Clear Sky Science · en
MHC class II functions as a host-specific entry receptor for representative human and swine H3N2 influenza A viruses
Why this flu study matters
Seasonal flu seems familiar, but the virus that causes it is constantly searching for new ways to infect our cells and jump between species. This study reveals that a common flu virus strain, H3N2, can use not just its classic doorway into cells but also a second, unexpected one that differs between humans and pigs. Understanding this backup entry route helps explain how flu adapts to new hosts and could improve how we watch for, and prepare against, strains with pandemic potential. 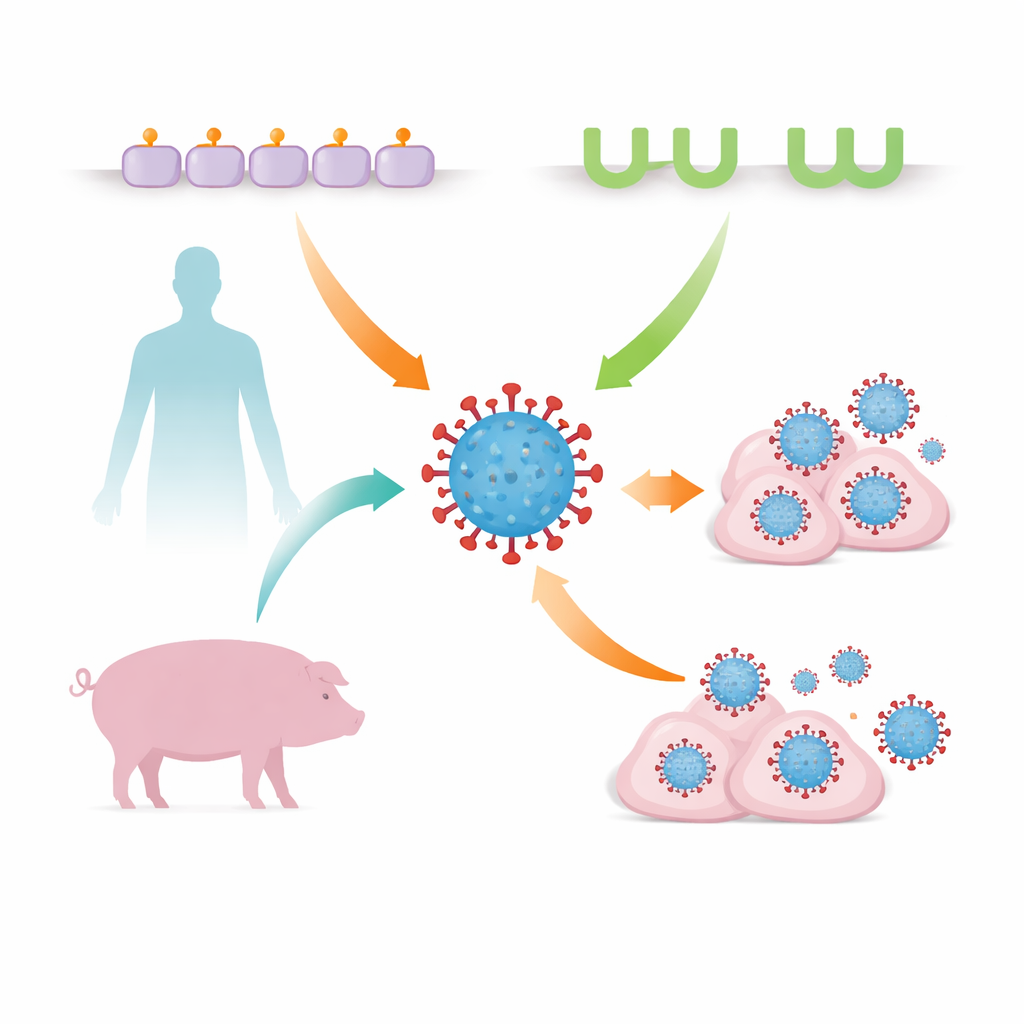
The usual way flu gets in
For decades, scientists have known that influenza A viruses typically enter cells by latching onto sugar molecules called sialic acids that decorate the cell surface. The viral "grappling hook," a protein called hemagglutinin, recognizes specific shapes and linkages of these sugars, which differ between birds, humans, and other animals. Small changes in hemagglutinin can shift its sugar preference, helping a bird virus adapt to mammals. Recently, however, unusual bat and duck flu viruses were found that ignore sialic acids entirely and instead use a protein called MHC class II as their doorway, hinting that our picture of flu entry was incomplete.
A second doorway appears
The new work shows that more familiar H3N2 viruses can also use MHC class II as an alternative entry route, in addition to sialic acids. MHC class II normally helps immune cells display pieces of germs to alert the immune system. The team focused on two closely related viruses built on the same genetic backbone: one resembling a human seasonal H3N2 strain (hVIC/11) and another adapted to pigs (sOH/04). In pig lungs, the swine-adapted virus was found closely associated with cells rich in pig MHC class II, especially alveolar macrophages—front-line immune cells in the air sacs of the lung—suggesting that this molecule might be acting as a handle the virus can grab.
Disabling one door to test the other
To test whether MHC class II truly acts as an independent entry receptor, the researchers chemically stripped sialic acids from pig macrophages and from engineered human cell lines. When only the classic sugars were removed, both viruses still managed some infection. But when sialic acids were removed and MHC class II was simultaneously blocked with antibodies, infection dropped sharply, especially for the swine-adapted virus. In human kidney and lung cells that were genetically prevented from making any sialic acid at all, adding the human version of MHC class II enabled efficient infection by the human-like virus, while adding the pig version favored the swine virus. Treating cells with drugs that stop the acidification of internal compartments also blocked this MHC-driven infection, indicating that once inside, the virus follows the same internal route as in standard flu entry. 
How small changes help the virus switch hosts
The authors then asked what parts of the viral hemagglutinin control this new type of binding. Earlier work had shown that when a human H3N2 virus passes through pigs, it often picks up certain single–letter changes near, but not directly inside, the sialic acid binding pocket. Viruses carrying any one of three such changes (at positions labeled 138, 186, or 193 in the protein) were now tested. These mutant viruses could use both human and pig MHC class II to enter cells that lacked sialic acid, and in some assays they even preferred the pig version. At the same time, these same mutations altered how strongly the viruses bound to human- or bird-like sialic acids. In other words, tweaks near the usual sugar-binding spot can tune both the classic and the new receptor at once, giving the virus flexibility as it adapts to a new species.
What this means for flu and future threats
Taken together, the findings show that common H3N2 flu viruses are not limited to a single type of cell-surface handle: they can use both sialic acids and MHC class II, and they do so in a way that reflects whether the virus is better suited to humans or pigs. During early adaptation to a new host, a virus can temporarily engage MHC class II from both species, widening its target range and helping it get established. Because MHC class II is abundant on key immune cells in the lung, this alternative route may allow flu to selectively attack cells that are supposed to defend us, potentially aiding viral spread and disease. Recognizing MHC class II as a host-specific entry receptor adds an important piece to the puzzle of how flu crosses species barriers and underscores the need to monitor not just sugar binding but also protein receptors when assessing the pandemic risk of emerging strains.
Citation: Cardenas, M., Compton, S., Caceres, C.J. et al. MHC class II functions as a host-specific entry receptor for representative human and swine H3N2 influenza A viruses. Nat Commun 17, 2560 (2026). https://doi.org/10.1038/s41467-026-69267-6
Keywords: influenza A H3N2, virus entry receptors, MHC class II, host adaptation, zoonotic transmission