Clear Sky Science · en
Convergent extreme reductive evolution in ancient planthopper symbioses
Tiny partners with a big impact
Plant-sap sucking insects called planthoppers depend on invisible bacterial partners to survive on their sugary, nutrient-poor diet. This study explores how some of those bacteria have shrunk their DNA to the smallest known bacterial genomes, becoming so dependent on their insect hosts that they are edging toward the status of cellular “organelles” like mitochondria. For readers interested in how life can be pared down to its bare essentials, this work offers a window into the minimum toolkit needed for a cell to keep going inside another organism.
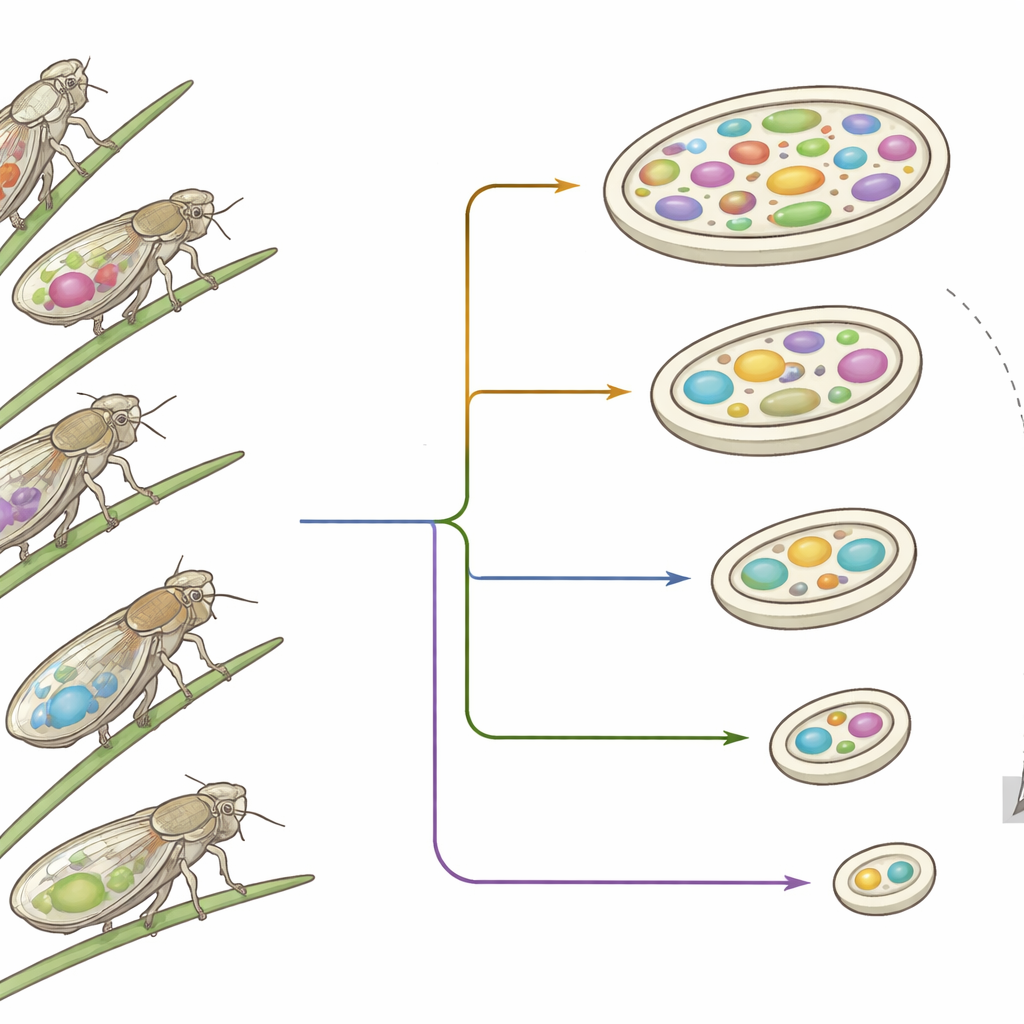
Hidden allies in sap-feeding insects
Planthoppers are an ancient group of sap-feeding insects that arose about 263 million years ago. Because plant sap lacks many vital nutrients, these insects rely on internal bacteria, passed from mother to offspring, to manufacture missing amino acids and vitamins. Two such long-term partners, known as Sulcia and Vidania, occupy specialized cells in the insect body and have co-diversified with their hosts over hundreds of millions of years. Earlier work on similar insect–bacteria partnerships showed that these symbionts tend to streamline their genomes, keeping mostly genes needed for nutrient production and basic cellular housekeeping.
How small can a genome get?
Using metagenomic sequencing from 149 planthopper species, the authors reconstructed 131 complete genomes of Sulcia and Vidania. Most Sulcia genomes clustered in a relatively narrow size range, whereas Vidania varied far more widely. Two Vidania strains, from different planthopper superfamilies, stood out as record-breakers: their genomes were only about 50–52 thousand DNA letters long and carried just over 60 recognizable protein-coding genes. That is smaller than any previously reported bacterial genome outside of organelles such as mitochondria and chloroplasts. Despite evolving separately for roughly a quarter of a billion years, these two ultra-small genomes turned out to be strikingly similar in structure and content.
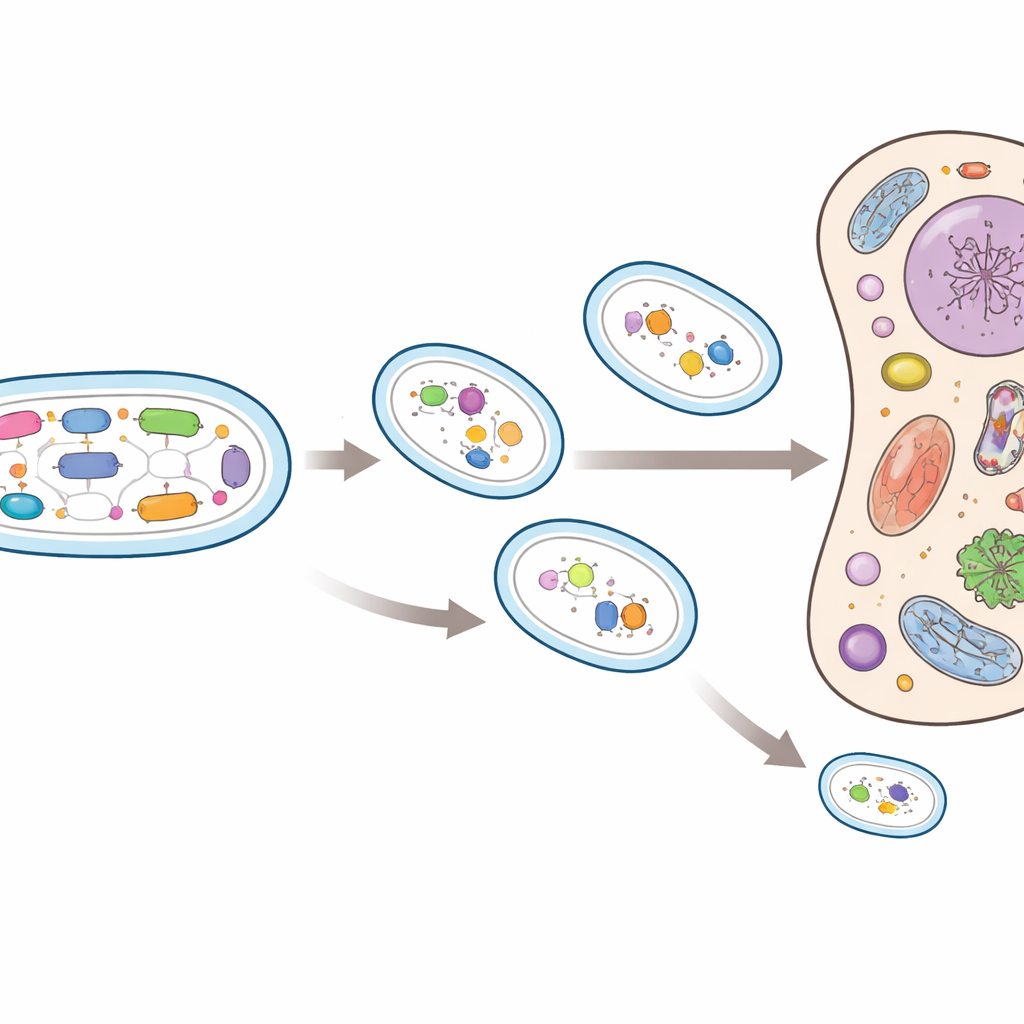
Parallel paths to extreme simplification
By comparing gene content across the planthopper family tree, the researchers reconstructed the larger ancestral gene sets of Sulcia and Vidania and traced which genes were lost in each lineage. In most hosts, gene loss in both bacteria was gradual, mainly affecting metabolic and information-processing functions. In a few lineages, however, Vidania underwent dramatic erosion, shedding dozens of genes in single evolutionary branches. The most reduced Vidania strains retained only a handful of genes for DNA and RNA handling, ribosome components, and one complete pathway to make a single essential amino acid, phenylalanine. Every other amino acid pathway was gone. Notably, these extreme cases evolved independently in different planthopper groups yet converged on nearly the same tiny set of remaining genes, suggesting that there may be a common endpoint to this kind of reductive evolution.
When other partners pick up the slack
The insects hosting the tiniest Vidania genomes had all lost Sulcia entirely, breaking a symbiosis that had persisted since early in planthopper history. In many of these species, other bacteria or fungi now live alongside Vidania and appear to provide some of the nutrient-making functions once handled by the ancestral pair. Some planthopper groups have also shifted their lifestyles in ways that change their nutrient needs or access: certain larvae feed on fungal threads instead of plant sap, some species maintain close food-sharing partnerships with ants, and one species lives in caves. These ecological shifts, together with the arrival of new microbial partners, likely relaxed the pressure to maintain full nutrient factories inside Vidania, allowing further gene losses that would otherwise have been lethal.
Blurring the line between bacteria and organelles
As Vidania sheds more and more of its own cellular toolkit, it must rely increasingly on proteins and processes supplied by the insect host, much like mitochondria do in our own cells. The smallest Vidania genomes still make phenylalanine, a building block important for hardening the insect’s outer shell, but little else nutritionally. The study suggests that such symbionts can be whittled down to a few dozen genes and a single key role, while the host and other microbes cover everything else. This pushes our understanding of how far evolution can strip down a living cell and shows how long-term partnerships can trap both sides in an “evolutionary rabbit hole” where escape may require replacing old partners, radically changing lifestyles, or facing eventual collapse of the alliance.
Citation: Michalik, A., Franco, D.C., Deng, J. et al. Convergent extreme reductive evolution in ancient planthopper symbioses. Nat Commun 17, 2473 (2026). https://doi.org/10.1038/s41467-026-69238-x
Keywords: endosymbiosis, genome reduction, planthoppers, insect microbiome, bacterial evolution