Clear Sky Science · en
Trifunctional flavoenzyme-catalyzed asymmetric 4-alkyl-butenolide assembly in avenolide biosynthesis
Why tiny rings in bacteria matter to us
Many life‑saving medicines, from parasite killers to crop protectants, depend on a small chemical motif called a butenolide. Today these rings are mostly made from oil‑derived starting materials in multi‑step industrial processes that consume energy and create waste. This study reveals how soil bacteria build one such ring, a hormone called avenolide that switches on production of the blockbuster antiparasitic drugs avermectins. Understanding this natural route points toward cleaner, cheaper ways to make useful chemicals and could help unlock new antibiotics.
The special ring at the heart of many drugs
Butenolides are compact, five‑membered rings that chemists love because they react in versatile ways and appear in many natural products and modern drugs. They help shape molecules with anticancer, antifungal, anti‑inflammatory, and insecticidal activities used in medicine and agriculture. Yet traditional synthetic routes to these rings usually require several carefully controlled steps, expensive catalysts, and petrochemical feedstocks. That combination drives up cost and environmental impact, pushing researchers to look for biological shortcuts that nature has already perfected.
A bacterial hormone that turns on a blockbuster medicine
In the soil bacterium Streptomyces avermitilis, the butenolide‑containing molecule avenolide acts as a tiny hormone. At extremely low concentrations it binds to a regulatory protein and lifts a molecular brake on the genes that make avermectins, powerful agents that paralyze parasitic worms and some insects. Earlier genetic work had hinted that two enzymes, named SavA and SavB, build avenolide, but the actual steps were unknown. The team first moved the relevant genes into a more cooperative relative, Streptomyces albidoflavus, and optimized growth conditions until the engineered strain produced milligram quantities of pure avenolide—enough to dissect the pathway in detail. 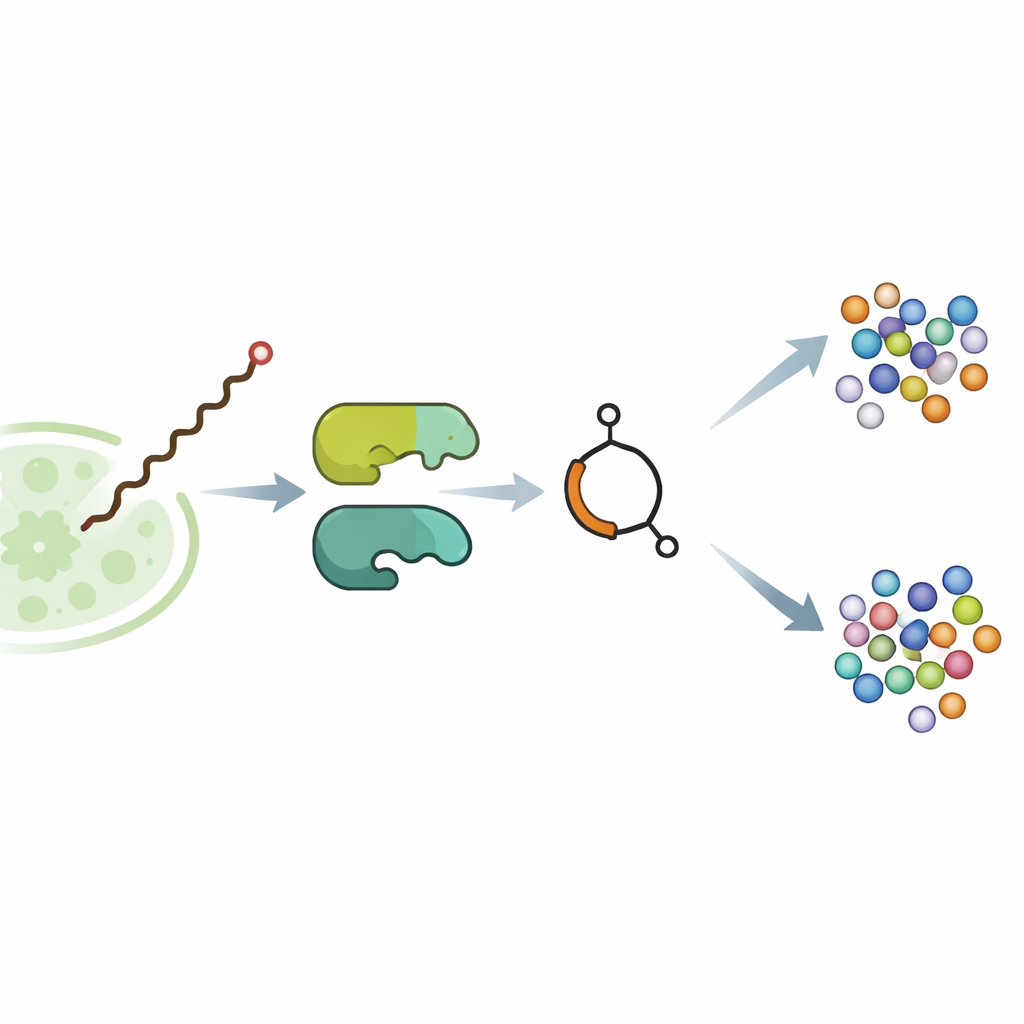
A single enzyme that does three jobs in a row
The core surprise of the study is SavA, a flavin‑containing enzyme that performs three distinct chemical transformations on a fatty‑acid‑like starter piece taken from normal cell metabolism. Working in test‑tube reactions with carefully synthesized mimic substrates, the researchers showed that SavA first removes hydrogen atoms to introduce a double bond, then adds an oxygen‑containing group at a specific position, and finally promotes closure of the chain into the butenolide ring. Isotope experiments using oxygen gas enriched with a heavier form of oxygen confirmed that the ring oxygen comes directly from the air. Structural modeling and targeted mutations pinpointed a single amino acid as the base that kicks off the reaction, and revealed how the bound flavin cofactor cycles between oxidized and reduced forms without ever being consumed.
A finishing enzyme that fine‑tunes the side chain
Once SavA has built the chiral butenolide scaffold, SavB—a cytochrome P450 enzyme—takes over to decorate the attached carbon chain. In the presence of its redox partners and a common cellular cofactor, SavB performs a precise series of oxidations at two adjacent carbon atoms. Time‑resolved analysis uncovered two intermediate molecules: first a singly hydroxylated product, then a keto form, before the fully functional avenolide appears. Nuclear magnetic resonance measurements confirmed the positions and three‑dimensional arrangements of these new groups. The work shows that SavB introduces these oxygen atoms in a defined order and with strict control over orientation, an important feature for the hormone’s biological activity. 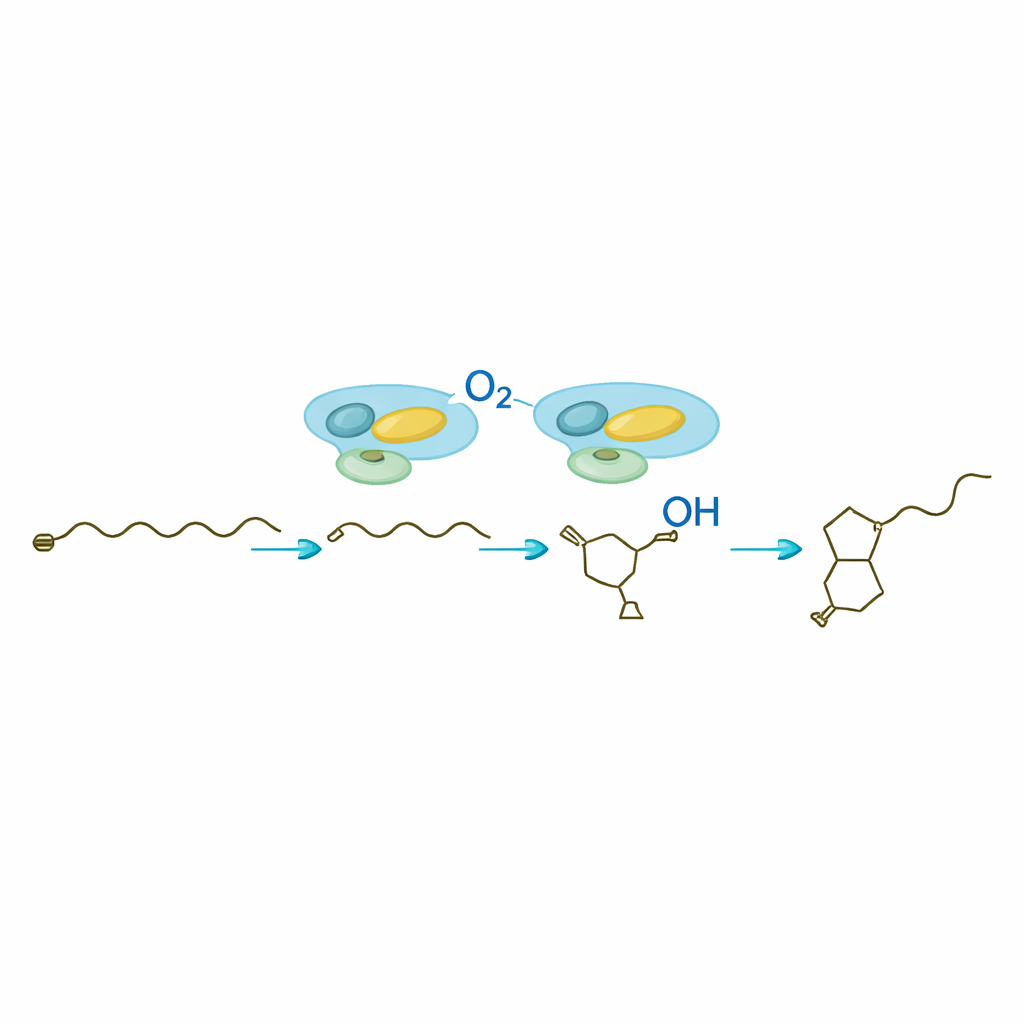
Green chemistry lessons from bacterial enzymes
Together, SavA and SavB convert an ordinary fatty‑acid‑derived building block into a finely tuned signaling molecule using only oxygen from the air and standard cellular helpers. Unlike many industrial processes, SavA does not need extra reducing agents or sacrificial reagents; its flavin cofactor simply shuttles electrons while the substrate itself provides the driving force. The authors highlight SavA as a new kind of multi‑talented flavoenzyme with strong potential as a biocatalyst for sustainable manufacturing of butenolides and related motifs. In practical terms, harnessing or engineering such enzymes could one day allow factories—or engineered microbes—to make important drug fragments and agricultural chemicals from renewable feedstocks under mild conditions, reducing both cost and environmental footprint.
Citation: Li, W., Zhao, J., Zeng, W. et al. Trifunctional flavoenzyme-catalyzed asymmetric 4-alkyl-butenolide assembly in avenolide biosynthesis. Nat Commun 17, 2459 (2026). https://doi.org/10.1038/s41467-026-69265-8
Keywords: butenolide biosynthesis, flavoenzyme SavA, avenolide hormone, biocatalysis, Streptomyces signaling