Clear Sky Science · en
Hepatocytes functionally reprogrammed by KIAA1199-high colorectal cancer cells favour the accumulation of pro-metastatic Egr1+ neutrophils
When Colon Cancer Targets the Liver
Most people think of cancer spreading as rogue cells simply breaking away and drifting to new organs. This study shows a more unsettling story: colorectal (colon) tumors can quietly rewire the liver long before any cancer cells arrive, building a welcoming “soil” in which future tumors can easily take root. Understanding this hidden preparation phase could open the door to treatments that stop liver metastases—the main cause of death in colorectal cancer—before they begin.
A Stealthy Setup Before Tumors Arrive
Colorectal cancer often spreads to the liver, yet only some tumors do so aggressively. The authors focused on a molecule called KIAA1199, which is abundant in high‑risk colorectal cancers. In mouse models and patient samples, tumors with high KIAA1199 did not just grow faster; they also primed the liver to become a “pre‑metastatic niche,” a microenvironment that is unusually friendly to incoming cancer cells. Even when liver tumors were seeded experimentally from a separate source, animals carrying KIAA1199‑high colon tumors developed many more liver metastases and died sooner. This showed that signals from the primary tumor, not just wandering cancer cells, were reshaping the liver in advance.
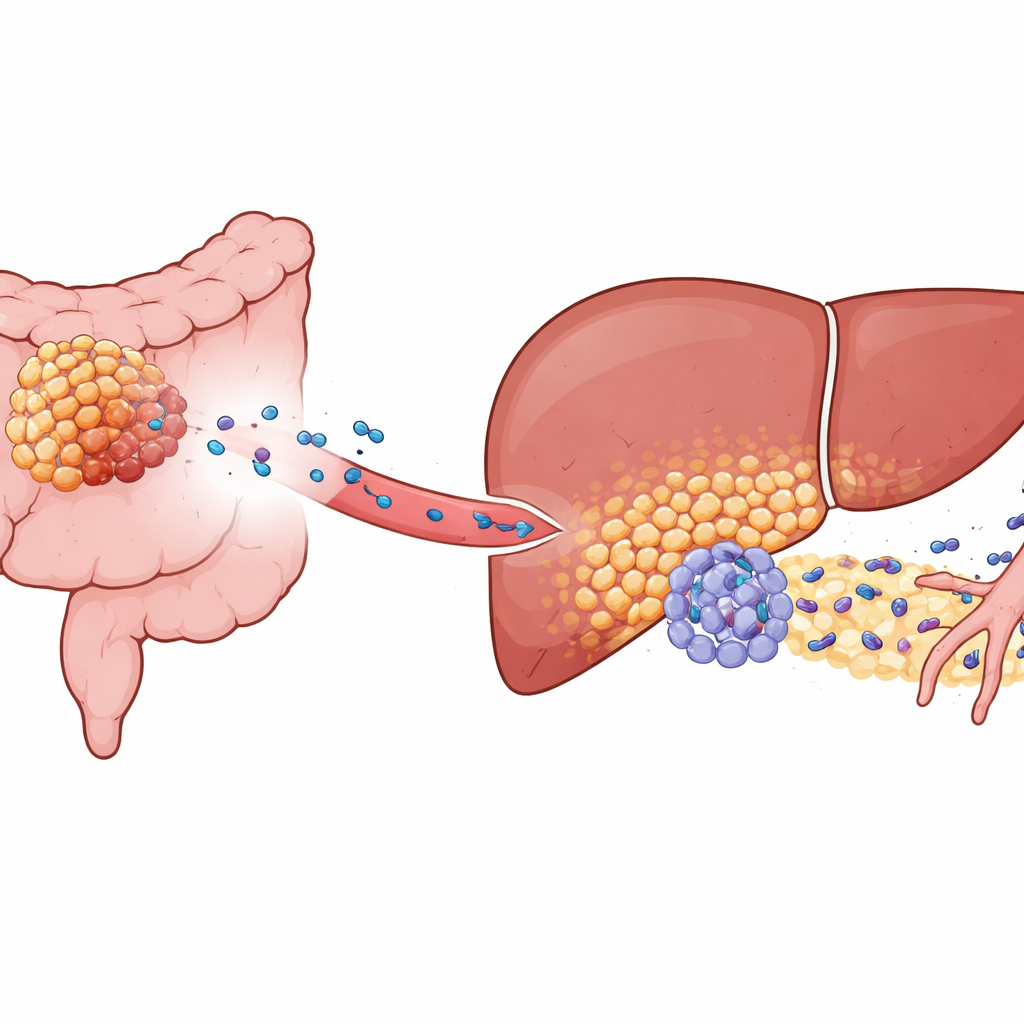
Liver Cells Reprogrammed into Tumor Helpers
Digging deeper, the researchers used single‑cell sequencing and spatial mapping to examine liver tissue at high resolution. They discovered a distinct subset of hepatocytes—normally the workhorse cells of the liver—that had been “functionally reprogrammed.” These cells appeared only when KIAA1199‑high tumors were present. Instead of quietly managing metabolism, the altered hepatocytes switched to a more malignant‑like metabolic style and dialed down a protective regulator called PPARγ, which usually helps keep inflammation and stress responses in check. With PPARγ suppressed, these hepatocytes began producing high levels of a protein called SAA2, which they released into their surroundings, particularly at the border where liver tissue meets invading tumor cells.
Turning Ordinary Defenders into Pro‑Metastatic Neutrophils
Neutrophils—white blood cells best known for fighting infections—also behaved strangely in this primed liver. The authors found a special subset carrying an “Egr1+” signature, named after a transcription factor that reshapes how these cells function. Rather than arriving from the bloodstream already altered, neutrophils seemed to be re‑educated locally by the reprogrammed hepatocytes. SAA2 released from hepatocytes bound to a receptor called FPR2 on nearby neutrophils, flipping on an internal PI3K‑AKT signaling circuit that stabilized Egr1 activity. The result was a longer‑lived, highly active neutrophil that secreted large amounts of VEGFA, a potent driver of new blood vessel growth. These Egr1+ neutrophils clustered at the tumor–liver interface, exactly where new, fragile blood vessels were sprouting.
Building Blood Vessels that Welcome Tumor Cells
The study shows that these re‑educated neutrophils are not bystanders—they actively remodel the liver’s vasculature. In cell culture, factors released by Egr1+ neutrophils made endothelial cells grow, migrate, and form tube‑like structures reminiscent of new vessels. In mice, adding Egr1+ neutrophils boosted liver metastases and sped up death. Blocking key steps in the chain—either the SAA2–FPR2 signal to neutrophils or the downstream PI3K‑AKT pathway—cut off neutrophil survival and VEGFA production, reducing blood vessel growth and metastatic spread. Likewise, restoring PPARγ activity in hepatocytes with the diabetes drug rosiglitazone lowered SAA2 levels, curbed the emergence of Egr1+ neutrophils, and sharply reduced liver metastasis in preclinical models.
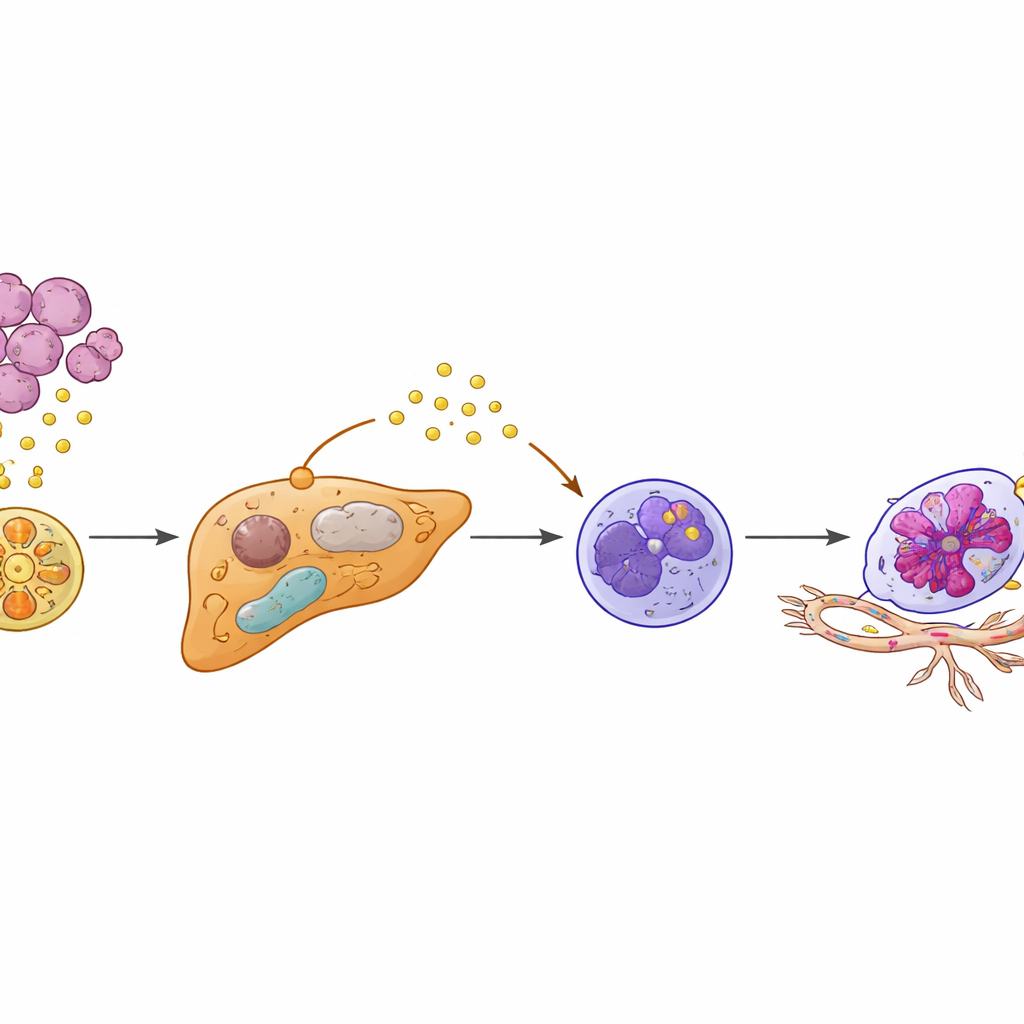
From Mechanism to Prediction and Prevention
Because KIAA1199 in tumors and SAA2 in the liver sit near the top of this cascade, the authors tested whether these proteins could help flag patients at high risk of liver metastasis. In several patient cohorts, people with high blood levels of both markers were far more likely to develop liver metastases and to do so sooner. A simple combined KIAA1199–SAA2 score outperformed either marker alone in predicting risk and was packaged into a clinical “nomogram” that estimates an individual patient’s likelihood of liver spread over the next two years. Together, the findings sketch a vivid picture: KIAA1199‑high colon cancers send out vesicle‑borne signals that rewire hepatocytes, which in turn convert nearby neutrophils into long‑lived, vessel‑building accomplices. By targeting this KIAA1199–PPARγ/SAA2–Egr1 axis—through metabolic drugs, immune‑modulating agents, or both—it may be possible not just to treat existing liver metastases, but to prevent them from taking hold at all.
Citation: Li, L., Zhao, L., Cao, K. et al. Hepatocytes functionally reprogrammed by KIAA1199-high colorectal cancer cells favour the accumulation of pro-metastatic Egr1+ neutrophils. Nat Commun 17, 2462 (2026). https://doi.org/10.1038/s41467-026-69250-1
Keywords: colorectal cancer liver metastasis, pre-metastatic niche, neutrophils, hepatocyte reprogramming, tumor microenvironment