Clear Sky Science · en
Enhanced sodium storage in hard carbon via solvent co-intercalation electrolyte enabling Ah-level pouch cells at low temperatures
Why cold-proof batteries matter
From electric cars in snowy climates to sensors in the deep Arctic, many modern devices need batteries that keep working in the cold. Today’s lithium and sodium batteries often lose power or fail completely at very low temperatures because their internal chemistry slows down. This study explores a new way to build sodium-ion batteries—using a specially designed liquid inside the battery—so they can store and deliver energy reliably even at temperatures as low as −50 °C.
The challenge of freezing batteries
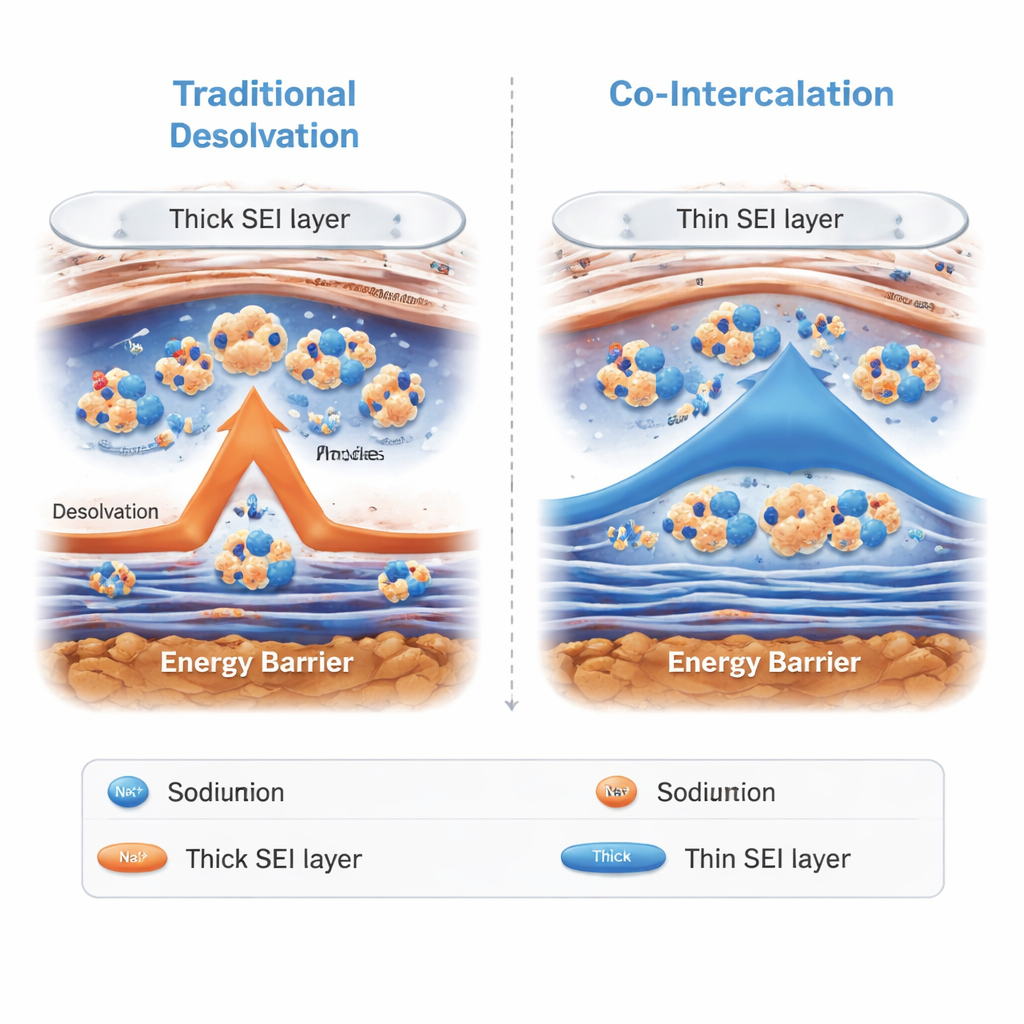
Batteries store energy by shuttling charged atoms, called ions, between two solid electrodes through a liquid electrolyte. In sodium-ion batteries, sodium ions must squeeze through a thin surface film and into a carbon-based negative electrode known as hard carbon. At low temperatures, two things go wrong: ions move more slowly through the liquid, and they struggle to shed the solvent molecules that surround them before entering the hard carbon. At the same time, the protective surface film—called the solid electrolyte interphase—tends to get thicker and more resistive in the cold. All of this makes it harder for sodium ions to move, so the battery can deliver far less energy when it is needed most.
A new liquid mix for easier ion travel
The researchers tackled this problem by redesigning the electrolyte so that sodium ions no longer have to fully strip off their solvent shell before entering the hard carbon. They mixed two ether-based solvents: diethylene glycol dimethyl ether (G2), which clings tightly to sodium ions and supports fast ion motion, and 2-methyloxolane (MO), a less polar liquid that stays fluid at very low temperatures. In the resulting “co-intercalation electrolyte,” sodium ions coordinate mainly with G2, while MO largely acts as a free, non-binding solvent that helps keep the mixture liquid down to −50 °C. Computer simulations and spectroscopy measurements showed that this mixture forms a stable structure in which sodium ions and G2 travel together as a small cluster.
Letting ions enter carbon without undressing
Instead of forcing sodium ions to shed their solvent shell at the electrode surface, the new electrolyte allows the sodium–G2 clusters to slip directly through the surface film and into the layered spaces within the hard carbon. This process, called solvent co-intercalation, bypasses the slow “undressing” step that usually limits performance in the cold. Microscopic and spectroscopic tests revealed that the surface film formed with this electrolyte is thinner and richer in inorganic compounds than in conventional systems. That combination protects the electrode while still allowing ions to cross quickly. Measurements of ion diffusion and electrical resistance confirmed that ions move faster inside the carbon and across the interface, especially at low temperatures.
Strong performance even at −50 °C
When the team tested coin-sized cells using hard carbon and the new electrolyte, they found that the batteries retained high capacity and efficiency from room temperature down to −50 °C. At −50 °C, the hard carbon electrode still delivered about 80% of its initial charge efficiency and maintained over 90% of its capacity after 200 charge–discharge cycles. Going beyond small cells, the researchers built pouch cells—flat batteries similar to those in consumer electronics—with a capacity of about 1.2 ampere-hours. These full sodium-ion batteries achieved a specific energy of 163 watt-hours per kilogram at room temperature and 107 watt-hours per kilogram at −50 °C, while continuing to power LED lights for more than 10 hours in a −50 °C chamber.
What this means for future cold-climate batteries
For non-specialists, the key message is that the authors found a way to let sodium ions keep their helpful solvent coat as they enter the battery’s carbon electrode. By crafting an electrolyte that stays liquid in the cold and forms a thin, ion-friendly surface film, they removed a major bottleneck in low-temperature performance. This approach could help make sodium-ion batteries—a cheaper alternative to lithium-ion—more practical for use in winter climates, high-altitude regions, and other harsh environments where dependable, affordable energy storage is urgently needed.
Citation: Li, M., Liu, Z., Zhao, Y. et al. Enhanced sodium storage in hard carbon via solvent co-intercalation electrolyte enabling Ah-level pouch cells at low temperatures. Nat Commun 17, 1478 (2026). https://doi.org/10.1038/s41467-026-69237-y
Keywords: sodium-ion batteries, low-temperature batteries, electrolyte design, hard carbon anodes, energy storage