Clear Sky Science · en
Heterogeneous multicopy of blaCTX-M variants on the same plasmid enhances evolutionary adaptability in clinical Klebsiella pneumoniae
Why this matters for modern medicine
Antibiotic-resistant infections are a growing threat in hospitals worldwide, and doctors are increasingly relying on last-resort drugs to save patients. This study explains how a common hospital bacterium, Klebsiella pneumoniae, can use a subtle genetic trick to withstand powerful antibiotic combinations that were designed to overcome resistance. By uncovering this strategy, the research helps explain why some infections keep coming back despite aggressive treatment—and offers clues for how doctors might stay one step ahead.
A hospital germ under pressure
The story begins in an intensive care unit, where two patients were infected by nearly identical strains of K. pneumoniae. One strain was easily treated with the modern drug pair ceftazidime/avibactam, while the other was strongly resistant. Genetic comparisons showed that both strains carried the same broad family of resistance enzymes, called β-lactamases, on a shared plasmid—a small, mobile DNA circle inside the bacteria. But in the resistant strain, one of these enzymes had subtly changed, and that variant, named CTX-M-249, allowed the bacterium to shrug off the drug combination that should have killed it.
A small change with big consequences
Closer biochemical tests revealed that CTX-M-249 trades one type of protection for another. The usual version, CTX-M-65, is excellent at breaking down certain antibiotics like cefotaxime, but remains vulnerable to the inhibitor avibactam. CTX-M-249, altered at just two positions in the protein, becomes good at handling ceftazidime plus avibactam but loses much of its strength against cefotaxime. On paper, this looks like a classic evolutionary trade-off: gaining one defense while weakening another. Yet the clinical strain avoided this drawback by carrying several closely related copies of the gene at once, so different versions of the enzyme could coexist in the same bacterial lineage.
Many copies, many options
Using long-read DNA sequencing and precise counting methods, the researchers found that the plasmid in the resistant strain carried two separate blaCTX-M sites, and one of them could exist in multiple, slightly different versions. Under no drug pressure, about half the population carried the older CTX-M-65 version and nearly half carried CTX-M-249, with a small fraction bearing intermediate forms. When the bacteria were exposed to increasing doses of ceftazidime/avibactam, both the number of gene copies and the share of CTX-M-249 rose sharply. Sometimes this happened by increasing the number of plasmids per cell; at higher drug levels, the plasmid itself sprouted short tandem repeats of the resistance gene. In effect, the bacteria used DNA duplication as a dial they could turn up or down to match the surrounding antibiotics.
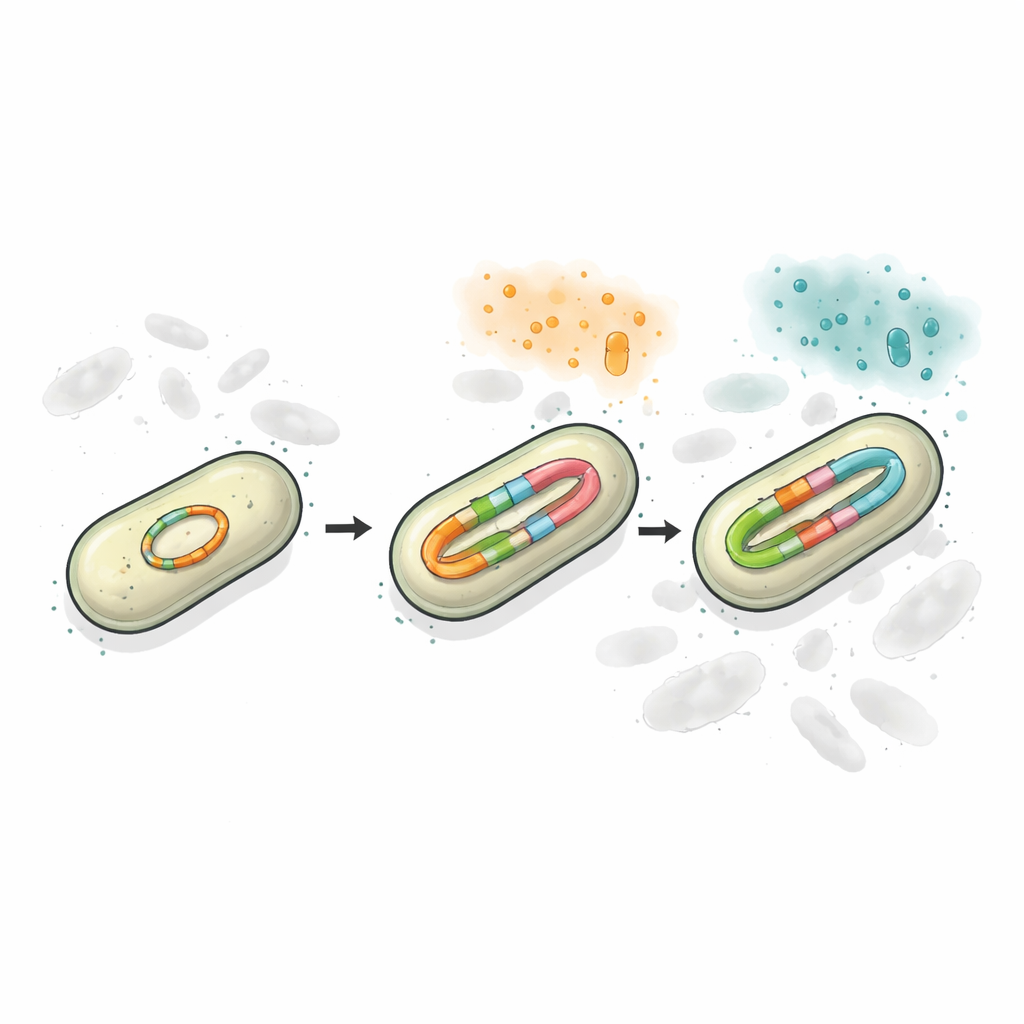
Keeping diversity on one DNA circle
To test how this arrangement shapes survival, the team built simplified laboratory models in which bacteria carried either one resistance version, two versions on separate plasmids, or both versions encoded together on a single plasmid. When challenged with two different cephalosporin drugs, the mixed systems outperformed single-gene strains, because at least one version of the enzyme could cope with each drug. However, the configuration where both gene variants sat on the same plasmid proved the most stable. When antibiotics were applied for several days or switched from one drug to another, cells with two separate plasmids often lost one of them, sacrificing part of their protection. In contrast, the “two-in-one” plasmid was inherited as a package, preserving both resistance options even when it imposed a short-term growth cost.
A wider pattern in dangerous bacteria
Mathematical models reproduced these experiments, showing that, above certain antibiotic levels, bacteria with a single plasmid carrying multiple resistance variants ultimately dominate mixed populations. The researchers then searched thousands of K. pneumoniae genomes from hospitals, farms, food, and the environment. They frequently found multiple, slightly different copies of key resistance genes—especially in human clinical isolates that face heavy drug exposure. This suggests that building “multicopy heterogeneity” into plasmids is not a rare oddity, but a widespread tactic that bacteria use to hedge their bets against changing treatments.
What this means for patients and treatment
To a non-specialist, the core message is that some bacteria do not simply carry a single resistance gene; they carry families of related versions bundled onto the same mobile DNA element, giving them a flexible toolkit against different drugs. This arrangement allows them to maintain resistance over long periods, even as doctors change therapies, and helps explain why certain infections are so hard to clear. At the same time, the study shows that carefully chosen drug combinations—such as pairing ceftazidime/avibactam with cefotaxime—can exploit weaknesses in this system and suppress even these well-armed strains. Understanding how bacteria engineer and use these multicopy plasmids is therefore crucial for designing smarter antibiotic strategies and slowing the march of resistance.
Citation: Weng, R., Zhu, J., Wu, X. et al. Heterogeneous multicopy of blaCTX-M variants on the same plasmid enhances evolutionary adaptability in clinical Klebsiella pneumoniae. Nat Commun 17, 2460 (2026). https://doi.org/10.1038/s41467-026-69266-7
Keywords: antibiotic resistance, Klebsiella pneumoniae, plasmids, beta-lactamases, multidrug therapy