Clear Sky Science · en
Urea photosynthesis over a MOF-on-MOF S-scheme heterojunction
Turning Waste into Fertilizer with Sunlight
Modern farming depends on urea fertilizer, but making it the conventional way burns large amounts of fossil fuel and releases extra carbon dioxide. At the same time, waterways around the world are polluted with nitrate, and the air holds rising levels of carbon dioxide. This study explores a way to turn these two wastes into useful urea using only sunlight, offering a glimpse of cleaner chemistry that could help both food production and the environment.
Why Rethinking Urea Matters
Today’s urea is mostly made by reacting ammonia with carbon dioxide at high temperatures and pressures inside large industrial plants. That process is energy-hungry and tied tightly to fossil fuels. Scientists have long imagined a gentler route: using sunlight to power reactions that stitch together carbon and nitrogen directly from simple molecules like nitrogen gas and carbon dioxide dissolved in water. However, nitrogen gas is stubbornly unreactive and poorly soluble, so early “solar urea” experiments delivered very little product. The authors of this paper take a different tack by replacing hard-to-activate nitrogen gas with nitrate, a much more reactive nitrogen source that is already abundant as a pollutant in many wastewaters.
Building a Layered Light-Driven Sponge
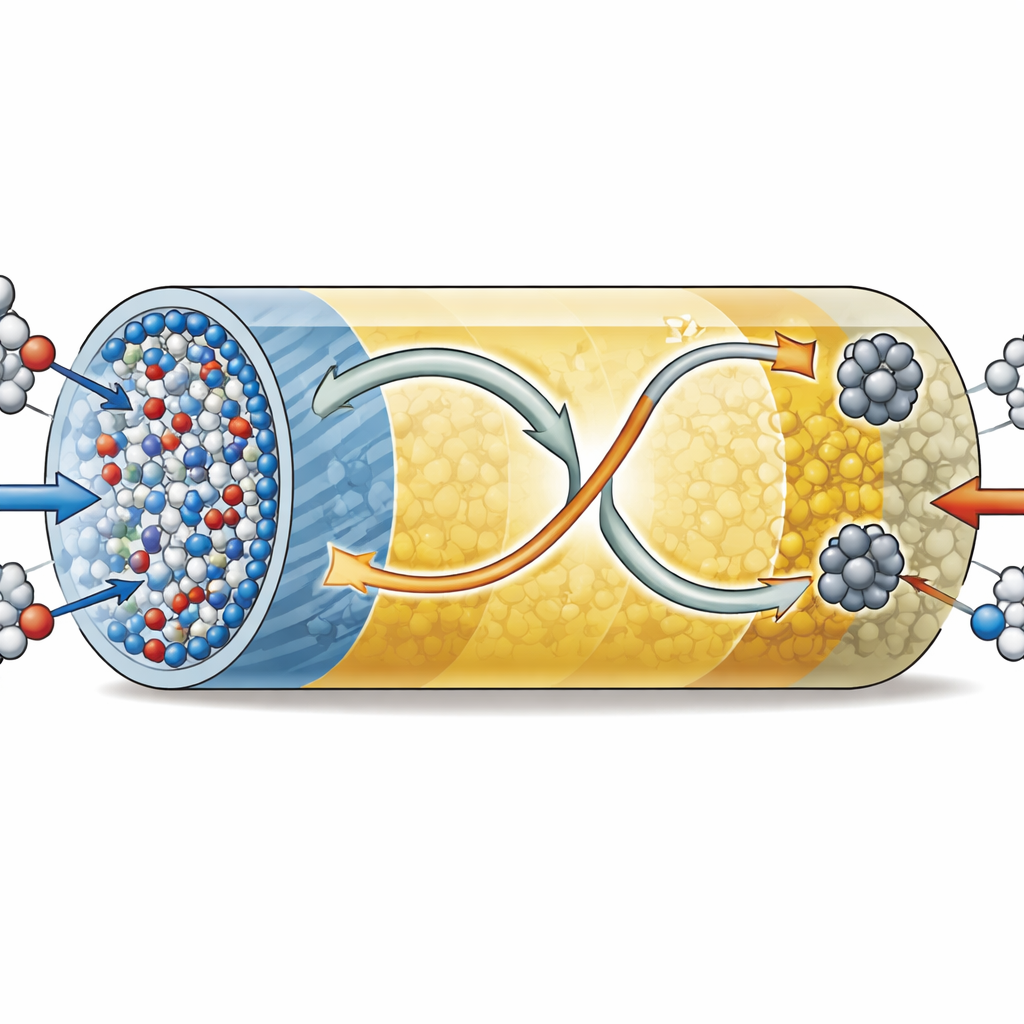
To make this solar-driven chemistry work, the team designed a tiny, porous structure called a metal–organic framework, or MOF, in which metal atoms are linked by organic molecules to form an ordered sponge. They did not rely on just one MOF, but instead grew a second MOF as a thin shell on the first, creating a “MOF-on-MOF” rod with a solid zirconium-based core (called NU-1000) and a cobalt-based shell (called Co-HHTP). High-resolution electron microscopy and elemental mapping confirm that the inner rods and outer nanorods form a well-defined core–shell architecture, with zirconium concentrated in the center and cobalt on the outside. This layered structure provides a huge internal surface area and, crucially, brings zirconium and cobalt atoms into close contact at the interface, where the important chemistry happens.
Guiding Light and Charges in the Right Direction
Sunlight excites electrons in a photocatalyst, but these charges must be separated and steered efficiently to drive useful reactions instead of simply recombining as heat. Optical and electrochemical tests show that the combined MOF-on-MOF structure absorbs a broader range of light than either component alone and behaves as an “S-scheme” junction. In essence, when the two MOFs touch, electrons naturally flow from cobalt sites to zirconium sites until their energy levels line up, creating an internal electric field. Under illumination, this built-in field and the bent energy bands push electrons and holes in opposite directions within the rod, keeping the most energetic electrons on cobalt sites and the strongest oxidizing holes on zirconium sites. Measurements of photocurrent, fluorescence, and charge lifetimes all indicate that this arrangement greatly improves charge separation and transport compared with the single-layer materials or a simple physical mixture.
Making and Tracing Solar-Made Urea
When the MOF-on-MOF rods are suspended in water containing dissolved nitrate and saturated with carbon dioxide, and then exposed to simulated sunlight, they produce urea much more rapidly than either of the individual MOFs. The authors report a urea production rate over three thousand micrograms per gram of catalyst per hour and a measurable quantum yield at ultraviolet wavelengths, both competitive with the best photocatalysts reported so far. By using specially labeled nitrate and carbon dioxide, they confirm that both atoms in the urea product truly come from these two sources. Real-time infrared monitoring reveals key reaction intermediates: nitrate is first reduced on cobalt sites to nitrogen–oxygen fragments, while carbon dioxide is held on zirconium sites. These fragments then couple at the Co–Zr interface to build the carbon–nitrogen bonds that define urea, with only modest amounts of side products such as ammonia, carbon monoxide, and hydrogen.
How the Dual-Site Design Does the Heavy Lifting
The researchers use computer simulations to dig deeper into why the interface is so effective. Calculations show that nitrate binds particularly strongly to cobalt atoms, while carbon dioxide prefers zirconium atoms, and both molecules are more strongly adsorbed in the combined structure than on either MOF alone. The crucial step—joining a nitrogen-containing fragment from nitrate with a carbon-containing fragment from carbon dioxide—faces a noticeably lower energy barrier on the dual-site interface than on cobalt alone. This means that once the molecules are in place on the catalyst surface, they can link up to form urea more easily and with less wasted energy.
A Step Toward Cleaner Fertilizer Production
In everyday terms, this work shows that it is possible to design tiny, layered sponges that use sunlight to pull harmful nitrate from water and carbon dioxide from air, then knit them together into a valuable fertilizer ingredient. While the technology is still far from replacing today’s massive urea factories, the MOF-on-MOF “S-scheme” design offers a blueprint for future photocatalysts: combine different active metals at well-controlled interfaces, guide light-driven charges intelligently, and turn pollution into useful products under mild conditions.
Citation: Xi, Y., Zhang, C., Bao, T. et al. Urea photosynthesis over a MOF-on-MOF S-scheme heterojunction. Nat Commun 17, 2423 (2026). https://doi.org/10.1038/s41467-026-69281-8
Keywords: solar urea synthesis, photocatalyst, metal–organic framework, nitrate reduction, carbon dioxide utilization