Clear Sky Science · en
Adgrg6/Gpr126 is required for compact wall integrity and establishing trabecular identity during cardiac trabeculation
Why Heart Wall Architecture Matters
Every heartbeat depends on a finely sculpted inner heart wall. In developing embryos, this wall first forms as a smooth layer and then grows a forest of muscular ridges called trabeculae that help the heart pump efficiently. When this process goes wrong, children and adults can develop serious cardiomyopathies. This study explores how a little-known receptor, Gpr126, helps heart cells decide whether to stay in the smooth outer wall or become part of these inner ridges.
A Split-Function Receptor in the Heart
Gpr126 belongs to a family of adhesion G protein–coupled receptors that sit in the cell membrane and sense the cell’s surroundings. These receptors are particularly interesting because many existing drugs already target related receptors, making them attractive therapeutic candidates. Gpr126 is unusual in that it is naturally cut into two major pieces: an N-terminal fragment (NTF), which sticks out of the cell and can aid in cell–cell adhesion, and a C-terminal fragment (CTF), which spans the membrane and triggers internal signaling. Previous work linked Gpr126 to heart development, but it was unclear what each fragment actually does during the formation of trabeculae.
Using Zebrafish to Watch the Heart Take Shape
The researchers used zebrafish, whose transparent embryos allow real-time imaging of the beating heart. They studied two genetic variants of gpr126. One (stl47) produces only a tiny, severely truncated protein; the other (st49) produces a stable, secreted NTF fragment that lacks the CTF signaling part. By combining these mutants with fluorescent reporter lines, the team could see individual heart cells, track the formation of trabeculae, and monitor key molecules such as N-cadherin (which helps cells stick together) and Notch (a signaling pathway that tells cells what identity to adopt).
Holding the Wall Together vs. Making the Ridges
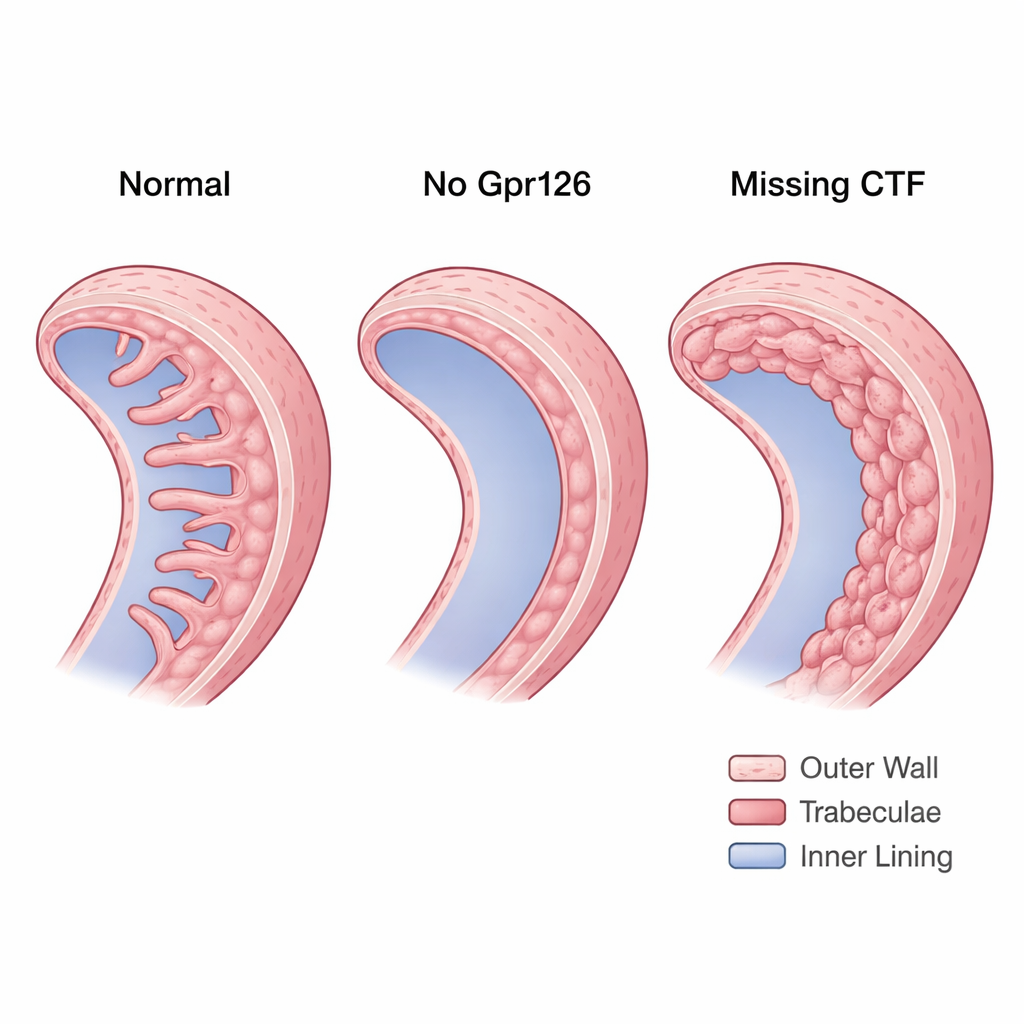
The stl47 mutants, which essentially lack both functional fragments, revealed that Gpr126 is needed to form normal trabeculae. In maternal-zygotic stl47 embryos—where no healthy Gpr126 comes from either parent—many hearts developed with missing or strongly reduced trabeculae. N-cadherin, which in healthy hearts becomes neatly concentrated at the sides of compact-wall cardiomyocytes, appeared scattered around the cell surface. This disorganization hinted that without the NTF, cell–cell adhesion in the compact wall becomes unstable, undermining the foundation on which trabeculae should build.
Too Much Wall, Not Enough Identity
The st49 mutants, which still make the NTF but lack the CTF, showed the opposite problem. Instead of missing trabeculae, their hearts formed a thick, multilayered inner wall. The extra layers were not true trabeculae: the cells remained polarized like compact-wall cells and failed to remodel their N-cadherin contacts into the punctate pattern typical of ridge-forming cells. Using a Notch activity reporter, the team found that these hearts had abnormally high Notch signaling within the myocardium. High Notch is known to keep cells in the compact wall and prevent them from delaminating into trabeculae. Importantly, when the researchers reintroduced just the CTF into endocardial cells, trabeculae reappeared and Notch activity fell back toward normal, showing that CTF signaling is crucial for giving cardiomyocytes a trabecular identity.
Balancing Signals for a Healthy Heart
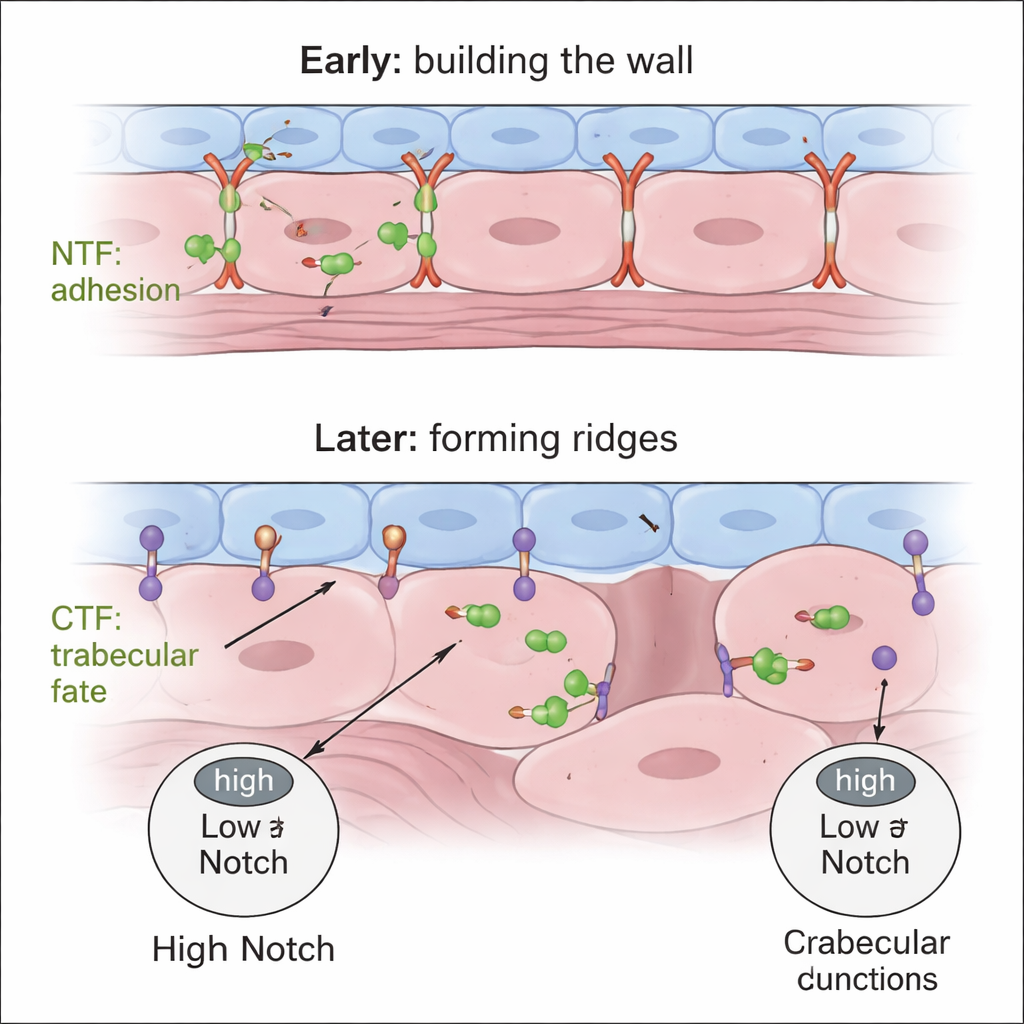
Taken together, the findings support a two-step, two-part model. First, the secreted NTF from Gpr126 helps compact-wall cardiomyocytes maintain tight, correctly positioned junctions, ensuring a sturdy single-layered wall. Later, the CTF—acting from endocardial cells—modulates myocardial Notch signaling so that only some cardiomyocytes depolarize, loosen their junctions, and protrude inward to form trabeculae, while their neighbors remain in the compact wall. If the NTF is missing, the wall never organizes properly; if the CTF is missing, the wall overgrows and cells fail to adopt trabecular identity. This domain-specific control of heart wall architecture highlights Gpr126 as a potential link between cell adhesion, signaling pathways like Notch, and structural heart disease, and suggests that tuning different parts of the same receptor could one day help correct developmental defects.
Citation: Srivastava, S., Gunawan, F., Vergarajauregui, S. et al. Adgrg6/Gpr126 is required for compact wall integrity and establishing trabecular identity during cardiac trabeculation. Nat Commun 17, 1484 (2026). https://doi.org/10.1038/s41467-026-69292-5
Keywords: cardiac trabeculation, Gpr126, heart development, Notch signaling, zebrafish model