Clear Sky Science · en
Engineering d-orbital of copper single-atom sites toward industrial-level electrocatalytic methanation
Turning Power Plant Exhaust into Fuel
Burning coal and gas for electricity releases huge amounts of carbon dioxide into the air, driving climate change. This study explores an emerging idea: instead of simply treating carbon dioxide as waste, can we use electricity to turn it back into energy-rich fuel right at the power plant? The researchers focus on converting carbon dioxide into methane, the main component of natural gas, using a highly efficient and durable catalyst made from copper and titanium oxide. Their goal is to reach performance levels suitable for industry, not just the laboratory.
Why Methane from Carbon Dioxide Matters
Many existing power plants will keep running for years, especially overseas coal plants that currently emit hundreds of millions of tons of carbon dioxide annually. Capturing this carbon dioxide and electrochemically converting it into methane offers a way to both cut emissions and create a usable fuel. Methane is attractive because it stores a lot of energy and can be burned in existing turbines and gas infrastructure. However, most current copper-based catalysts that turn carbon dioxide into methane work too slowly, waste a lot of energy, or break down under the high currents needed for real-world devices.
Designing a Smarter Copper Site
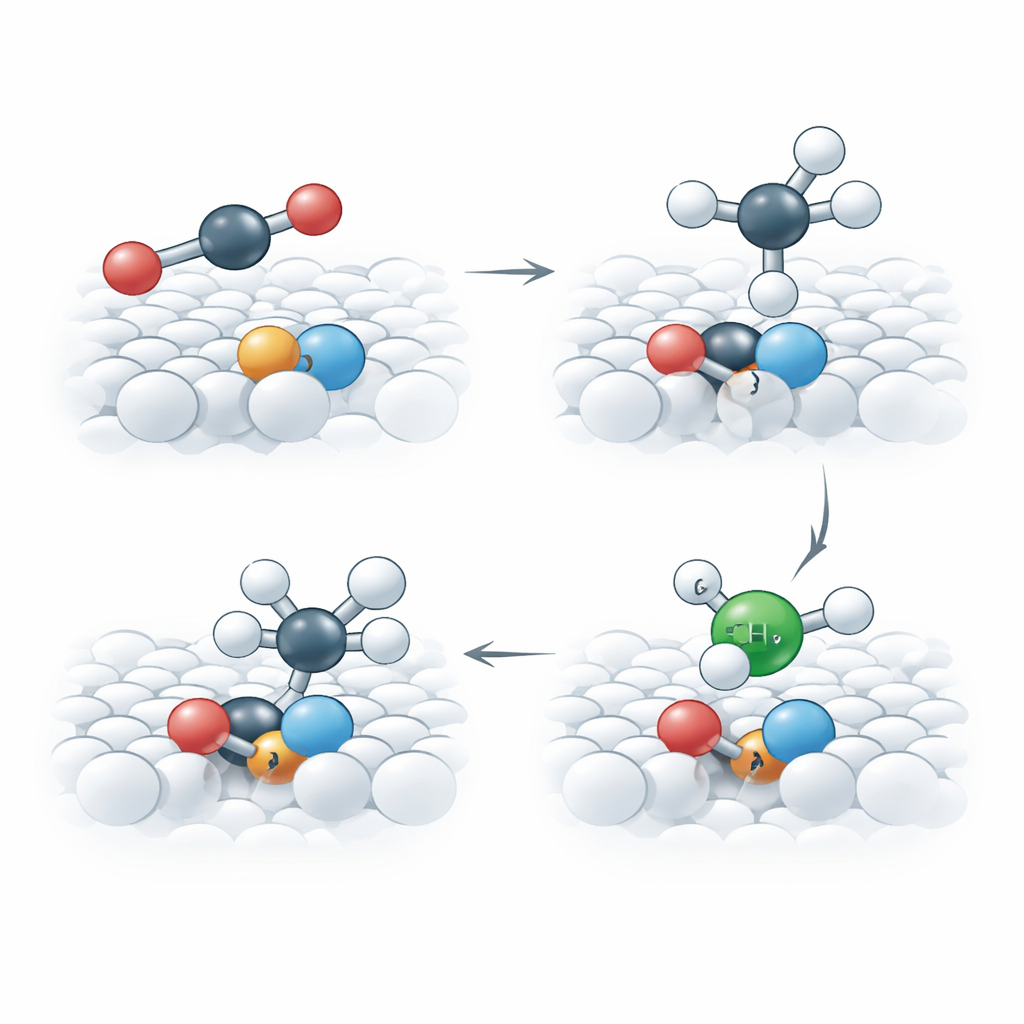
The heart of this work is a new kind of copper catalyst called a single-atom catalyst, in which isolated copper atoms are anchored on a solid support rather than clumped into particles. The team uses titanium oxide as the support and deliberately removes some oxygen atoms from its crystal lattice, creating tiny “vacancies” that change how nearby metal atoms interact. By carefully treating a copper‑doped titanium oxide in hydrogen, they form a compound the authors call Cu–Ti1O3, where single copper atoms sit next to titanium atoms and share electrons directly. These copper–titanium pairs behave very differently from conventional copper sites that are surrounded mainly by oxygen.
How Tiny Vacancies Control the Reaction
Advanced simulations and measurements reveal what is special about these engineered copper sites. The missing oxygen atoms encourage a strong electronic link between copper and titanium, which makes the copper more localized and “harder” in chemical terms. This helps carbon dioxide bind in a bent, activated form and stabilizes a critical reaction intermediate that contains carbon, oxygen, and hydrogen. The study shows that oxygen from this intermediate can temporarily slip into the nearby vacancy, behaving like a reversible part of the crystal lattice. This clever rearrangement makes it easier to break the carbon–oxygen bond and continue the sequence of steps leading to methane, without damaging the catalyst itself.
From Theory to Industrial-Scale Performance
To test whether these microscopic improvements matter in practice, the researchers build flow-cell reactors and a zero-gap electrolyzer similar to systems being developed for industry. In alkaline solution, the Cu–Ti1O3 catalyst converts carbon dioxide to methane with a Faradaic efficiency of about three-quarters, meaning most of the input current goes to methane rather than unwanted by-products like hydrogen. It also reaches very high methane production rates—far above many earlier copper catalysts—while using electricity efficiently. Perhaps most impressively, in a larger 5 cm² cell running at industrial-level current, the catalyst maintains high methane selectivity for more than 1,200 hours, vastly outlasting a comparison copper catalyst that quickly degrades and forms copper nanoparticles.
Implications for Cleaner Power Plants
In simple terms, this work shows that reshaping how electrons are shared around single copper atoms can turn a fragile, mediocre catalyst into a fast, long-lived “machine” for turning carbon dioxide into methane. By using oxygen vacancies in titanium oxide to strengthen the copper–titanium partnership, the researchers unlock a reaction pathway that favors methane and protects the active sites over long operation times. Although real power plants involve many additional engineering and economic questions, the demonstrated performance and durability suggest that such catalysts could form the core of future devices that recycle flue-gas carbon dioxide into useful fuel, easing the path toward lower-carbon electricity.
Citation: Liu, Z., Cai, J., Dong, S. et al. Engineering d-orbital of copper single-atom sites toward industrial-level electrocatalytic methanation. Nat Commun 17, 2723 (2026). https://doi.org/10.1038/s41467-026-69260-z
Keywords: electrocatalytic CO2 reduction, methane fuel, copper single-atom catalyst, oxygen vacancies, power plant decarbonization