Clear Sky Science · en
Cooperative anion activation at a cobalt center through ion pairing and ligand design
Why tiny partners around metals matter
Chemists often focus on the main “actor” in a reaction — a metal atom that helps break and make bonds. But this paper shows that the quiet partners hovering nearby, invisible to the naked eye, can completely change what the metal does. By carefully shaping the space around a cobalt atom, the authors reveal how two almost interchangeable anions, often treated as passive bystanders in chemistry and batteries, can lead to sharply different outcomes. 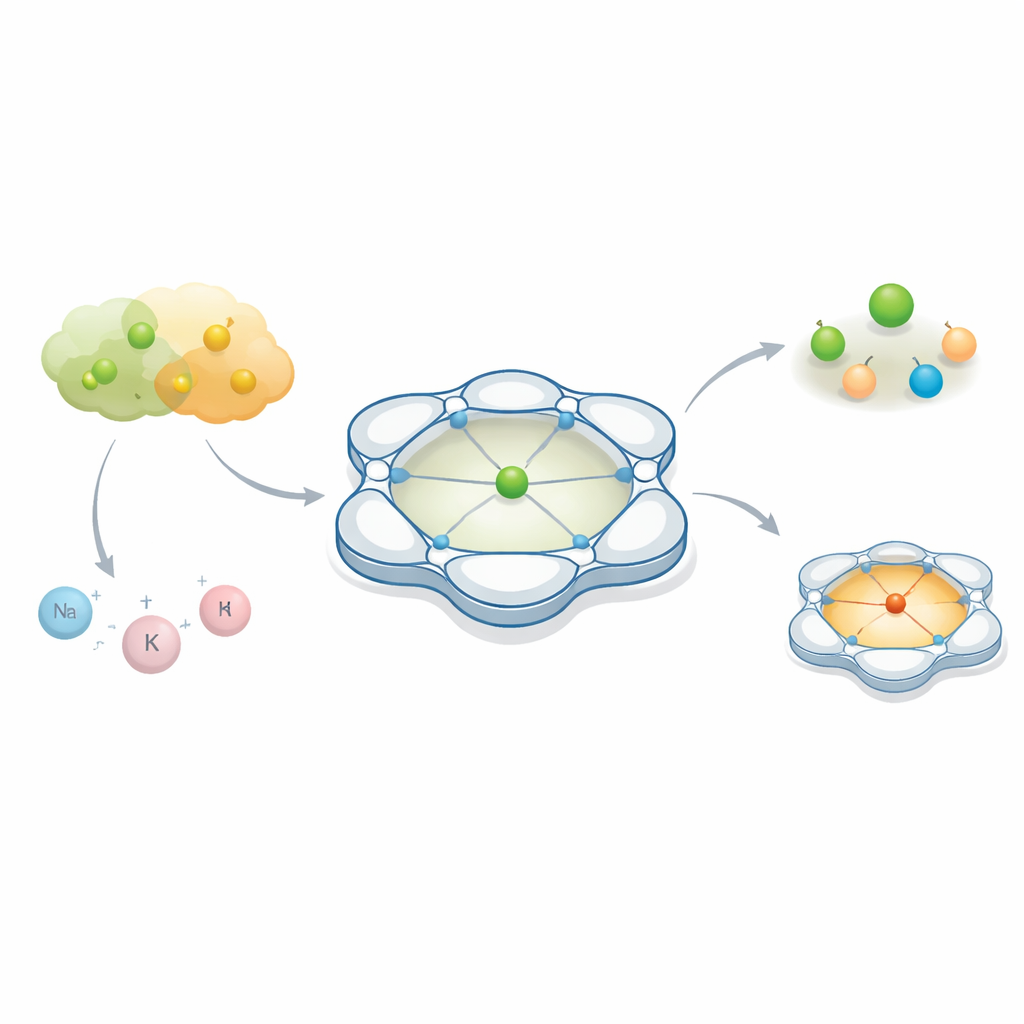
Building a tailored pocket around cobalt
The researchers designed a cage-like organic framework called Py4Im that wraps tightly around a cobalt ion. This framework is built from several linked pyridine rings and an imidazolidine unit, which together create a rigid, bowl-shaped cavity containing a single N–H group. That small cavity is “protic,” meaning it can form hydrogen bonds, and it points in a specific direction, like a tiny docking bay for incoming anions. When cobalt is assembled with this ligand, the result is a family of positively charged complexes whose overall shape and internal pocket stay the same while the counterion — the negatively charged partner — can be swapped. This makes the system an ideal test bed for seeing how different anions behave in the same controlled environment.
Two look‑alike anions, two very different behaviors
The team compared two workhorse anions, tetrafluoroborate (BF4−) and hexafluorophosphate (PF6−), which are widely used as so‑called weakly coordinating anions. They are popular because they usually keep their distance from metal centers, helping stabilize highly charged species without getting directly involved. Surprisingly, inside the Py4Im cavity they do not behave alike. Under mild conditions, PF6− donates a fluoride ion to cobalt, breaking a strong P–F bond and forming a well‑defined cobalt–fluoride complex. In contrast, BF4−, which is usually considered the more “fragile” of the two, refuses to give up fluoride in the same setting. Instead, it settles into a stable arrangement bound to the cobalt center without completing the bond‑breaking step.
Watching ions pair up and move
To understand these contrasting behaviors, the authors used a combination of high‑resolution NMR techniques and quantum‑chemical calculations. Diffusion NMR experiments measured how fast the positive and negative species move in solution, which reveals how tightly they are paired. These measurements showed that BF4− forms a closer, more persistent ion pair with the cobalt complex than PF6− does. The Py4Im cavity positions the anion directly beneath the imidazolidine ring, where hydrogen bonds from the N–H and nearby C–H groups hold it in place. Computer calculations confirmed that this stronger, directional pairing with BF4− stabilizes the starting state so much that breaking the B–F bond becomes slightly unfavorable, even though that bond is intrinsically weaker than the P–F bond in PF6−. PF6−, pairing more loosely, can approach, transfer fluoride to cobalt, and depart as PF5, leading to a fluoride‑bearing cobalt complex that is thermodynamically accessible. 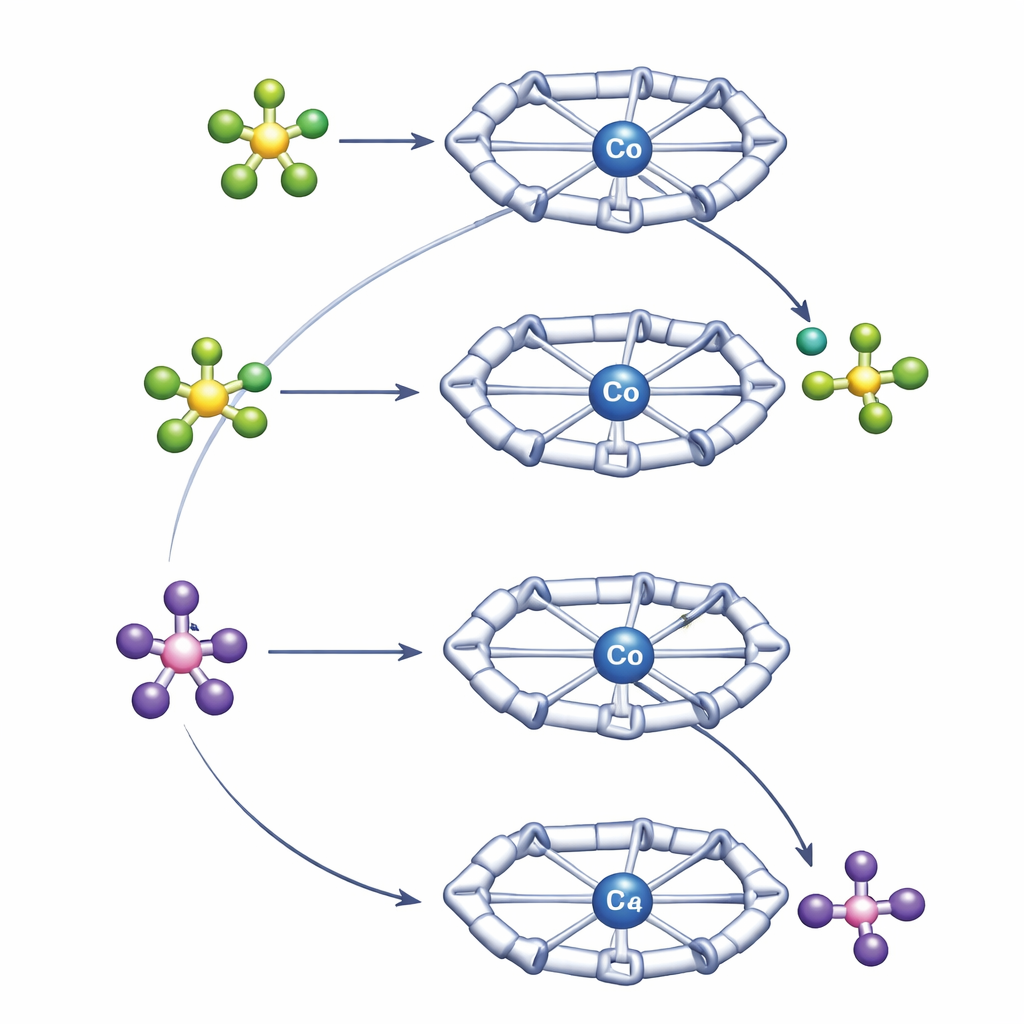
Turning a bound fluoride into a useful tool
Once formed, the cobalt–fluoride complex is not a dead end. The authors show that it behaves as a nucleophilic fluoride source — in simpler terms, it can deliver fluoride to positively charged or electron‑poor partners. In solution, this cobalt–fluoride species cleanly transfers fluoride to reactive carbon centers, to silicon atoms in chlorosilanes, and to acyl chlorides, generating organic fluorides while the cobalt complex switches back to its chloride form or binds the new partner. In a clever twist, the team also demonstrates that BF4− can be coaxed into giving up fluoride if a separate base is present to “catch” the BF3 fragment that is left behind. This converts the otherwise reluctant BF4− pathway into one that also yields the same cobalt–fluoride product.
What this means for designing smarter catalysts
To a non‑specialist, the key message is that ions once treated as inert background salts can in fact steer chemical reactions in powerful ways. By sculpting a precise pocket around a metal center and controlling how tightly anions pair with it, the authors flipped the expected reactivity of two nearly identical species. PF6−, normally viewed as very robust, becomes the easier source of fluoride, while BF4− is locked down by stronger pairing unless the system is helped along. This work provides a blueprint for using ligand design and counterion choice together to tune how reactive a metal complex is — an idea that could influence fields ranging from homogeneous catalysis to electrolyte design for advanced batteries, where the “silent partners” in solution may be anything but passive.
Citation: Tarifa, L., Cano-Asensio, J., López, J.A. et al. Cooperative anion activation at a cobalt center through ion pairing and ligand design. Nat Commun 17, 2469 (2026). https://doi.org/10.1038/s41467-026-69257-8
Keywords: cobalt complexes, weakly coordinating anions, fluoride transfer, ion pairing, ligand design