Clear Sky Science · en
Degraders of the dengue virus capsid protein exhibit differentiated pharmacology relative to capsid inhibitors
Turning the Virus’s Shell Against Itself
Dengue fever infects hundreds of millions of people each year, yet doctors still lack reliable antiviral drugs to treat it. This study explores a fresh way to disarm dengue virus by destroying one of its most important building blocks inside infected cells, rather than simply trying to block its activity. The work shows that carefully designed molecules can tag a key viral protein for disposal by the cell’s own waste‑management system, cutting virus production and blunting its ability to hide from our immune defenses.
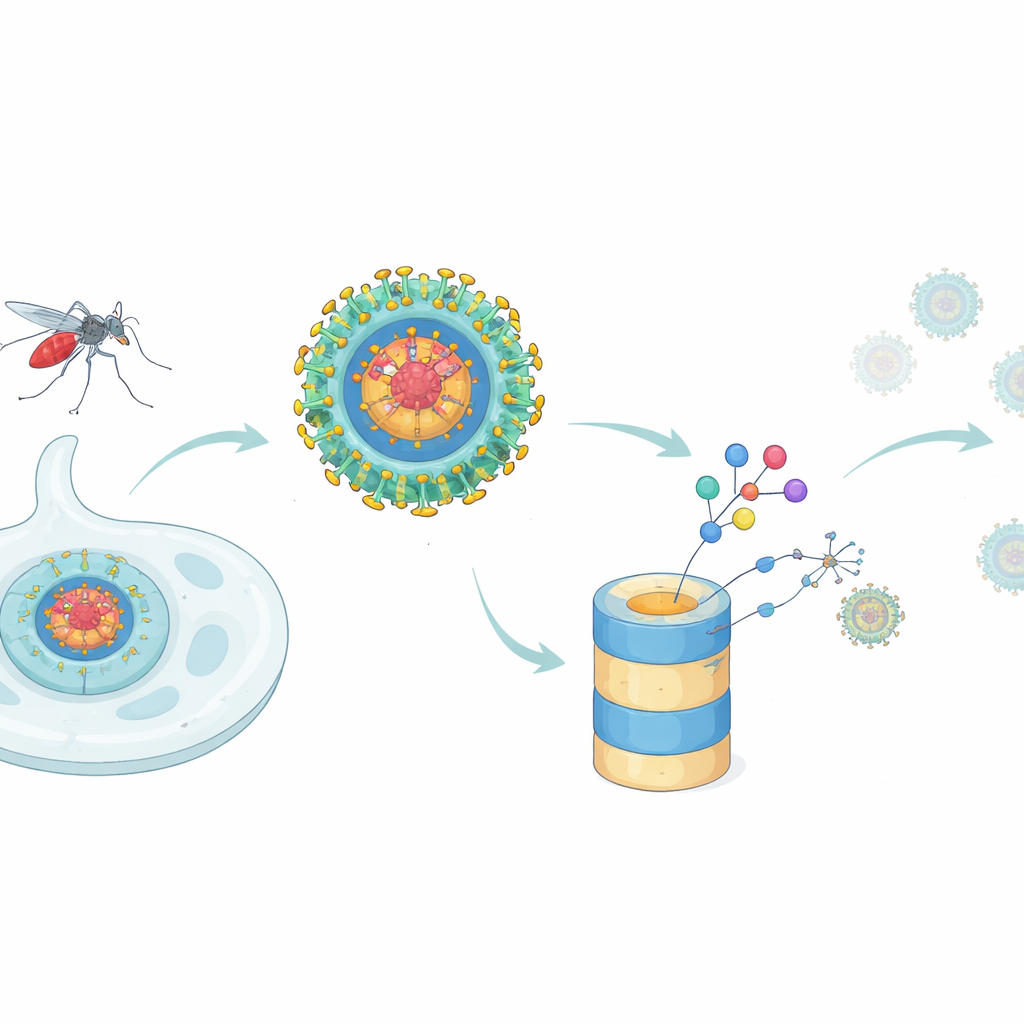
A New Tactic for Stopping Dengue
Most antiviral drugs act like plugs in a machine: they sit on one functional spot of a viral protein and try to block it. That approach struggles when a single viral protein does many different jobs, or when small mutations weaken the drug’s grip. Dengue’s capsid protein is a prime example. It forms the core shell that packages viral genetic material into new particles, but it also meddles with the host cell by interacting with many human proteins and by dampening the body’s interferon response, a frontline antiviral alarm. The authors asked whether a newer drug concept—targeted protein degradation—could go beyond classic inhibitors by actually removing the capsid protein from infected cells.
Designing a Molecular “Bounty Hunter”
To build such a degrader, the researchers started from ST148, a known small molecule that binds the dengue capsid and interferes with assembly of new viral particles. They chemically linked ST148 to a second module that recruits a human enzyme complex responsible for tagging proteins for destruction. The resulting chimeric molecules, called PROTACs, are shaped to grab the capsid with one end and an E3 ligase complex with the other, bringing them together so the capsid can be marked with ubiquitin tags and fed into the cell’s proteasome, the main protein shredder. By testing many linker lengths and ligase‑recruiting groups, they identified one standout compound, dubbed RPG‑01‑132, that reliably reduced capsid levels in infected cells in a manner dependent on the CRBN form of the ligase and on normal proteasome function.
Shutting Down Virus Production and Immune Evasion
Once they had a working degrader, the team examined what it actually did to dengue infection. In liver‑derived cells infected with dengue virus, RPG‑01‑132 caused a strong drop in the number of infectious viruses released, at concentrations where it only partially lowered total capsid levels. Importantly, other viral proteins and viral RNA remained unchanged, showing that the compound was not simply poisoning overall replication but specifically acting through capsid loss. Electron microscopy gave a visual readout: cells treated with the original inhibitor ST148 accumulated stacks of partially formed virus particles in the endoplasmic reticulum, consistent with a block in assembly, whereas cells treated with the degrader showed almost no visible virions at all. The degrader also relieved the capsid’s ability to silence interferon‑β signaling in a reporter system, suggesting that capsid’s non‑structural role in disarming innate immunity can be reversed by degrading it.
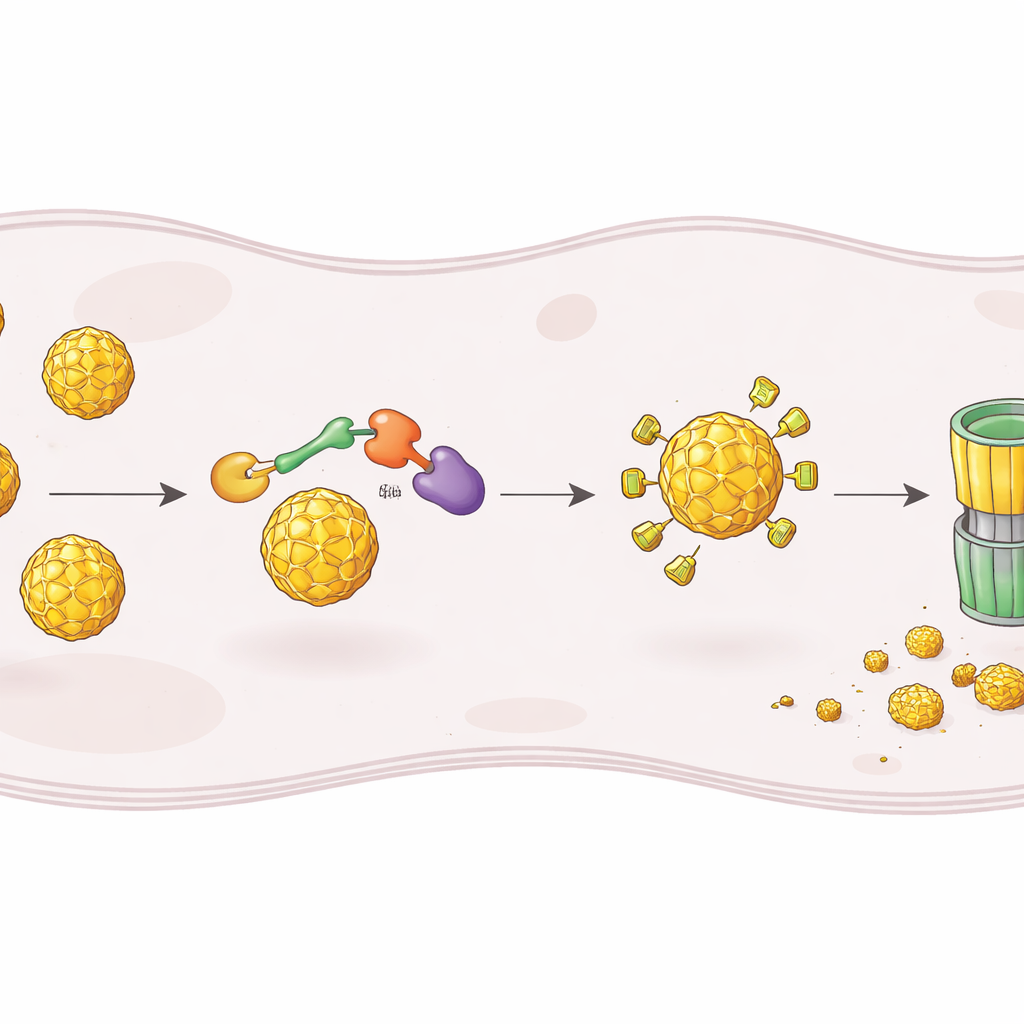
Beating Viral Diversity and Drug Resistance
Dengue virus comes in four major serotypes that differ in how well they respond to capsid‑targeting inhibitors, and resistance can arise from single mutations. The authors compared the degrader to ST148 across representative strains of all four serotypes and against a virus carrying a mutation (S34L in the capsid) previously shown to make ST148 ineffective. As expected, ST148 worked best on one serotype and lost activity against the resistant mutant. RPG‑01‑132, in contrast, showed similar antiviral potency across all serotypes and retained activity against the S34L mutant, while still acting through the same CRBN‑dependent degradation pathway. This illustrates a key advantage of “event‑driven” pharmacology: the degrader does not need to cling tightly to every capsid molecule at all times, as long as it can trigger enough degradation events to tip the balance.
What This Means for Future Dengue Treatments
This study demonstrates that dengue’s capsid protein can be attacked not only as a structural component of the virus but as a removable hub that supports multiple steps of infection and immune evasion. By converting a traditional inhibitor into a degrader, the researchers created a compound that blocks virus production, lifts some of the virus’s suppression of innate immunity, and remains effective across diverse viral strains and a known resistance mutation. RPG‑01‑132 itself will need optimization—better cell entry, stronger activity in animal models, and careful safety checks—but it provides a compelling proof of concept. More broadly, the work suggests that hijacking the cell’s own disposal machinery could open new routes to antiviral drugs that are harder for viruses to evade and that can neutralize proteins with many different roles inside infected cells.
Citation: Chakravarty, A., Wang, LN., Golden, R.P. et al. Degraders of the dengue virus capsid protein exhibit differentiated pharmacology relative to capsid inhibitors. Nat Commun 17, 2594 (2026). https://doi.org/10.1038/s41467-026-69263-w
Keywords: dengue virus, capsid protein, targeted protein degradation, antiviral drug design, PROTACs