Clear Sky Science · en
Electrostatic properties of disordered regions control transcription factor search and pioneer activity
How protein “tails” help genes switch on
Every cell in your body must rapidly decide which genes to turn on, despite its DNA being tightly packed into chromatin. This paper explores how floppy, electrically charged extensions of key gene‑control proteins act like built‑in search helpers, allowing some proteins to find buried DNA switches and pry open the surrounding material, while others struggle to do so. Understanding this hidden layer of control sheds light on how stem cells stay flexible and how cells change identity.
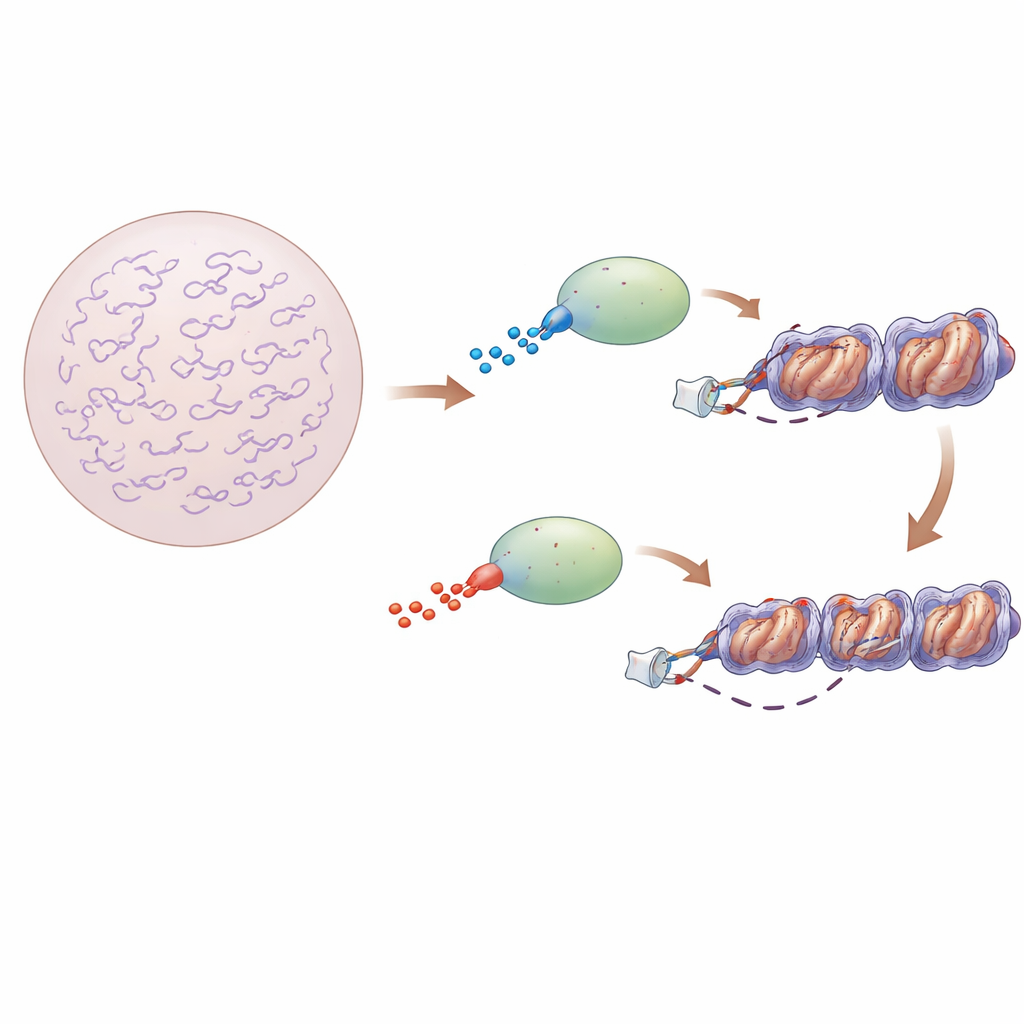
Gene switches in a crowded DNA landscape
Transcription factors are proteins that locate short DNA sequences and trigger nearby genes. In bacteria, the DNA is relatively exposed, and classic models describe these proteins as repeatedly jumping into solution and then sliding along bare DNA until they hit their targets. In animal cells, however, DNA is wrapped around spool‑like proteins to form nucleosomes and further folded into compact chromatin. This crowding makes it unclear how transcription factors still manage to find the right spots quickly enough to control thousands of genes.
Floppy protein regions with hidden influence
Many transcription factors contain structured DNA‑gripping cores flanked by long, disordered stretches of amino acids. These floppy regions do not fold into fixed shapes, but they do carry electrical charges. The authors focused on two closely related factors, Sox2 and Sox17, which share almost the same DNA‑gripping core yet behave very differently in cells. Sox2 is a classic “pioneer” factor that can bind to DNA hidden in compact chromatin and is essential for keeping stem cells in a flexible, pluripotent state. Sox17, in contrast, normally acts later during development and is much less able to bind to tightly packed DNA. The key difference: the region just after the DNA‑binding core of Sox2 is more positively charged, whereas the equivalent region in Sox17 is more negatively charged.
Watching individual molecules search DNA
To see how these charge differences affect the search process, the researchers used single‑molecule microscopy both in living mouse stem cells and with purified components on glass surfaces. They engineered versions of Sox2 and Sox17 that swap these charged “tails,” and also versions containing only the DNA‑gripping core. In cells, they tracked fluorescently tagged proteins one by one, measuring how fast they diffuse, how long they stay bound, and how often they land on DNA. Proteins carrying the positively charged Sox2 tail bound chromatin more frequently and spent more time in long‑lived interactions than those with the Sox17 tail, even though all versions recognized essentially the same DNA letters.
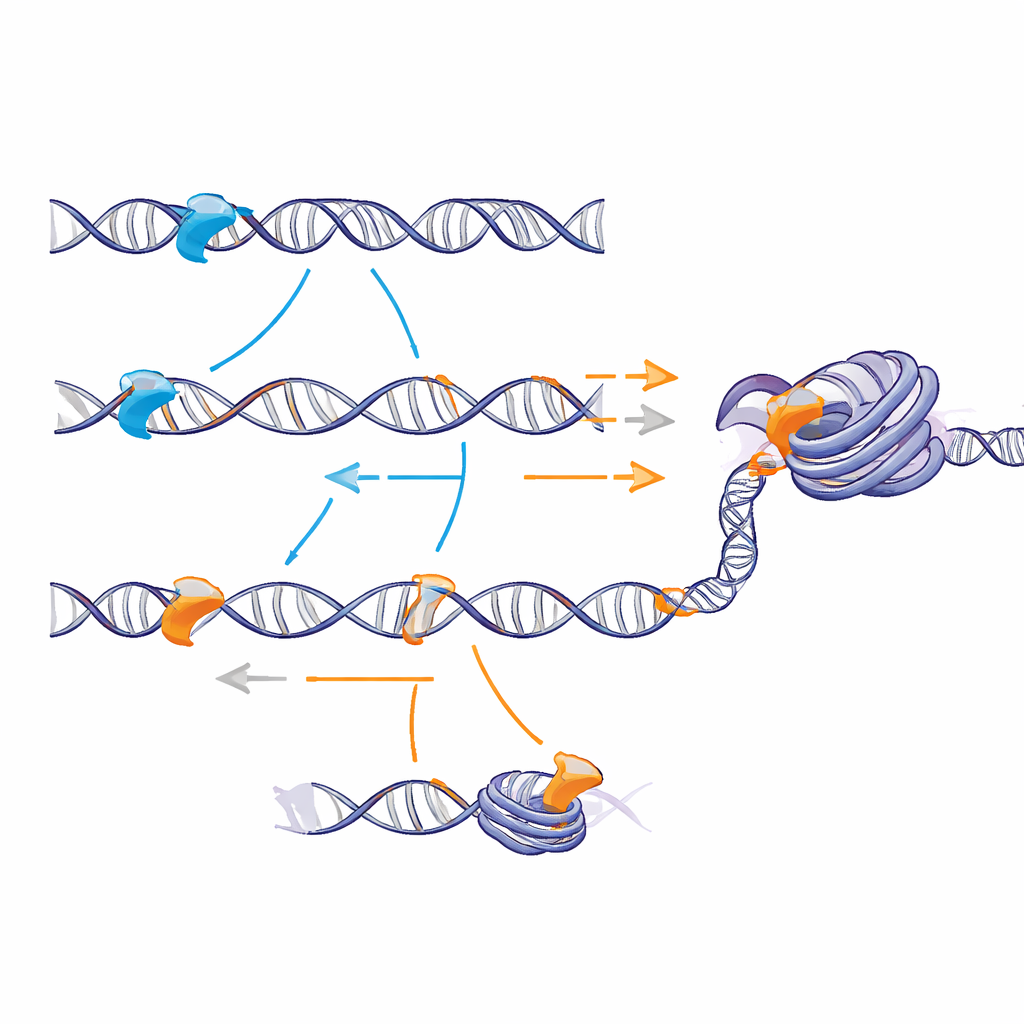
Slow sliding but better recognition
In carefully controlled test‑tube experiments with pieces of bare DNA, the charged tails did not change how often proteins initially collided with DNA from solution. Instead, they altered what happened once a protein had landed. By combining experiments with mathematical modeling, the authors showed that the Sox2 tail makes the protein slide more slowly along DNA but more likely to “notice” its specific target when it passes over it. The Sox17 tail allows faster motion but increases the chance of simply gliding past the right site without locking on. This reveals a trade‑off between speed and recognition: a stickier, positively charged tail roughens the energy landscape enough to boost the odds of successful target capture.
Invading and opening compact chromatin
When the team rebuilt nucleosomes and short chromatin fibers in vitro, the contrast became sharper. The Sox2 tail promoted frequent, short‑lived contacts with both the wrapped DNA and the histone spools, which occasionally converted into longer, specific binding at buried target sites. On model chromatin fibers, this led to more stable binding and greater access to internal sites than seen with the Sox17 tail. In stem cells, artificially expressed Sox2 increased binding in naturally closed chromatin regions and made these regions more accessible, as measured by an assay that detects how easily enzymes can cut the DNA. The version of Sox2 carrying the Sox17 tail bound less well and opened chromatin less efficiently, even though it still recognized the same DNA motifs.
What this means for cell identity
Overall, the study shows that the electrical charge of disordered protein “tails” can tune how transcription factors search DNA and how effectively they can invade and loosen compact chromatin. A more positively charged tail, as in Sox2, promotes frequent nonspecific contacts and sharpens target recognition, supporting strong pioneer activity and helping maintain the open chromatin landscape of stem cells. These principles likely extend to many other gene‑control proteins, adding a new design rule for how cells program, and reprogram, their genetic activity.
Citation: Sakong, S., Fierz, B. & Suter, D.M. Electrostatic properties of disordered regions control transcription factor search and pioneer activity. Nat Commun 17, 2512 (2026). https://doi.org/10.1038/s41467-026-69284-5
Keywords: transcription factors, chromatin, Sox2, intrinsically disordered regions, pioneer activity