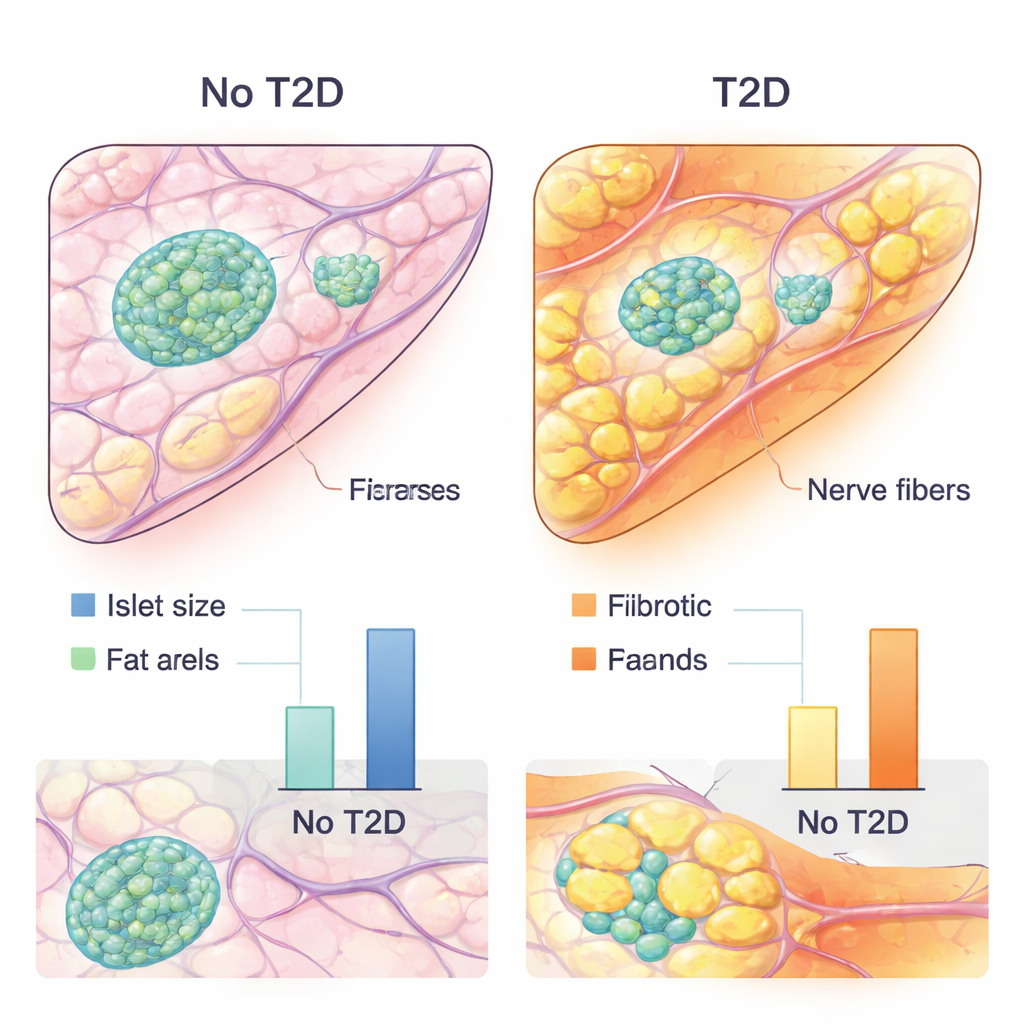
New clues: fat, nerves, scarring, and shrinking islets
The AI-based analysis uncovered several consistent traits of the diabetic pancreas. People with type 2 diabetes tended to have smaller islets and larger clusters of fat cells inside the pancreas. Importantly, islets in diabetic donors sat closer to these fat deposits, suggesting more direct local influence of the fat tissue on hormone-producing cells. The amount of connective, scar-like tissue was also higher in diabetes and was linked to poorer insulin secretion. At the same time, the AI models placed surprisingly strong emphasis on structures stained for tubulin beta 3, a marker of nerve fibers, especially when these fibers coursed through or near islets. This points to changes in pancreatic innervation—how nerves interface with islet cells—as a potentially important and underappreciated contributor to type 2 diabetes. Together, these findings suggest that it is not just the beta cells themselves that matter, but also their neighborhood: nearby fat cells, scarring, blood vessels, and nerves all seem to shape islet health.
What this means for future care
For non-specialists, the key message is that type 2 diabetes leaves a subtle but detectable footprint in the architecture of the pancreas. By combining high-resolution imaging with explainable AI, this study translates those patterns into measurable features—such as islet size, fat distribution, nerve presence, and fibrosis—that correlate with diabetes and with how well the body can still produce insulin. While this approach is not yet a diagnostic tool for routine care, it offers a powerful roadmap for discovering new drug targets and refining our understanding of how diabetes develops. In the long run, insights from such AI-guided tissue analysis may help doctors better predict who is at risk, monitor disease progression, and design therapies that protect not only beta cells but also the surrounding pancreatic environment.
Citation: Klein, L., Ziegler, S., Gerst, F. et al. Explainable AI-based analysis of human pancreas sections identifies traits of type 2 diabetes. Nat Commun 17, 1558 (2026). https://doi.org/10.1038/s41467-026-69295-2
Keywords: type 2 diabetes, pancreas, artificial intelligence, histopathology, biomarkers