Clear Sky Science · en
Phyllosilicate adsorption limited phosphorus bioavailability in early ferruginous oceans
Why ancient oceans matter today
Phosphorus is a quiet workhorse of life, building cell membranes, DNA, and the energy-carrying molecules in every organism. Yet on the early Earth, this vital element may have been surprisingly hard for microbes to get. This study explores how common clay-like minerals in iron-rich, oxygen-poor ancient oceans grabbed onto phosphorus, carried it around, and locked much of it away in seafloor sediments. Understanding that hidden traffic helps explain why life and oxygen took so long to transform our planet.

Life’s key ingredient with a supply problem
Today, phosphorus reaches the oceans mainly when rocks weather on land, rivers carry it seaward, and minerals and organisms grab, recycle, and finally bury it in sediments. Most of the phosphorus that living things can actually use is temporarily stuck to mineral surfaces or organic matter, not locked in tough crystals. Billions of years ago, however, Earth’s surface looked very different: the atmosphere lacked oxygen, oceans were rich in dissolved iron, and river and seawater chemistry diverged sharply from the modern case. Scientists have debated whether early oceans were starved of phosphorus or occasionally flooded with it, and what roles clay minerals—thin, sheet-like silicates known as phyllosilicates—played in shuttling and trapping this nutrient.
Experiments that recreate early waters
The authors recreated early river and seawater in the lab under oxygen-free conditions, using realistic mixtures of salts, iron, and dissolved silica. They then measured how much dissolved phosphate (the main dissolved form of phosphorus) stuck to several common phyllosilicates: aluminous clays such as kaolinite and montmorillonite, and iron- and magnesium-rich clays like lizardite and nontronite that form during seafloor alteration of volcanic rocks. Across many tests, adding moderate amounts of dissolved iron in its reduced form, Fe(II), dramatically boosted phosphate adsorption onto these minerals, while high levels of dissolved silica tended to weaken it. Microscopy and spectroscopy confirmed that phosphorus was attaching to existing mineral surfaces rather than forming new phosphate crystals.
How iron helps clays grab phosphorus
Why is Fe(II) so effective? Using molecular simulations, the team showed that divalent metal ions, especially Fe(II), act as nanoscale bridges between negatively charged phosphate groups in the water and the likewise negatively charged clay surfaces. These metals can sit near the mineral surface and simultaneously bind phosphate, overcoming electrical repulsion and anchoring phosphorus to the clay. Fe(II) binds both to phyllosilicates and to phosphate more strongly than calcium or magnesium, the other major divalent ions in seawater, giving it an outsized impact in iron-rich ancient oceans. The simulations also revealed that phosphate species more common at slightly acidic pH bind less strongly, helping explain why adsorption changed with water acidity. Dissolved silica, in turn, competes with phosphate for the same surface sites, edging phosphorus aside when concentrations and pH are high enough.
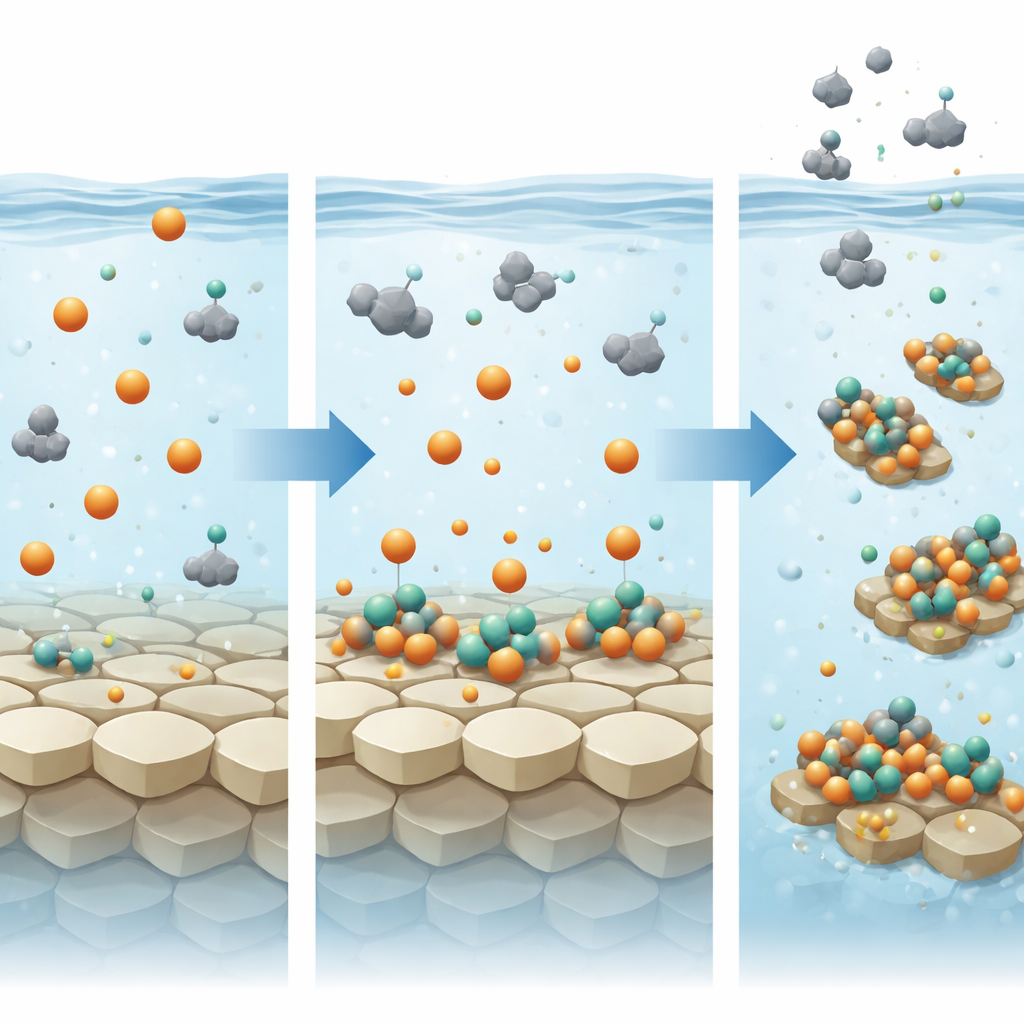
Moving and burying phosphorus on a changing planet
Armed with these mechanistic insights, the authors built simple probabilistic models to scale up from lab beakers to global budgets. As continents rose and weathering intensified late in the Archean Eon, rivers likely produced and transported abundant clay particles. The results suggest that in iron-rich rivers, these clays would have soaked up large amounts of phosphate, becoming the dominant form of biologically accessible phosphorus during transport. Once these particles reached coastal seas, instead of releasing their cargo, the presence of Fe(II), calcium, and magnesium in seawater would have encouraged them to hold on to even more phosphorus and sink rapidly into sediments. Separate simulations show that phyllosilicates generated directly by seafloor weathering of mafic and ultramafic crust also formed a powerful sink for dissolved phosphate, especially when continents were still small and river input was limited.
Implications for early life and oxygen
Putting the pieces together, the study argues that clay minerals in ancient, iron-rich oceans acted as both couriers and vaults for phosphorus. They likely helped move reactive phosphorus from land to sea, but then quickly locked much of it into sediments, where it was slowly transformed into more stable phosphate minerals. This double-edged role would have kept dissolved phosphorus concentrations low, throttling marine productivity and delaying the buildup of oxygen in the atmosphere, even after oxygen-producing microbes evolved. Over time, as Earth’s surface oxidized and iron chemistry shifted, other minerals took over as the main phosphorus adsorbers, easing these constraints. By tracing how humble clays sculpted the early phosphorus cycle, the work helps explain why the rise of complex life and an oxygen-rich world was a slow, stepwise process rather than an abrupt revolution.
Citation: Cui, X., Zhang, Z., Li, Q. et al. Phyllosilicate adsorption limited phosphorus bioavailability in early ferruginous oceans. Nat Commun 17, 2422 (2026). https://doi.org/10.1038/s41467-026-69293-4
Keywords: early Earth oceans, phosphorus cycle, clay minerals, Archean biosphere, nutrient limitation