Clear Sky Science · en
The genetic driver of Acute Necrotizing Encephalopathy, RANBP2, regulates the inflammatory response to Influenza A virus infection
When the Flu Harms the Brain
Most people think of influenza as a week of fever, cough, and bed rest. Yet in rare cases, especially in children, a routine flu infection can suddenly spiral into a life‑threatening brain disorder called acute necrotizing encephalopathy (ANE). Families and doctors have long suspected that genes may help determine who develops this catastrophic complication. This study uncovers how a single cellular gatekeeper protein, RANBP2, normally keeps the flu virus in check and prevents the body’s own inflammatory response from running out of control.
A Rare but Devastating Flu Complication
ANE appears abruptly after a feverish illness, often influenza, and can rapidly cause seizures, coma, and lasting neurological damage. Roughly half of known ANE episodes worldwide are linked to influenza A virus, particularly the H1N1 strain. Children who inherit certain changes in the RANBP2 gene face a much higher risk, in a condition known as ANE1. Until now, however, scientists did not understand what this protein actually does during flu infection, or why its alteration would make inflammation in the brain so severe.
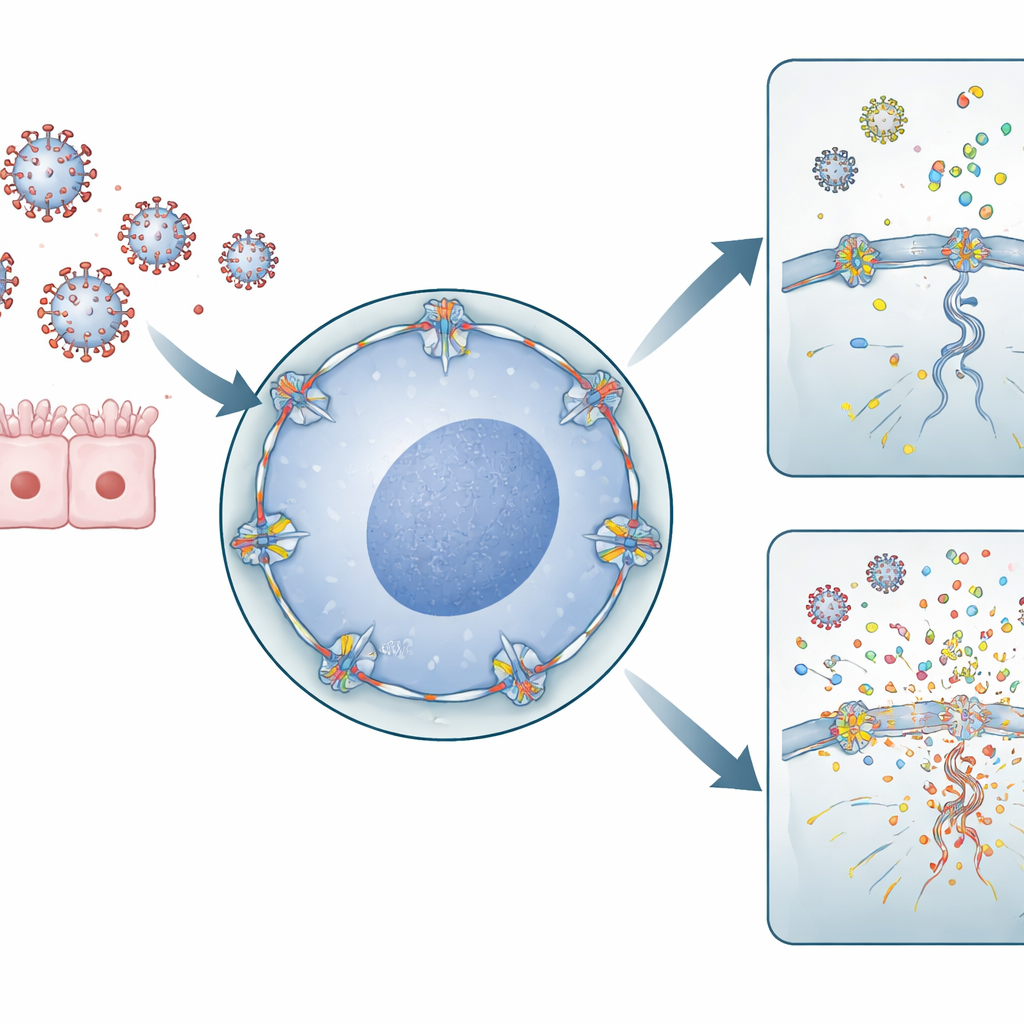
Cellular Gates and Viral Genomes
Influenza A virus has an unusual lifestyle for an RNA virus: it must enter the cell’s nucleus to copy its genetic material. To do so, it passes through nuclear pores—large gateways in the nuclear envelope that control traffic between the nucleus and the surrounding cytoplasm. RANBP2 is a major component on the outer side of these pores. The authors used human lung‑derived cells and immune cells to lower RANBP2 levels or introduce the ANE‑linked mutation. They found that when RANBP2 was missing or misplaced, flu genomes were copied more extensively inside the nucleus, and viral genetic segments were exported to the cytoplasm in an unbalanced way. Surprisingly, this surplus of viral RNA did not yield more infectious virus particles, but it did change where and how viral material accumulated inside the cell.
When Viral Debris Feeds the Alarm System
The immune system relies on molecular sensors that detect stray bits of viral RNA in the cytoplasm as danger signals. The team showed that, in cells lacking normal RANBP2, extra viral RNA segments built up in the cytoplasm, creating exactly the kind of patterns these sensors are tuned to recognize. In lung‑derived cells, this triggered higher levels of inflammatory molecules such as IL‑6 and IL‑1β. In primary human macrophages—front‑line immune cells from blood donors—RANBP2 loss led to a striking surge in pro‑inflammatory chemokines, including CXCL8, CXCL10, CCL2, CCL3, and CCL4. These chemical messengers recruit and activate additional immune cells, amplifying the overall reaction to infection.
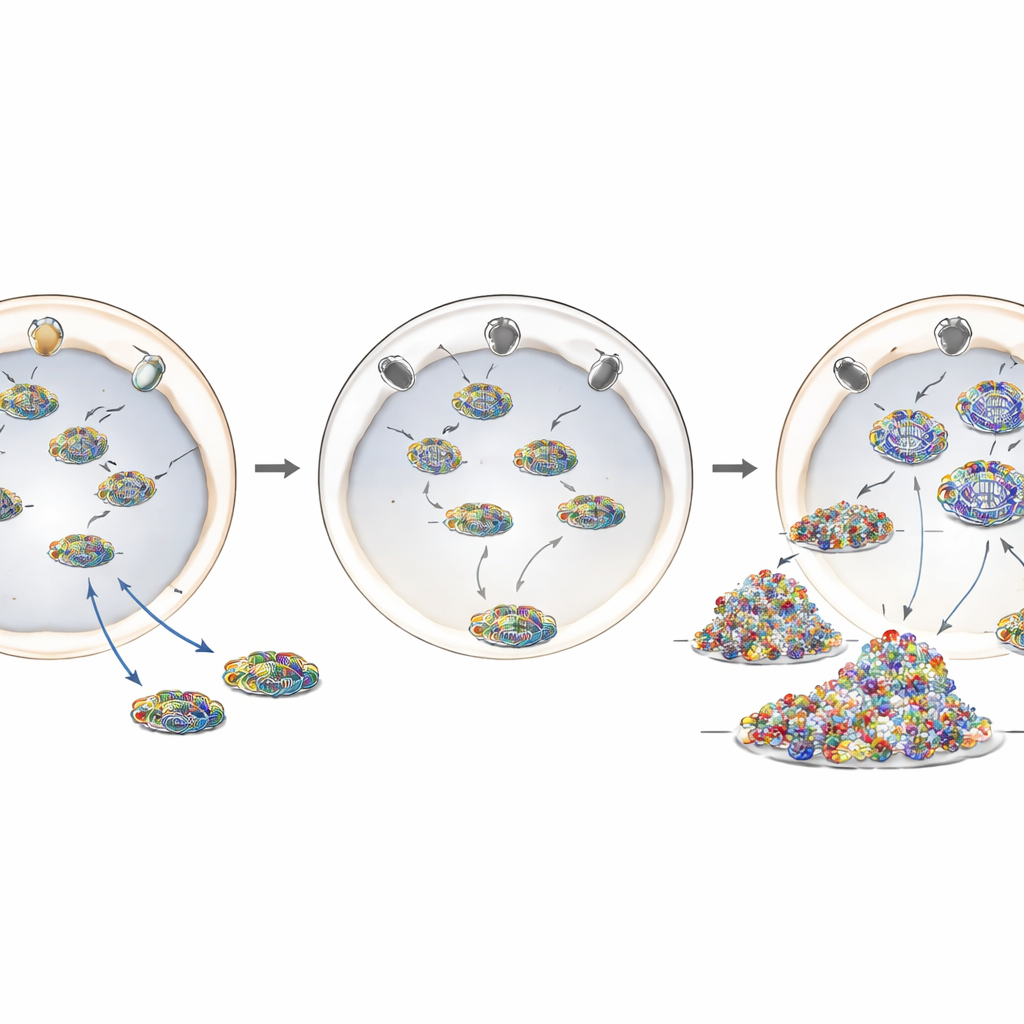
A Disease Mutation that Misplaces a Guardian
To model ANE1 more closely, the researchers used CRISPR‑Cas9 genome editing to introduce the most common disease‑associated change, T585M, into the human RANBP2 gene. In these edited cells, RANBP2 protein levels were largely normal, but its location was not: instead of forming a bright rim around the nuclear envelope, much of the protein was displaced into the cell interior. Cells carrying this mutation, whether in one or both gene copies, behaved much like RANBP2‑depleted cells. They supported increased influenza genome replication, showed more viral material in the cytoplasm, and mounted a stronger inflammatory response after infection. This suggests that correctly positioning RANBP2 at nuclear pores—rather than its overall abundance—is crucial to its protective role.
Why This Matters for Children at Risk
Taken together, the findings paint RANBP2 as a cellular guardian that fine‑tunes how influenza genetic material moves into and out of the nucleus. When RANBP2 is absent or mislocalized, viral RNA is over‑replicated and exported in a chaotic fashion, cluttering the cytoplasm with molecular debris that greatly intensifies immune signaling. For most tissues, this may mean a harsher but survivable bout of flu. In vulnerable children with ANE1, however, such runaway inflammation—especially if it reaches the brain—may help explain the sudden, severe neurological damage seen after otherwise ordinary infections. Understanding this pathway could guide future strategies to identify at‑risk patients early and to tailor anti‑inflammatory treatments before the immune response turns from protective to destructive.
Citation: Desgraupes, S., Decorsière, A., Perrin, S. et al. The genetic driver of Acute Necrotizing Encephalopathy, RANBP2, regulates the inflammatory response to Influenza A virus infection. Nat Commun 17, 2427 (2026). https://doi.org/10.1038/s41467-026-69288-1
Keywords: influenza, acute necrotizing encephalopathy, RANBP2, hyperinflammation, nuclear pore