Clear Sky Science · en
Decoupling phase separation and fibrillization preserves activity of biomolecular condensates
Why this matters for brain health
Many brain diseases, including Alzheimer’s, involve proteins that clump into hard, fibrous tangles over time. These same proteins can also form softer, droplet-like structures inside cells that help organize chemistry, a bit like tiny, liquid workstations. This study asks a crucial question: can we stop the harmful hardening of these droplets into fibers without destroying their useful, everyday function? The authors show that a common cell metabolite, the amino acid L-arginine, can do exactly that for a key Alzheimer’s-related protein called Tau.
From free-floating protein to tiny droplets
Inside cells, certain proteins do not always stay evenly mixed in the fluid interior. Instead, they can gather into droplets, called biomolecular condensates, that behave like very soft gels or thick liquids. The team focused on Tau, a protein that normally helps build and stabilize microtubules—hollow filaments that act like roads inside nerve cells. Tau is also infamous for forming amyloid fibrils, the rigid fibers found in the tangles that mark many neurodegenerative diseases. To study how Tau droplets evolve over time, the researchers engineered a version of the protein, dubbed SynTag-Tau, which forms active condensates that age into amyloid fibrils on an experimentally convenient time scale, without harsh additives.
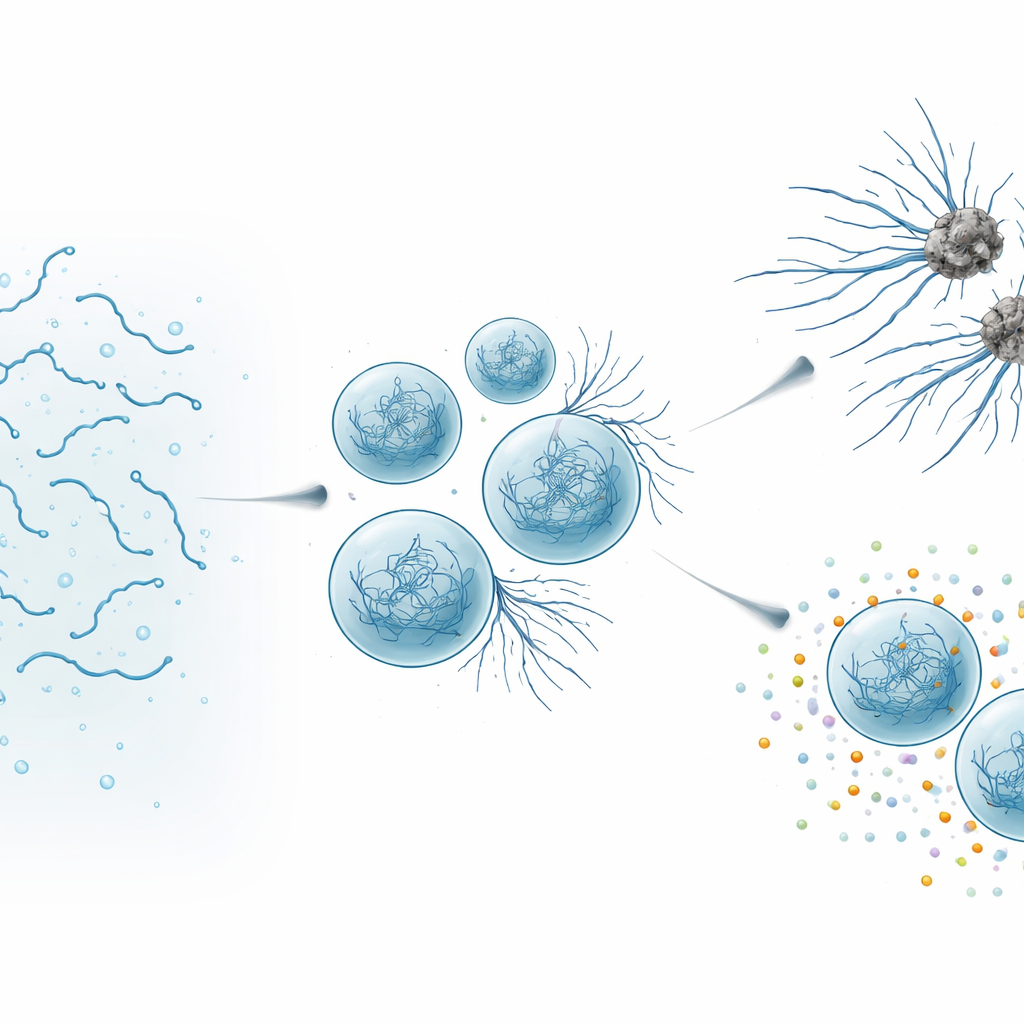
When helpful droplets turn harmful
Using high-resolution microscopy and biophysical measurements, the authors watched SynTag-Tau droplets change over hours. Fresh droplets behaved like liquids: they fused easily, their molecules moved quickly, and there was no sign of ordered structure. Over time, the droplets slowed down and stiffened, and thin fibers began sprouting from their surfaces into the surrounding solution. Sensitive optical methods showed that these fibers contained the tightly packed “cross–beta” architecture typical of amyloid. Importantly, the interface—the boundary between the dense droplet and the surrounding fluid—acted as a hot spot where fibrils first appeared. As droplets aged and sprouted more fibers, Tau’s normal job suffered: the condensates became worse at drawing in tubulin, the building block of microtubules, and eventually lost their ability to support microtubule assembly altogether.
Small molecules that tip the balance
The researchers then asked whether simple metabolites could slow or prevent this liquid-to-fiber transition while leaving the droplets themselves intact. Screening several naturally occurring small molecules, they found that the positively charged amino acids L-arginine and L-lysine strongly delayed or blocked the formation of amyloid fibrils from SynTag-Tau droplets, yet did not shut down droplet formation at realistic, low-millimolar concentrations. In contrast, negatively charged amino acids such as glutamate and aspartate sped up fibril formation, and general protein-disrupting chemicals either failed to help or disturbed the droplets themselves. A fluorescent analog of L-arginine revealed that this molecule prefers to sit inside Tau condensates rather than outside, suggesting it acts where it is most needed.
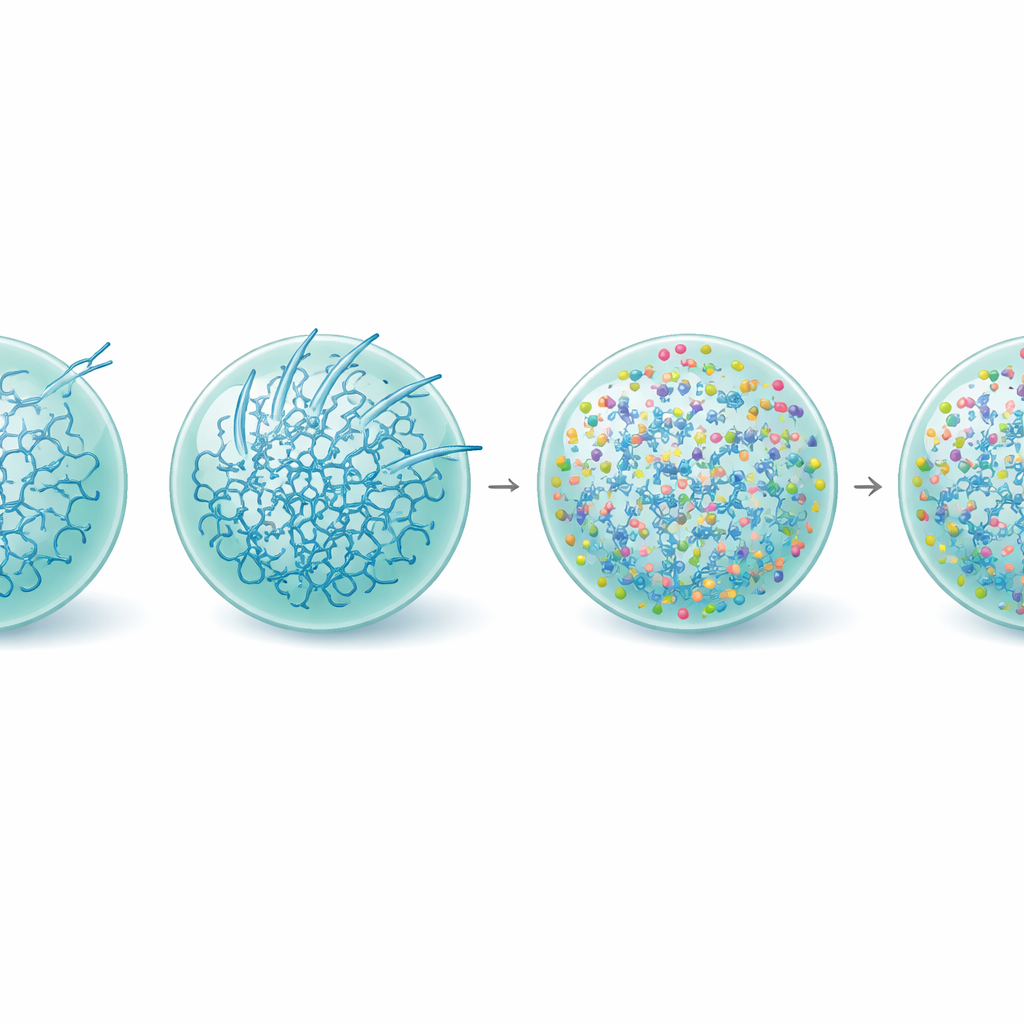
Stiffening the droplet, saving the function
Digging deeper, the team examined how L-arginine reshapes the inner life of Tau droplets. Measurements of protein structure showed that, with L-arginine present, the fraction of Tau molecules adopting the rigid beta-rich forms associated with amyloid was reduced, even in older droplets. Video-based nanorheology—tracking the motion of tiny beads inside condensates—revealed that L-arginine actually made Tau droplets more viscoelastic, meaning their internal network became stronger and more interconnected while remaining fluid-like. This strengthening appears to keep the system in a metastable, functional state and raises the energetic barrier to forming fibrils, particularly at the droplet surface. Consistent with this, L-arginine–treated condensates continued to recruit tubulin evenly and supported microtubule growth long after untreated droplets had become inactive.
What this means for future therapies
The work demonstrates that the forces driving protein droplet formation and those driving harmful fiber formation are related but separable. By tuning the chemistry inside condensates with a metabolite such as L-arginine, it is possible to preserve the beneficial, liquid-like state while delaying or preventing the transition to disease-linked amyloid fibers. Although this study uses a specially engineered Tau system in vitro, it offers a proof of principle: small molecules that selectively fortify condensate stability may one day help protect cells from the gradual buildup of toxic protein aggregates without blocking the normal, organizing roles of biomolecular condensates.
Citation: Mahendran, T.S., Singh, A., Srinivasan, S. et al. Decoupling phase separation and fibrillization preserves activity of biomolecular condensates. Nat Commun 17, 2841 (2026). https://doi.org/10.1038/s41467-026-69244-z
Keywords: Tau protein, biomolecular condensates, amyloid fibrils, L-arginine, neurodegeneration