Clear Sky Science · en
Axially engineered single atoms in enzyme-mimic-binding pocket steering dehalogenation–polymerization pathways toward water pollutant upcycling
Turning Toxic Water into Useful Materials
Many industrial chemicals that leak into rivers and lakes are both stubborn and toxic, especially those containing chlorine atoms. Instead of simply burning these pollutants away with harsh treatments, this research shows how to transform one such chemical into the building blocks of plastics, cleaning the water and creating a useful product at the same time.
Why Chlorine-Heavy Pollutants Are So Hard to Treat
Modern water treatment often relies on powerful oxidants—chemicals that rip electrons from pollutants until they break down into carbon dioxide and other simple molecules. While effective, this “burn everything” approach consumes large amounts of chemicals, can generate harmful by-products, and wastes the carbon content of the pollutants. Chlorine-containing compounds, such as the widely used 2,4,6-trichlorophenol, are especially troublesome. Their chlorine atoms pull electrons away from the molecule, making it harder for reactive species to start chain reactions. The result is slow clean-up, persistent chlorine in the leftover products, and a risk of new toxic substances.
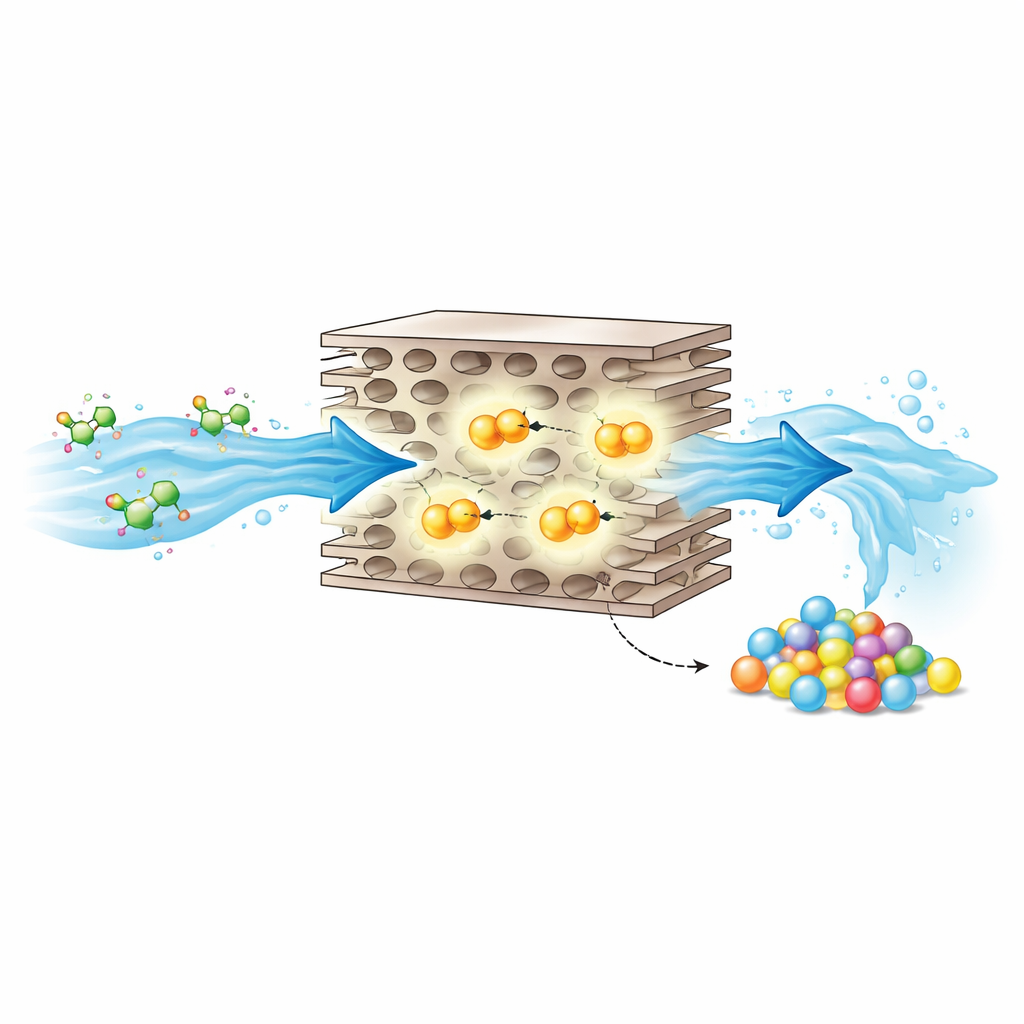
Borrowing Tricks from Natural Enzymes
Nature solves similar problems using enzymes that carefully position metal atoms inside pockets built from amino acids. These enzymes use water-derived hydroxyl groups to attack “activated” sites near halogen atoms, neatly removing chlorine while adding more reactive handles for later chemistry. Inspired by this, the authors designed a solid catalyst that mimics an enzyme’s active pocket at the atomic scale. They anchored individual iron atoms onto sheets of carbon nitride and attached an extra nitrogen atom above each iron, creating a five-fold “axially coordinated” site. Surrounding carbon and nitrogen atoms form a hydrophobic–polar microenvironment that behaves like a binding pocket, holding both pollutant and oxidant close to the iron center.
A New, Quiet Pathway for Oxidation
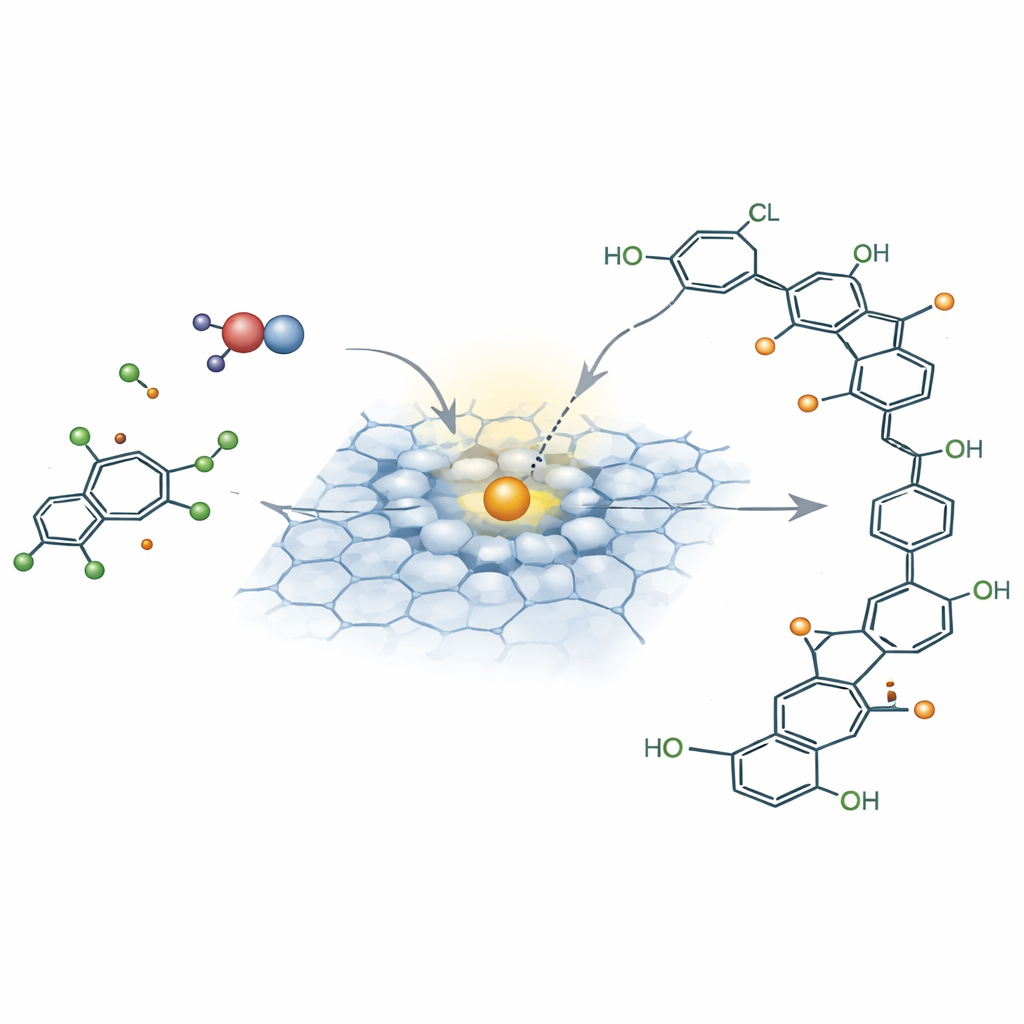
When the oxidant peroxymonosulfate meets this catalyst, it does not produce the familiar swarm of short-lived radicals or singlet oxygen that many advanced oxidation systems rely on. Instead, spectroscopic measurements and electrochemical tests show that the oxidant forms a stable surface complex with the iron site. This complex pulls electrons directly from the nearby chlorinated pollutant in a controlled, two-electron step. In the process, the oxidant is reshaped into a reactive surface-bound hydroxyl species, while the pollutant passes through a short-lived, positively charged intermediate. This “electron-transfer pathway” is short-range and highly selective: pollutant molecules must sit right next to the activated oxidant on the catalyst surface, rather than being attacked randomly in the water.
From Dechlorination to Polymer Building Blocks
Once the pollutant has given up electrons, its chlorine atoms become easier to dislodge. Water molecules step in as nucleophiles—electron donors—that substitute hydroxyl groups where chlorine once sat, first at the most exposed positions and then at neighboring sites. Computational modeling and isotope-tracing experiments show that these new hydroxyl groups come from water rather than the oxidant itself. As chlorine is stripped away, the molecule gains multiple hydroxyl groups that stabilize reactive intermediates and open up many new connection points. Instead of breaking down into gas, these modified molecules link together mainly through oxygen bridges to form polyphenylene ether–like oligomers: mid-sized, largely dechlorinated polymers that stick to the catalyst surface.
Scaling Up: Cleaning Water While Making Plastics
Crucially, these polymeric products can be collected and processed. By washing the catalyst with organic solvents, the researchers recovered a significant fraction of the carbon as solid oligomers, which were then turned into uniform plastic pellets using standard extrusion and granulation steps. Tests showed that the catalyst maintains high activity over many cycles and in realistic setups, including membrane reactors and fluidized beds treating chlorinated wastewater. Economic and environmental analyses suggest that this enzyme-inspired system could operate at lower cost and with a much smaller carbon footprint than traditional oxidation methods, especially once the value of the recovered plastics is counted.
What This Means for Future Water Treatment
Instead of choosing between pollution control and resource recovery, this work points to a future where contaminated water becomes a feedstock. By engineering single metal atoms to behave like the cores of natural enzymes, the authors direct the reaction away from total destruction and toward selective dechlorination and polymer formation. In simple terms, they turn a problematic chlorinated pollutant into safer, chlorine-free plastic precursors while simultaneously purifying the water, demonstrating a promising path toward cleaner and more circular water treatment technologies.
Citation: Wu, B., Li, Z., Zhang, J. et al. Axially engineered single atoms in enzyme-mimic-binding pocket steering dehalogenation–polymerization pathways toward water pollutant upcycling. Nat Commun 17, 2405 (2026). https://doi.org/10.1038/s41467-026-69253-y
Keywords: water pollution, chlorinated chemicals, advanced oxidation, single-atom catalysts, plastic upcycling