Clear Sky Science · en
Two-step voltage-sensor activation of the human KV7.4 channel and effect of a deafness-associated mutation
How Tiny Ear Gatekeepers Shape Our Hearing
Inside the inner ear, hearing depends on microscopic cells that convert sound vibrations into electrical signals for the brain. This study focuses on a specific protein, the Kv7.4 potassium channel, which acts like a tiny gate in the membrane of these cells. When the gate does not open and close properly, hearing can slowly fade over time. The researchers set out to understand exactly how this gate responds to electrical signals and why a particular inherited mutation linked to deafness causes the gate to malfunction.
The Gate in Our Hearing Cells
Outer hair cells in the cochlea help fine-tune sound and amplify vibrations. Their performance relies heavily on potassium channels, including Kv7.4, which let potassium ions flow out of the cell and help reset its electrical state after sound stimulation. The Kv7.4 protein has a “pore” that ions pass through and a “voltage sensor” that detects changes in the cell’s electrical charge and tells the pore when to open. Defects in the gene that encodes Kv7.4 (KCNQ4) are known to cause a form of progressive hearing loss in families. Yet, until now, the detailed motions of the voltage sensor and how they control pore opening were poorly understood.
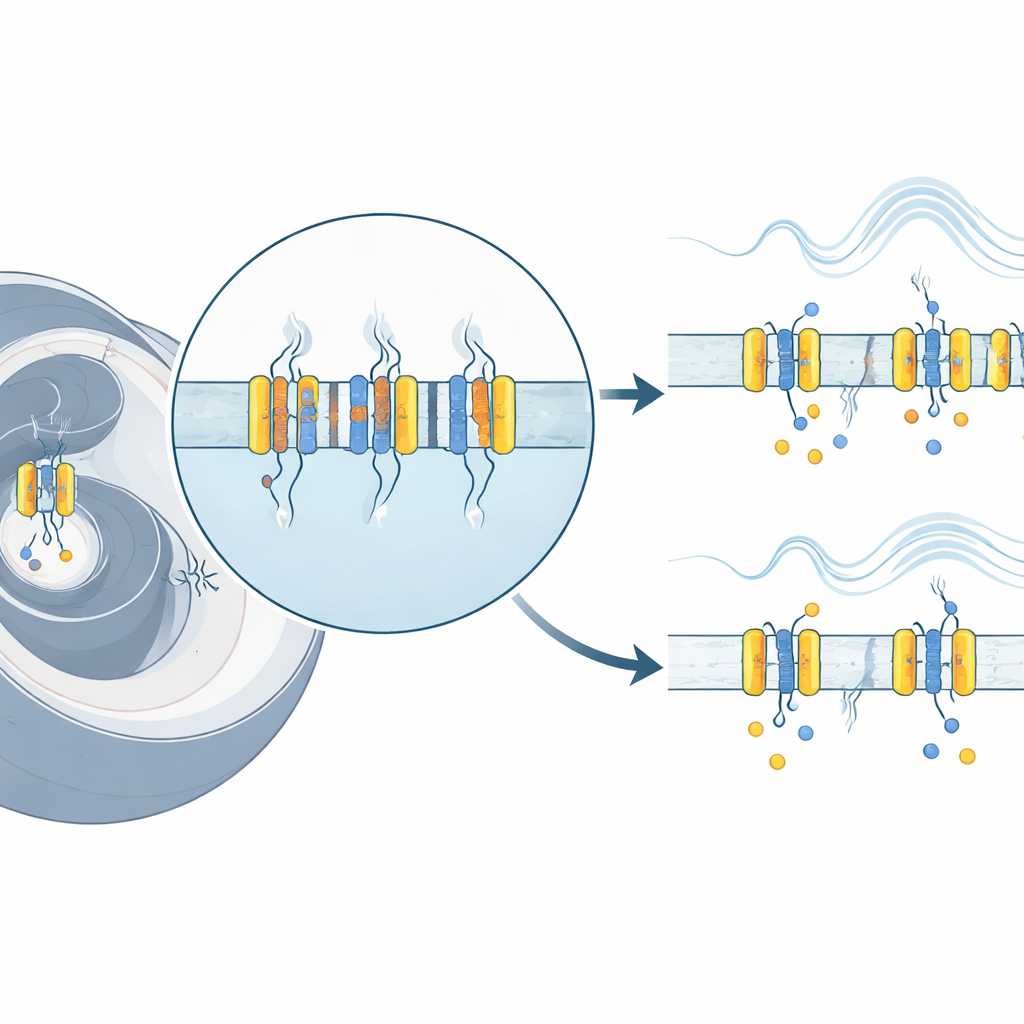
Watching a Molecular Switch in Real Time
To track the movements of the voltage sensor, the team used a technique called voltage-clamp fluorometry, which combines electrical recording with light-based reporting. They engineered a version of Kv7.4 that carries a single extra chemical handle on the outer side of the voltage-sensing region. To this handle they attached fluorescent dyes whose brightness changes when their surroundings shift. By stepping the cell’s voltage to different values and simultaneously measuring both electrical currents and changes in fluorescence, they could follow how the sensor moved as the channel switched between closed and open states. They also introduced a patient-derived mutation, R216H, into this engineered channel to see how it altered these motions.
A Two-Step Switch Behind a Slow-Opening Gate
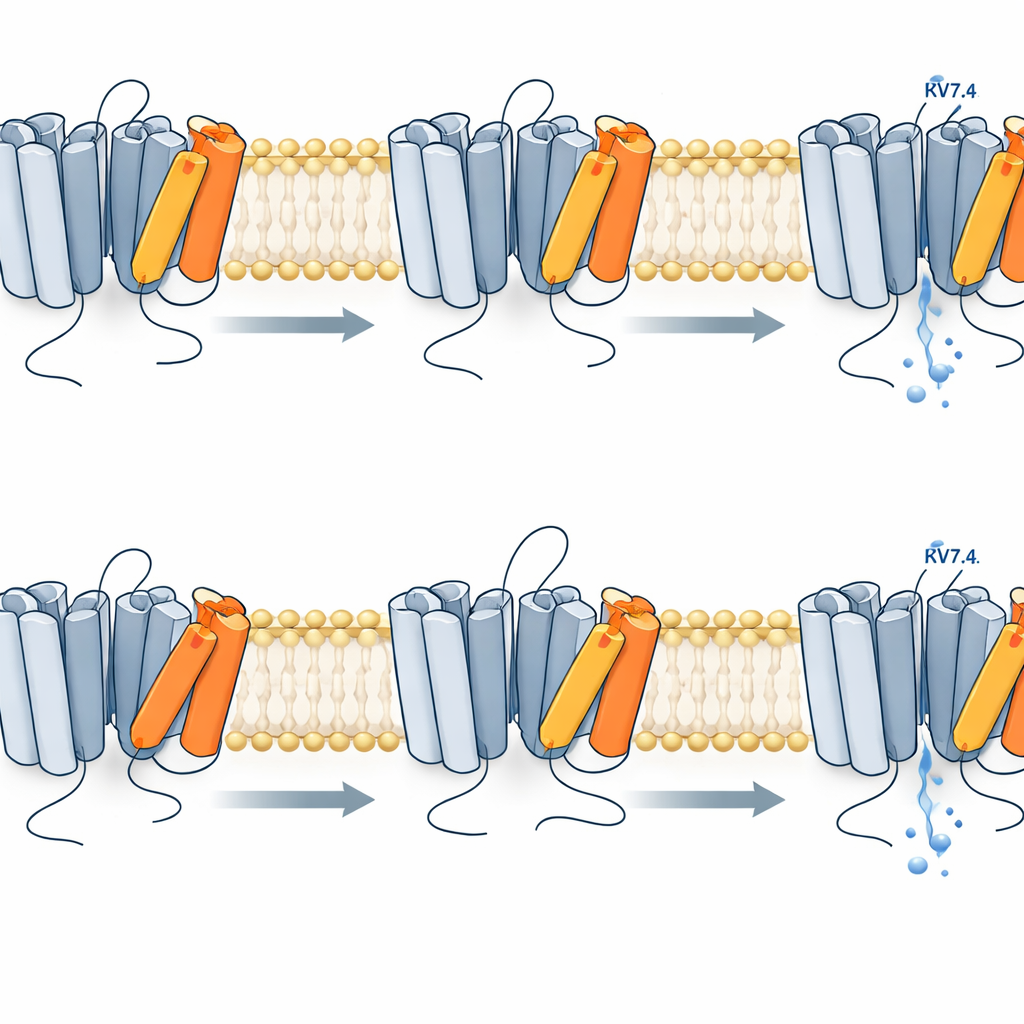
The experiments showed that the voltage sensor in Kv7.4 does not simply jump from “off” to “on.” Instead, it moves in at least two distinct steps. First, at relatively low voltages, the sensor rapidly shifts from a resting to an intermediate position while the pore remains closed. Only with stronger depolarization does the sensor complete a slower second movement into a fully active state, which is tightly linked to the pore opening and the appearance of potassium current. This two-step behavior emerged clearly when the researchers compared the timing and voltage range of the fluorescence signals with the channel’s electrical activity. The first step occurred at more negative voltages and much faster, whereas the second step matched both the voltage range and the sluggish time course of channel opening.
When a Single Substitution Destabilizes the Sensor
The deafness-associated R216H mutation changes one positively charged building block within the voltage-sensing helix. Using the same optical and electrical measurements, the team found that this mutation shifts both sensor steps and pore opening to more positive voltages and reduces their sensitivity to voltage changes. In other words, a stronger electrical push is needed to reach the same level of activation, and the channel is less likely to open. Computer simulations of the channel’s three-dimensional structure supported this view: in the mutant, the crucial helix that carries R216H wobbles more and forms fewer stabilizing interactions with nearby negatively charged residues. This makes the fully activated configuration less stable, so the sensor more readily falls back toward its resting position, and the pore tends to close sooner.
Why These Microscopic Movements Matter
By revealing that Kv7.4 relies on a two-step voltage-sensor movement to open, and showing how a single inherited change can weaken these steps, the study offers a clear mechanistic explanation for one form of progressive hearing loss. In healthy channels, the sensor reliably completes the slow second step that opens the gate and supports the steady potassium flow needed for normal sound amplification in the cochlea and proper blood vessel tone elsewhere in the body. In channels carrying the R216H mutation, this final step is destabilized, so fewer channels open under everyday conditions, leading over time to impaired hearing. Understanding this detailed gating mechanism provides a foundation for designing drugs that could stabilize the active sensor state or boost channel opening, with the long-term aim of protecting or restoring hearing in affected individuals.
Citation: Nappi, M., Frampton, D.J.A., Kusay, A.S. et al. Two-step voltage-sensor activation of the human KV7.4 channel and effect of a deafness-associated mutation. Nat Commun 17, 2381 (2026). https://doi.org/10.1038/s41467-026-69249-8
Keywords: hearing loss, ion channels, Kv7.4, voltage sensor, inner ear