Clear Sky Science · en
Metallic molybdenum sulfide catalyses protometabolic carbon dioxide reaction networks under extreme conditions
How Rocks May Have Jump-Started Life’s Chemistry
Long before living cells evolved enzymes, Earth still needed ways to turn simple gases like carbon dioxide into the organic molecules that life depends on. This study explores whether certain naturally occurring minerals, similar to those found near undersea hot springs, could have acted as primitive “chemical engines.” By showing that a metallic form of molybdenum sulfide can transform carbon dioxide into a surprisingly rich web of organic compounds, the work offers a concrete scenario for how life’s core chemistry might have begun on a lifeless planet.

Hot Vents and Hidden Chemical Engines
Modern life runs on intricate networks of reactions that shuttle carbon through cells and ecosystems. These networks rely on proteins called enzymes, which are themselves products of evolution. The big question is what came before. The authors looked to hydrothermal vents—natural hot-water chimneys on the ocean floor rich in metal sulfides and hydrogen gas—as likely settings for the earliest carbon chemistry. In such places, hot, pressurized water meets rocks containing metals like iron, nickel, and molybdenum. Past experiments showed that some of these metals can turn carbon dioxide into small organics, but usually only along fragments of life’s pathways, not full networks.
A Mineral Imitator of Ancient Enzymes
The team focused on a particular form of molybdenum sulfide, called the 1T′ phase, whose atomic arrangement mimics the metal-sulfur centers found in modern enzymes that process carbon dioxide. Under high temperature and pressure in water, with hydrogen as a fuel, this mineral catalyzed the conversion of dissolved carbon dioxide (modeled as bicarbonate) into an unexpectedly broad range of organic acids. By carefully varying temperature, reaction time, and gas pressure, and by tracking products with chromatography, mass spectrometry, and NMR, they identified 32 different intermediates and end products emerging from the same simple starting carbon source.
Rebuilding Life’s Core Carbon Pathways Without Enzymes
Those 32 compounds are not random. Many are the same molecules that sit at the heart of modern metabolism—such as acetate, pyruvate, oxaloacetate, succinate, and alpha-ketoglutarate. Collectively, they map onto five major carbon-fixing routes used by contemporary microbes: the acetyl–CoA pathway and variants of the Krebs cycle and related loops. In the experiments, these pathways appeared as an interconnected network: acetate linked several cycles; larger molecules broke down and re-formed; and key intermediates disappeared and later reappeared as conditions changed. The production of multi-carbon acids reached high efficiency, with up to about 70% of the converted carbon ending up in molecules containing two or more carbons. The reaction network also showed oscillations in which the amounts of certain products rose and fell over time, echoing the dynamic behavior of living chemical systems.
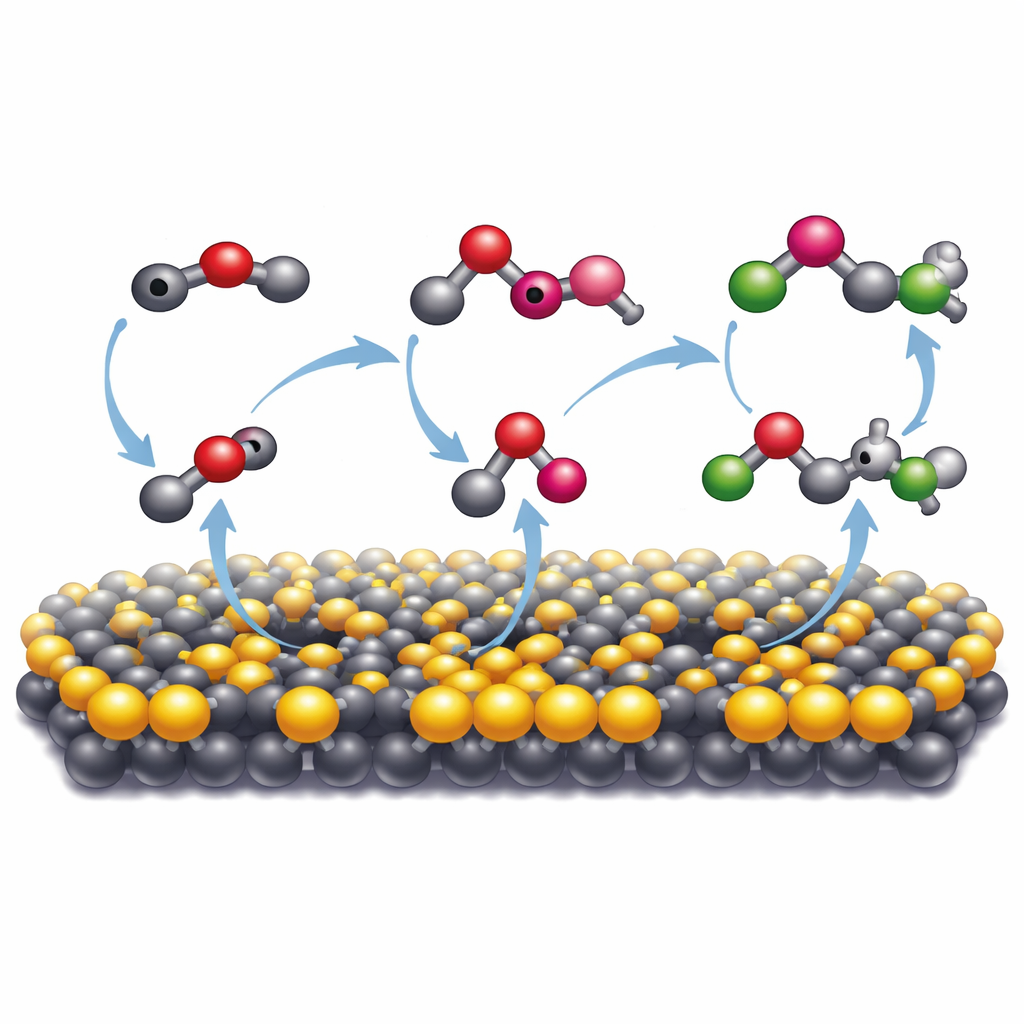
How the Mineral Makes Carbon Atoms Join Forces
To understand why this particular mineral was so effective, the researchers compared it to a more common form of molybdenum sulfide that has a different atomic structure. Only the metallic, distorted form—with many missing sulfur atoms—drove extensive formation of larger organics. Spectroscopic techniques revealed that on this surface, carbon monoxide derived from carbon dioxide binds strongly and is further reduced to highly reactive fragments, or radicals. These radicals are stabilized just enough on the mineral surface to bump into one another and form new carbon–carbon bonds. Electron-spin measurements confirmed the presence of such radicals, and adding a radical “trap” chemical sharply suppressed multi-carbon products. Computer simulations backed this picture, showing that sulfur vacancies and the altered electronic structure of the 1T′ phase make it easier for the surface to donate electrons and to pull carbon fragments close together so they can combine.
From Stone Chemistry to the First Metabolism
Put together, these findings suggest that certain metal sulfide minerals on the early Earth could have run complex, self-organizing carbon chemistry long before enzymes existed. In hydrothermal vent settings, where hot, hydrogen-rich fluids meet carbon dioxide and metal-bearing rocks, a mineral like 1T′ molybdenum sulfide could have continuously converted inorganic carbon into a web of interconnected organic pathways resembling modern metabolic cycles. To a non-specialist, the key message is that life’s central chemistry may not have needed life to get started: under the right conditions, simple rocks, water, and gas can spontaneously build many of the same molecules and reaction loops that living cells still use today.
Citation: Chen, P., Liu, X., He, D. et al. Metallic molybdenum sulfide catalyses protometabolic carbon dioxide reaction networks under extreme conditions. Nat Commun 17, 2395 (2026). https://doi.org/10.1038/s41467-026-69255-w
Keywords: origin of life, prebiotic chemistry, hydrothermal vents, carbon fixation, molybdenum sulfide