Clear Sky Science · en
Epilepsy-associated FOXJ3 variants link a transcriptional program of the PTEN-mTOR pathway to neuronal specification and cortical lamination
When Brain Wiring Goes Slightly Off Course
Epilepsy often begins in childhood, but in many young patients doctors cannot see anything obviously wrong on brain scans. This study tackles one hidden cause: tiny changes in a gene called FOXJ3 that quietly reshape how the brain’s outer layer, the cortex, is built. By following this gene’s actions in families with focal epilepsy and in developing mouse brains, the researchers show how early missteps in cell birth, movement, and layering can eventually give rise to seizures.
A Subtle Malformation Behind Tough-to-Treat Seizures
Many children with drug-resistant focal epilepsy harbor a condition called focal cortical dysplasia (FCD), in which patches of cortex are malformed. These regions contain misplaced and abnormally large nerve cells and are a common reason why medicines fail to control seizures. Yet the molecular cause of FCD is often unknown, and standard MRI can miss small or shallow defects. The authors began with a family in which several members had focal epilepsy and evidence of FCD. Careful genome analysis pointed to rare changes in FOXJ3, a gene not previously linked to epilepsy. Additional cases from large genetic databases revealed more individuals with unusual FOXJ3 variants and focal seizures, suggesting that this gene may be a recurring player in such disorders.
How FOXJ3 Helps Shape the Thinking Surface of the Brain
During fetal development, the cortex is assembled like a many-story building. Stem-like cells lining the brain’s inner surface divide, then send newborn neurons outward along radial “scaffolds” to form six ordered layers. The team found that FOXJ3 is active both in these stem-like progenitors and in young neurons, but its activity in progenitors naturally drops after a specific time point in mid-gestation. Using gene-silencing tools in mouse embryos, the researchers reduced Foxj3 levels at different stages and watched what happened. Early loss of Foxj3 caused newborn neurons to migrate more slowly and settle in the wrong layers. Cells that should have become deep-layer neurons instead adopted identities typical of upper layers, and callosal projection neurons, which link the two brain hemispheres, were produced in excess. Later in development, however, reducing Foxj3 had far milder effects, showing that its role is highly time-sensitive.
Keeping Cell Division and Growth in Balance
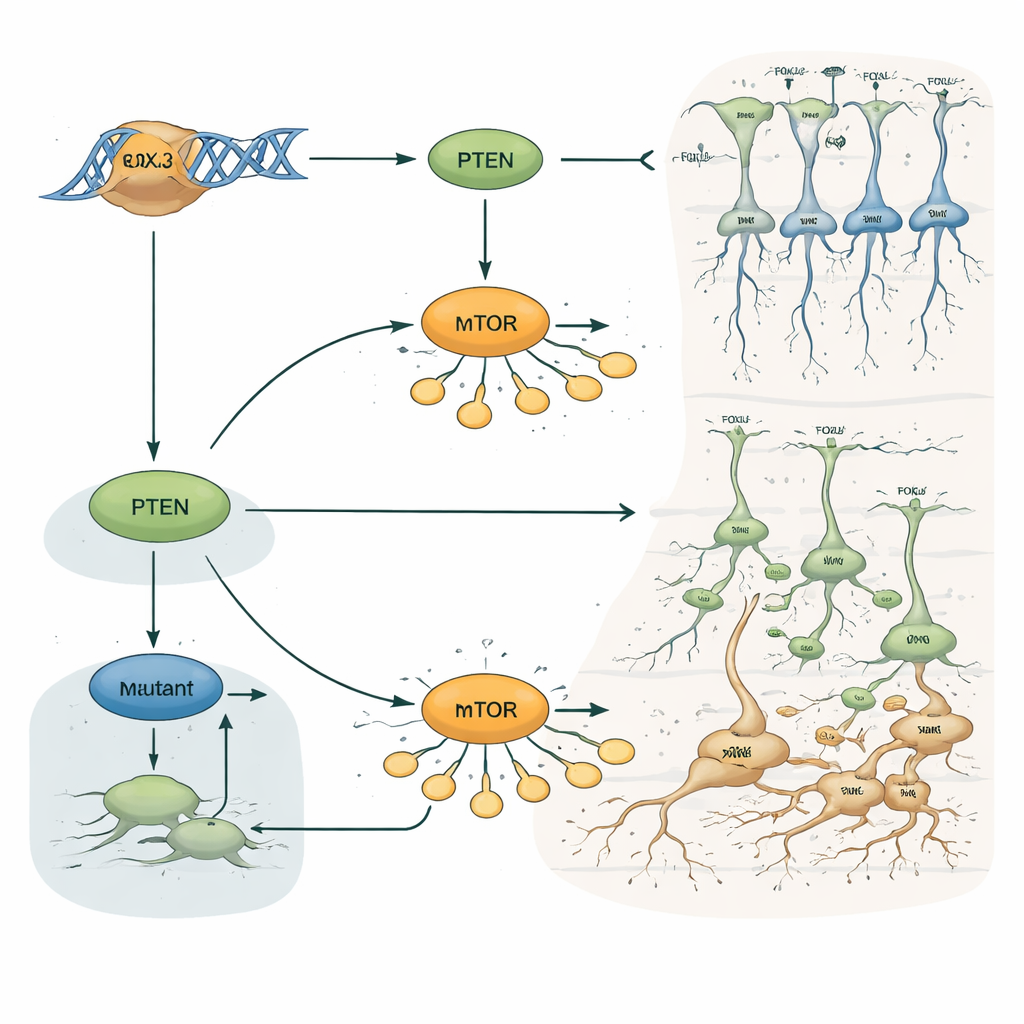
To understand why FOXJ3 has such influence, the researchers combined several large-scale approaches. They mapped where FOXJ3 binds on DNA and which genes are active in thousands of individual cortical cells. This pointed strongly to genes that govern the cell cycle and brain growth, especially PTEN, a major brake on a growth pathway known as mTOR. In progenitor cells, FOXJ3 binding boosted PTEN activity, which in turn helped cells exit the cycle of division, transform into neurons, and migrate outward in an orderly fashion. When Foxj3 was knocked down, more progenitors stayed in a dividing state, fewer exited the cycle on time, and migration stalled in intermediate zones. Crucially, restoring PTEN levels in Foxj3-deficient brains largely reversed the migration, layering, and cell-fate problems, whereas boosting another growth regulator, TSC1, did not. This places PTEN at the heart of FOXJ3’s control over cortical construction.
From Misregulated Pathways to Enlarged Neurons
The team next asked how patient-like FOXJ3 variants behave. A disease-associated version of FOXJ3 failed to turn up PTEN levels and allowed the mTOR pathway to become overactive, as seen by increased phosphorylation of a key growth-related protein. In adolescent mice engineered to express this mutant form in developing cortex, neurons later appeared noticeably swollen, with larger cell bodies and perimeters, mimicking the dysmorphic cells seen in human FCD. Yet the mutant protein still reached the nucleus correctly, indicating that the problem lies in faulty gene regulation, not mislocalization. Together, these findings tie FOXJ3 mutations to a cascade of events—less PTEN, more mTOR signaling, delayed cell-cycle exit, scrambled migration, and hypertrophic neurons—that can set the stage for epileptic networks.
Why This Matters for Epilepsy and Hidden Brain Lesions
For a non-specialist, the key message is that a single transcription factor, FOXJ3, helps decide when and where cortical neurons are born, how they move, and which layer they ultimately occupy. By acting through the PTEN–mTOR growth pathway, FOXJ3 keeps brain building on schedule. Rare changes in this gene can tilt that balance, causing small patches of cortex to be wired and layered incorrectly, even when MRI scans appear normal. This work not only proposes FOXJ3 as a new genetic cause of focal cortical dysplasia and epilepsy, but also illustrates how subtle shifts in early brain development can echo decades later as hard-to-treat seizures.
Citation: Cheng, HY., Liu, C., Nien, CW. et al. Epilepsy-associated FOXJ3 variants link a transcriptional program of the PTEN-mTOR pathway to neuronal specification and cortical lamination. Nat Commun 17, 1815 (2026). https://doi.org/10.1038/s41467-026-69241-2
Keywords: focal cortical dysplasia, FOHJ3 gene, PTEN mTOR pathway, cortical development, focal epilepsy