Clear Sky Science · en
Reducing methylation of histone 3.3 lysine 4 in the medial ganglionic eminence and hypothalamus recapitulates neurodevelopmental disorder phenotypes
How Tiny Chemical Tags Shape Brain and Body
Why do some genetic changes cause both learning problems and unusual growth, such as being very small in childhood but obese in adulthood? This study looks at tiny chemical tags on DNA-packaging proteins in the brain and shows how disturbing them in just two key regions can ripple outward into seizures, anxiety-like behavior, memory problems, and dramatic changes in body size in mice.
Switches on the Genome’s Dimmer Knobs
Inside every brain cell, DNA is wrapped around spool-like proteins called histones. Chemical marks placed on these histones act like dimmer knobs, turning groups of genes up or down. One such mark, added to a spot called H3K4, is strongly linked to turning genes on. Human genetic studies have revealed that people born with faults in enzymes that add or remove this mark often have neurodevelopmental disorders that combine intellectual disability, epilepsy, and abnormal body growth. However, it has been unclear which brain cells are most sensitive to this disruption and how their malfunction could tie together brain excitability and whole-body metabolism.
Targeting Two Critical Brain Hubs
The researchers engineered mice in which a mutant version of a histone protein (H3.3K4M) was switched on only in cells arising from two embryonic brain regions: the medial ganglionic eminence, which produces many of the brain’s inhibitory “brake” cells, and the developing hypothalamus, which helps regulate appetite, hormones, and energy balance. This mutation specifically blocks H3K4 methylation without eliminating histones themselves. Tests confirmed that the mutant protein was widely present in the targeted areas and that normal H3K4 marks were strongly reduced there while overall histone levels stayed stable. This design mimics many human conditions where only one copy of an H3K4-related gene is faulty, rather than completely absent.
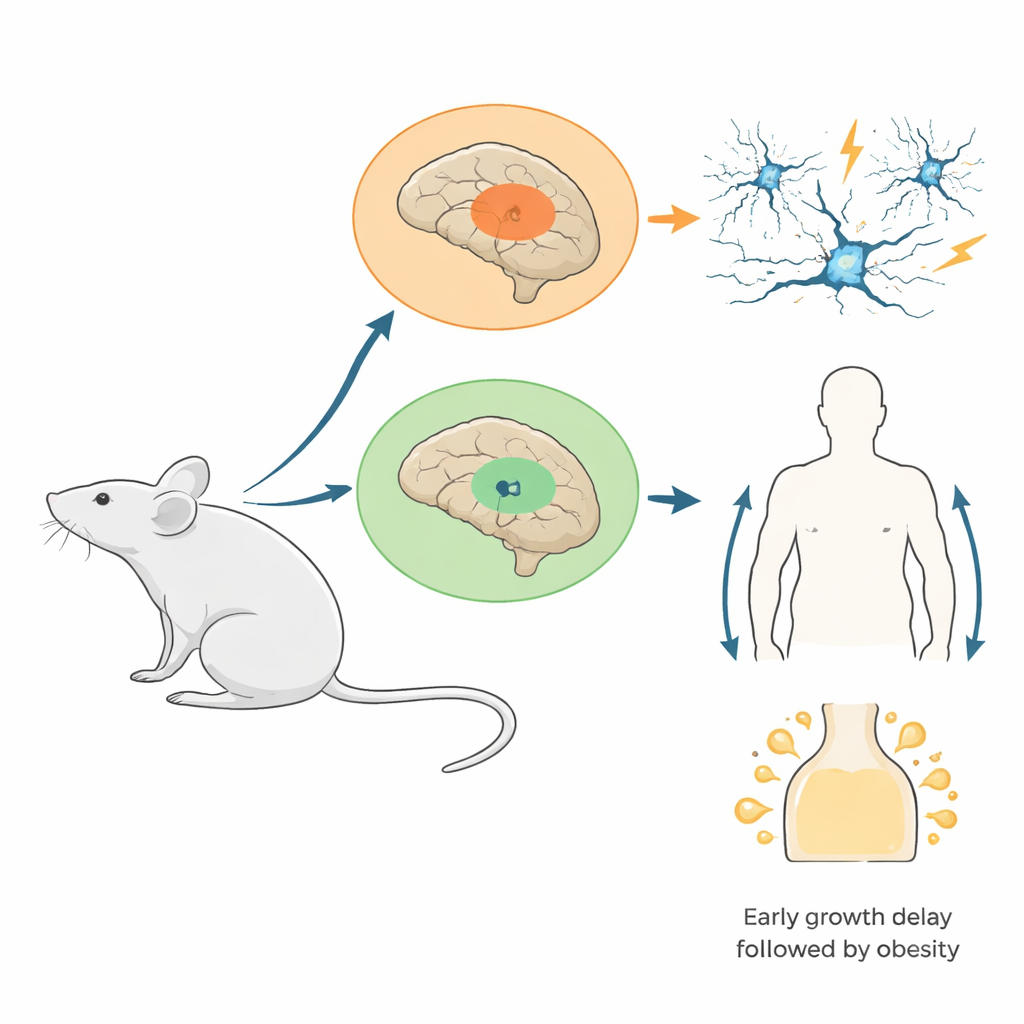
From Missing Brake Cells to Seizure-Prone Circuits
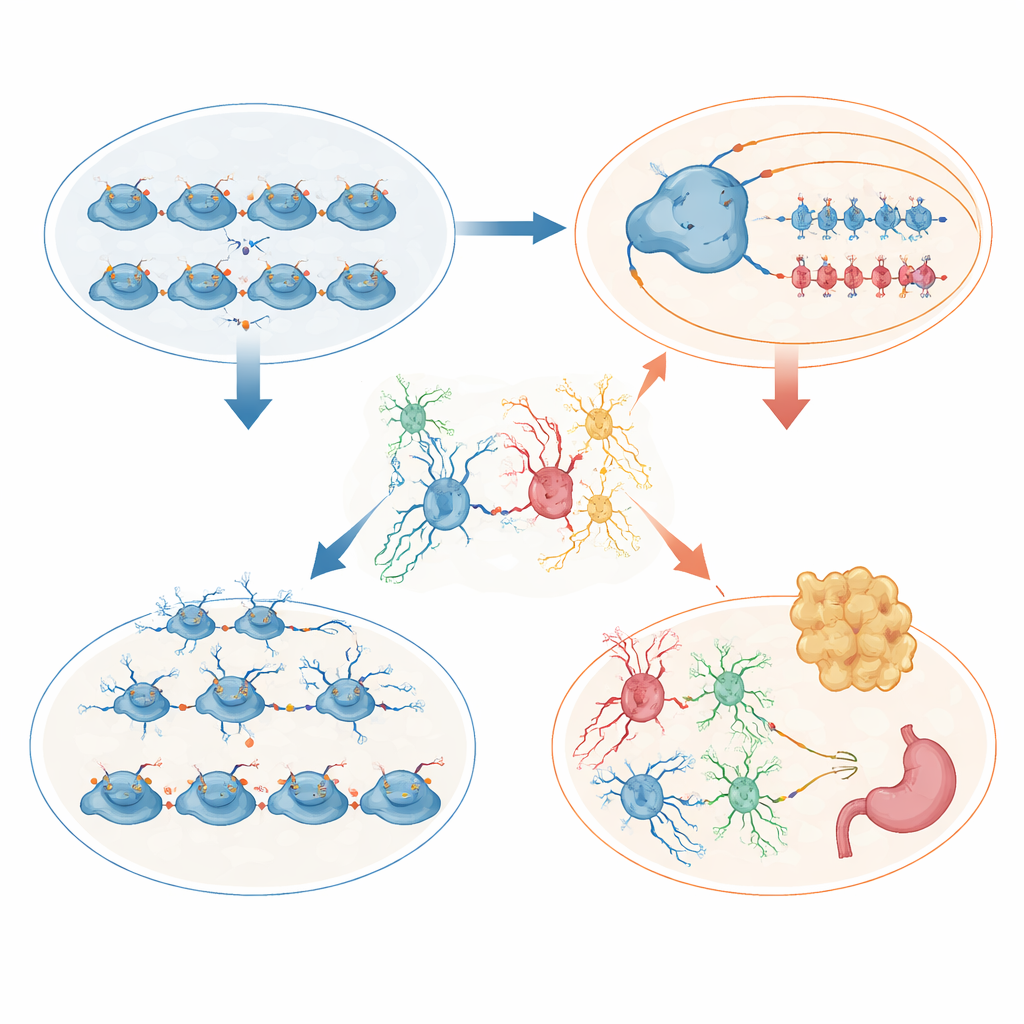
As these mice grew, the consequences for brain circuits became clear. Young mutants had fewer inhibitory interneurons in the cortex and hippocampus, especially a fast-firing type that normally keeps neural rhythms tightly timed. Detailed recordings showed that remaining interneurons were present but more variable in their electrical properties, suggesting that their maturation was disturbed. When the hippocampus was stimulated in brain slices, the usual high-frequency “gamma” oscillations—electrical rhythms linked to information processing—were weaker and slower, and abnormal burst-like events appeared. In living animals, many mutants, particularly females, developed spontaneous seizures and were far more sensitive to drug-induced seizures. Early in development, the team traced this cell loss mainly to poor migration of interneurons into the cortex, not to increased cell death or reduced cell division.
Rewiring Gene Activity in Seizure-Linked Cells
To connect these physical changes to gene control, the team used single-nucleus sequencing that reads both gene activity and DNA accessibility from individual cells. In embryonic medial ganglionic eminence, most genes that changed in mutants were turned down, including key regulators that steer cells toward specific inhibitory fates. In adult interneurons, the balance between subtypes shifted, and groups of genes involved in forming connections and controlling potassium currents—the flows that help set firing speed—were mis-regulated. Network analysis highlighted coordinated disruption of potassium channel genes that are already known to influence epilepsy and brain rhythms, providing a direct molecular link between the loss of a histone mark, altered interneuron identity, and seizure susceptibility.
Hypothalamic Imbalance and a Two-Phase Growth Curve
The hypothalamus told a complementary story centered on body growth. As pups, mutant mice were smaller and a substantial fraction died early. Survivors, however, later ate more, accumulated fat, and developed high levels of the hormone leptin, signaling obesity and probable leptin resistance. Single-cell analysis of embryonic hypothalamus revealed more dividing progenitor cells but fewer cells destined for key feeding-related nuclei, especially regions that normally sense nutritional status and regulate growth hormone. In adult hypothalamus, the cellular mix was reshaped: astrocytes increased sharply, oligodendrocytes diminished, and specialized barrier cells called tanycytes and neighboring glia lost their orderly arrangement at the brain’s interface with circulating hormones and nutrients. These structural and gene-expression changes likely distort how the brain senses energy stores and controls appetite.
Behavioral Echoes of Human Neurodevelopmental Disorders
The mice’s behavior echoed symptoms seen in many neurodevelopmental conditions. They showed stronger anxiety-like behavior, altered gait, reduced spontaneous movement at home, and poor performance on tasks that probe memory, object recognition, and filtering of startling sounds. Some tests revealed increased impulsive-like actions. Across multiple measures, females tended to be more severely affected than males, hinting that sex-specific hormone systems and gene regulation may interact with histone marks to shape vulnerability.
What This Means for Human Health
Together, the findings show that weakening one set of histone marks in just two embryonic brain hubs is enough to reproduce a broad constellation of problems: fewer inhibitory “brakes,” unstable brain networks, seizures, disturbed feeding circuits, and abnormal body growth. For non-specialists, the key message is that epigenetic marks such as H3K4 methylation are not vague add-ons but precise control dials that help developing brain cells become the right type, in the right place, at the right time. When those dials are mis-set, as in many rare genetic syndromes, the result can be a tightly linked combination of cognitive, behavioral, and metabolic symptoms. Understanding these shared roots may ultimately guide therapies that correct not just one symptom, like seizures or obesity, but the interconnected system that gives rise to them.
Citation: Li, J., Tanzillo, A.F., Pizzirusso, G. et al. Reducing methylation of histone 3.3 lysine 4 in the medial ganglionic eminence and hypothalamus recapitulates neurodevelopmental disorder phenotypes. Nat Commun 17, 2984 (2026). https://doi.org/10.1038/s41467-026-69248-9
Keywords: epigenetics, interneurons, hypothalamus, seizures, obesity