Clear Sky Science · en
Permeable intimate membrane electrode interface with optimized micro-environment for CO2 electroreduction in pure water
Turning Green Electricity into Useful Carbon
As the world works to cut carbon emissions, one appealing idea is to turn waste carbon dioxide (CO2) into useful fuels and chemicals using clean electricity. This study tackles a key roadblock: most efficient CO2-to-fuel devices need salty liquids to work well, which adds cost and complexity. The authors show how a redesigned "intimate" membrane–electrode structure lets a CO2 electrolyzer run on pure water instead, keeping performance high while simplifying the system.
Why Pure Water Matters
Today’s leading CO2 electrolysis systems often rely on dissolved salts such as potassium bicarbonate or potassium hydroxide. These salts help carry electric charge and shape the tiny environment where CO2 is turned into products, but they also create headaches: salt can crystallize and clog the device, and separating products from briny waste streams is costly. Running the device with pure water would avoid these issues and make large-scale plants easier to build and maintain. However, pure water conducts electricity poorly and lacks helpful metal ions, so current devices suffer from sluggish reactions, extra heat losses, and low selectivity for the desired product, carbon monoxide (CO).
Building a Closer Connection Inside the Device
The heart of this work is a new type of electrode called a permeable intimate membrane (PIM) electrode. In a standard design, a porous catalyst layer that activates CO2 is pressed against a separate ion-conducting membrane, leaving tiny gaps and dead zones that hinder the flow of water and charged species. In the PIM design, the researchers pour a liquid ion-conducting polymer directly onto a silver-based catalyst layer, letting it seep into the pores before solidifying into a thin membrane. This creates a tightly bonded sandwich of gas diffusion layer, catalyst, and ion-conducting layer with continuous internal channels for water and hydroxide ions to move. 
Better Performance with Less Energy
When tested in a membrane–electrode assembly powered by pure water, the PIM electrode made with a specific polymer (called QAPPT) delivers more than 90 percent of the electrical current into CO across a wide operating window, from 50 to 400 milliamps per square centimeter, and still about 84 percent at even higher load. Compared with the conventional pressed structure, the new design lowers the cell voltage at the same current, which means less wasted energy and fewer losses as heat. Overall energy efficiency improves by about 35 percent. The device also uses CO2 more effectively in a single pass, reaching over 80 percent conversion at certain flow rates—exceeding theoretical limits set by typical alkaline systems. 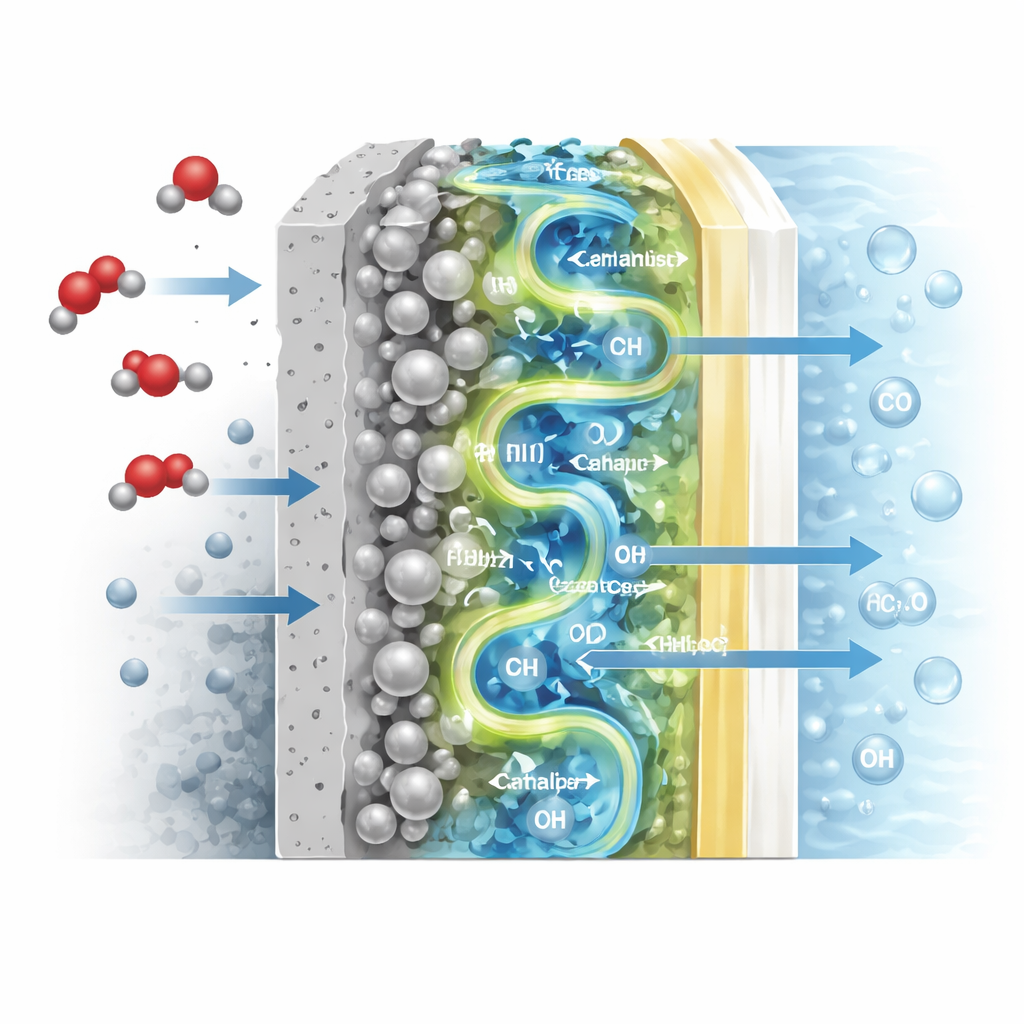
Stable, Scalable, and Versatile
Beyond raw efficiency, the new structure proves robust. In small cells, it runs for more than 200 hours with high CO output. A larger, 10 by 10 centimeter version operating at 3.2 amperes also maintains stable voltage and over 80 percent selectivity for CO over hundreds of hours. The approach works not only in pure water but also in alkaline, neutral, and even acidic solutions, and with different catalyst types, including silver particles of various sizes and bismuth for making formic acid. Economic modeling suggests that, at realistic scales and power prices, the improved design could cut the cost of producing CO to roughly half or less of its current market price, making this route attractive for industry.
Water at the Interface: The Hidden Helper
The authors go further to probe why the PIM structure works so well. Using advanced infrared methods and computer simulations, they show that the intimate contact between catalyst and polymer reorganizes the network of water molecules at the reaction surface. In the optimized structure, water forms a stronger, more ordered hydrogen-bond network that both speeds the key reaction step—adding hydrogen to a CO2-derived intermediate—and reduces a side reaction that simply makes hydrogen gas. Simulations confirm that CO2 diffuses more easily and binds in a more reactive bent shape on the silver surface when this water network is present. In effect, the redesigned interface quietly tunes the “personality” of water to favor CO formation.
What This Means Going Forward
By rethinking how the membrane and catalyst are joined, this work shows that efficient CO2 electrolysis does not have to depend on complex salty liquids. A tightly integrated, permeable electrode lets pure water feed high-performing cells that make valuable carbon-based products while using energy more wisely. For non-specialists, the take-home message is that smart control of the tiny environment at material interfaces—including how water behaves there—can unlock cleaner, cheaper routes to recycle CO2, bringing practical carbon-to-fuel technologies closer to reality.
Citation: Zheng, Z., Bi, S., Zhou, X. et al. Permeable intimate membrane electrode interface with optimized micro-environment for CO2 electroreduction in pure water. Nat Commun 17, 2570 (2026). https://doi.org/10.1038/s41467-026-69259-6
Keywords: CO2 electroreduction, pure water electrolyzer, membrane electrode assembly, interface engineering, carbon utilization