Clear Sky Science · en
A human electrophysiological signature of Fragile X pathophysiology is shared in V1 of Fmr1-/y mice
Why brain rhythms in Fragile X matter
Fragile X syndrome is a leading inherited cause of intellectual disability and autism. Families and clinicians have long hoped that promising treatments tested in mice would help people, yet many drugs that worked in animal models have failed in clinical trials. This study asks a simple but crucial question: can we find a shared, measurable brain signal in both humans and mice that captures what goes wrong in Fragile X, and that also changes when treatments act on the brain?
Slow “idling” rhythms as a window into the brain
Our brains are never truly quiet. Even at rest, billions of nerve cells produce rhythmic electrical activity that can be picked up by sensors on the scalp as an electroencephalogram (EEG). One of the strongest of these rhythms is called the alpha rhythm, a gentle 8–13 cycle-per-second sway that is especially strong over the back of the head and helps regulate how we process sights and sounds. In many brain conditions, including Fragile X, autism, schizophrenia, and others, this rhythm is altered. Here, researchers focused on how alpha rhythms differ in males with Fragile X compared with typically developing peers, and whether a similar rhythm exists in the visual cortex of Fragile X model mice.
What the researchers saw in people
The team recorded resting-state EEG from boys and men with Fragile X and age-matched controls, using many small electrodes across the scalp. Instead of looking only at standard frequency bands, they separated each person’s signal into a smooth, background “hiss” and distinct peaks that represent true rhythmic oscillations. Across ages, people with Fragile X showed a consistent shift in their main low-frequency peak—linked to alpha—toward slower frequencies. In children, this alpha-like peak was not only slower but also weaker, while in adults it was clearly slower but roughly equal in strength to controls. The strongest source of this altered rhythm was traced to the visual areas at the back of the brain, suggesting a good target for comparison with animal models.
Matching signatures in mouse visual cortex
Using the same style of analysis, the researchers then measured brain activity from the visual cortex of male Fragile X mice and their healthy littermates. Mice do not have a classic human alpha band, but they do show a prominent rhythm in the 3–6 cycles-per-second range in visual cortex when quietly viewing a gray screen or sitting in the dark. In Fragile X mice this rhythm was slowed, mirroring what was seen in humans. When tiny electrodes were placed directly into the visual cortex, an even richer picture emerged: the main low-frequency rhythm actually contained two sub-peaks. The lower one showed the slowing seen in Fragile X, while changes in the higher one appeared only in juveniles, echoing the child-specific power changes in humans.
Cells and chemistry behind the rhythm
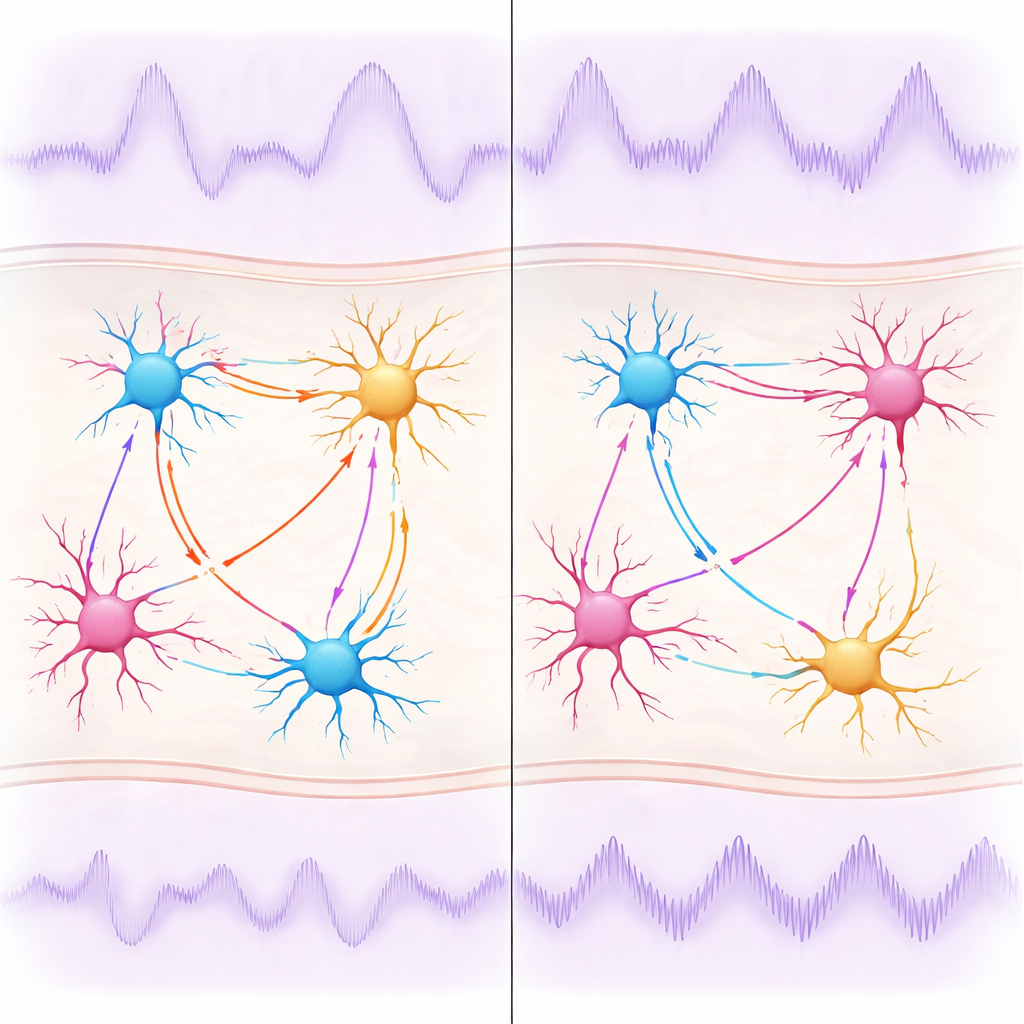
Because experiments in mice can probe cell types and drugs directly, the team next asked which neurons and chemical signals shape these alpha-like rhythms. They found that removing the Fragile X protein specifically from cortical excitatory neurons and nearby support cells was enough to recreate the abnormal rhythms, even when inhibitory cells were left genetically intact. By selectively dampening two major classes of inhibitory cells—parvalbumin-positive and somatostatin-positive interneurons—they showed that each class influences a different sub-peak of the rhythm. They then tested a drug, Arbaclofen, which boosts a particular type of inhibitory signal (GABAB receptors). In typical mice, this drug made the low-frequency rhythm stronger and slightly slower, confirming that the oscillation is sensitive to pharmacological intervention. In Fragile X mice, however, the same doses had a weaker effect on the rhythm, even though they still reduced excessive higher-frequency activity.
Bridging lab mice and human treatment
For non-specialists, the key message is that this study identifies a specific, shared brain rhythm abnormality in people with Fragile X and in a mouse model, and shows that this signal can be shifted by a drug in ways that depend on both age and genotype. The slowed alpha-like rhythm offers a practical “thermometer” of brain function that can be measured the same way in humans and mice, and that may better track how well treatments are working than previously used fast brain waves. By tying this rhythm to particular cell types and inhibitory pathways, the work also points toward more focused strategies for future therapies, while helping explain why some earlier drug trials may not have translated from mouse to human.
Citation: Kornfeld-Sylla, S.S., Gelegen, C., Norris, J.E. et al. A human electrophysiological signature of Fragile X pathophysiology is shared in V1 of Fmr1-/y mice. Nat Commun 17, 1497 (2026). https://doi.org/10.1038/s41467-026-69243-0
Keywords: Fragile X syndrome, brain rhythms, alpha oscillations, visual cortex, GABA inhibition