Clear Sky Science · en
Identification of protein precursor for thyroid hormone synthesis in basal chordate ascidian Styela clava
How a Tiny Sea Creature Illuminates Our Own Hormones
Thyroid hormones help control growth, development, and metabolism in humans, but where did this system come from in evolution? This study looks to an unexpected animal—a small, tube-shaped sea creature called an ascidian (Styela clava)—to answer that question. By uncovering how this animal makes thyroid-like hormones during its transformation from a free-swimming larva to a sedentary adult, the researchers reveal that key pieces of our own endocrine system may be far older and more widespread than previously thought. 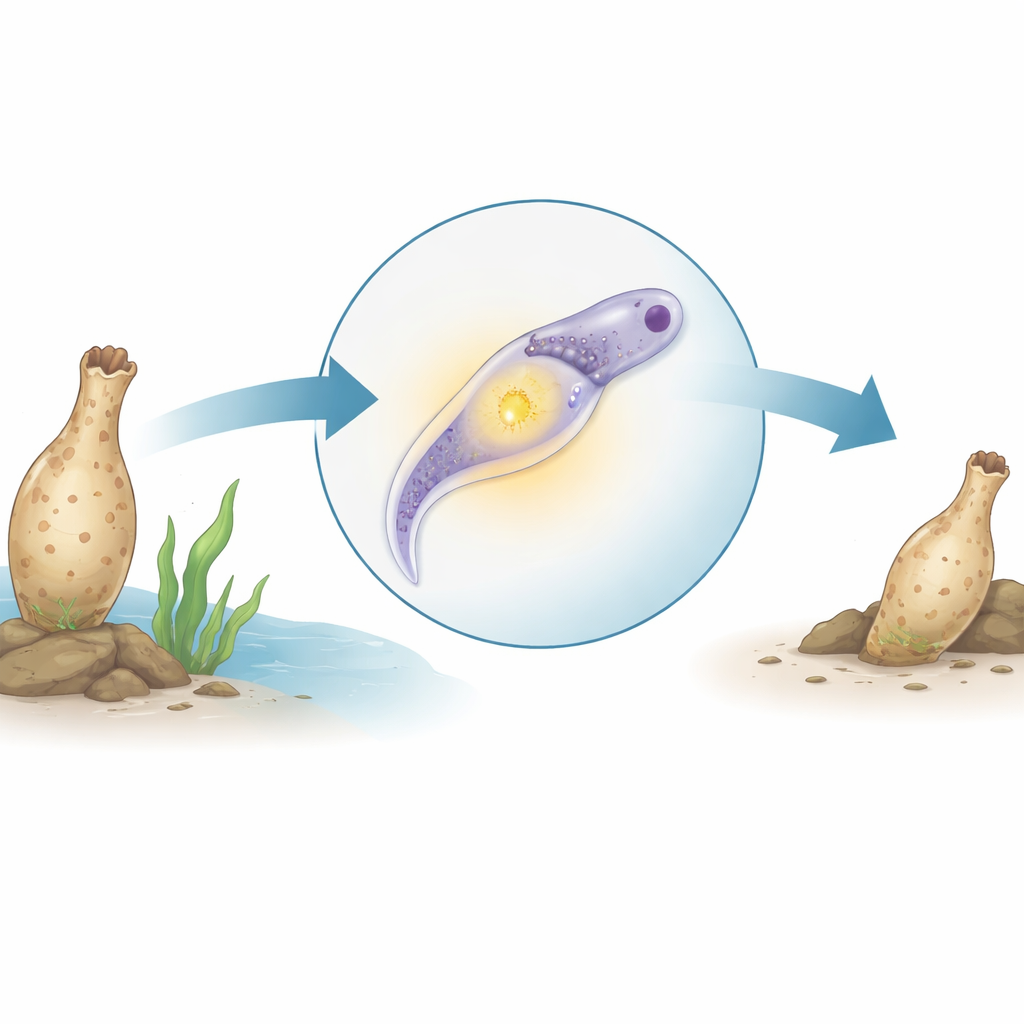
From Sea Squirts to Thyroid Science
In vertebrates, including humans, thyroid hormones are made using a huge protein called thyroglobulin inside the thyroid gland. In invertebrates, however, scientists had never clearly identified a comparable protein, leaving open the possibility that animals without backbones might rely entirely on hormone-like compounds from their environment. The authors revisited a classic but unsolved observation: antibodies against cow thyroglobulin strongly mark a region in the feeding organ (endostyle) of the ascidian Styela clava. Using modern protein analysis and evolutionary tools, they set out to discover exactly which protein was being recognized and whether it truly served as a hormone-making template.
Finding a Hidden Hormone Factory
The team first used an antibody that binds vertebrate thyroglobulin to “fish out” similar proteins from Styela clava tissue and identified several candidates by mass spectrometry. Among these, one very large protein, dubbed ScTG-like, stood out. Its gene switched on strongly during larval stages, in step with other components known to be involved in thyroid hormone production, and its messenger RNA and protein localized to the front of the swimming larva and to the thyroid-equivalent region of the adult endostyle. When the researchers produced part of this protein in cultured cells and exposed it to iodine in a test tube, it acquired chemical features characteristic of thyroid hormones, much like human thyroglobulin does. A related Styela protein failed this test, sharpening the case that ScTG-like is the true hormone precursor.
A Proto-Thyroid Gland in a Tadpole Body
Looking more closely inside the larvae, the scientists discovered a small invaginated pocket in the anterior trunk made of a handful of closely packed cells. This “follicle-like” structure glowed with signals for ScTG-like, thyroid hormones themselves, and several other proteins that in vertebrates help build and operate the thyroid gland. Stains that highlight sugar-rich secreted proteins, which are abundant in vertebrate thyroid follicles, also lit up this pocket, and similar staining was seen in zebrafish thyroid tissue. When ScTG-like was experimentally knocked down, both the protein and thyroid hormone signals vanished from this tiny pocket, indicating that it serves as a genuine hormone synthesis and storage site—a kind of proto-thyroid gland in an invertebrate larva. 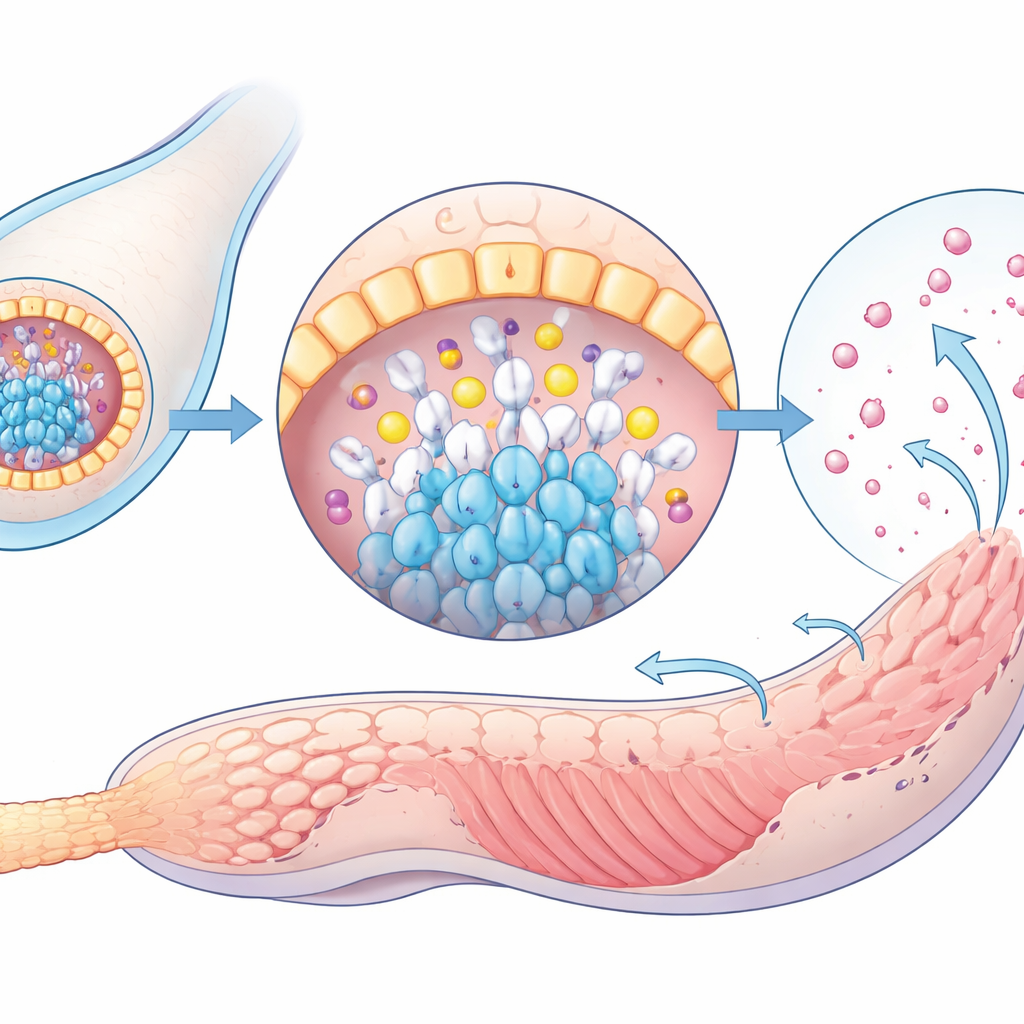
Hormones That Drive a Dramatic Transformation
To test whether ScTG-like–dependent hormones actually matter for the animal’s life cycle, the researchers reduced ScTG-like production using RNA interference. Larvae with lowered ScTG-like showed sharply reduced levels of thyroid hormones and struggled to undergo normal metamorphosis: their tails regressed poorly, their transformation was delayed, and the resulting juveniles had malformed siphons and immature internal organs. Treating these impaired larvae with a dose of active thyroid hormone (T3) rescued their development, restoring metamorphosis to near-normal levels. This direct rescue links the protein’s role in hormone synthesis to its control over the timing and quality of the animal’s dramatic shape change.
An Ancient Blueprint for Thyroid Hormone Making
Beyond this single species, the team compared protein architectures across many animal groups and found TG-like proteins with similar structural hallmarks—especially repeated domains thought to carry hormone-forming sites—in other bilaterian animals, from echinoderms to worms and mollusks. Even when amino-acid sequences differed greatly, the overall layout of these domains and the presence of many possible disulfide bonds resembled those in vertebrate thyroglobulin. These parallels suggest that internally produced thyroid-like hormones, made on large scaffold proteins, originated deep in animal evolution and were later refined into the well-organized thyroid glands of vertebrates.
Why This Matters for Understanding Ourselves
This work identifies, for the first time, a functional protein precursor for thyroid hormone synthesis outside vertebrates and pinpoints a follicle-like structure that acts as a primitive thyroid in an invertebrate chordate. For non-specialists, the takeaway is that the machinery our own bodies use to control growth and metabolism is not a late invention unique to vertebrates, but rather an ancient system already taking shape in simple sea animals. By tracing these hormonal roots back to creatures like Styela clava, scientists gain a clearer picture of how complex endocrine organs evolved and how conserved hormone pathways help sculpt the life cycles of animals across the tree of life.
Citation: Zhang, J., Yang, L., Beinsteiner, B. et al. Identification of protein precursor for thyroid hormone synthesis in basal chordate ascidian Styela clava. Nat Commun 17, 2463 (2026). https://doi.org/10.1038/s41467-026-69290-7
Keywords: thyroid hormone evolution, sea squirt metamorphosis, thyroglobulin-like protein, endocrine system origins, invertebrate hormone synthesis