Clear Sky Science · en
Molecular basis of collagen galactosylation by GLT25D1
How tiny sugar tags help build strong tissues
Collagen is the body’s most common protein, forming the scaffolding for skin, bones, blood vessels and many other tissues. But collagen doesn’t work alone: it must be decorated with small sugar molecules at precise spots to gain the right strength and flexibility. This study reveals, in atomic detail, how one key enzyme, GLT25D1, attaches a particular sugar to collagen and how faults in this process can lead to fragile vessels, muscle problems and perhaps even cancer.
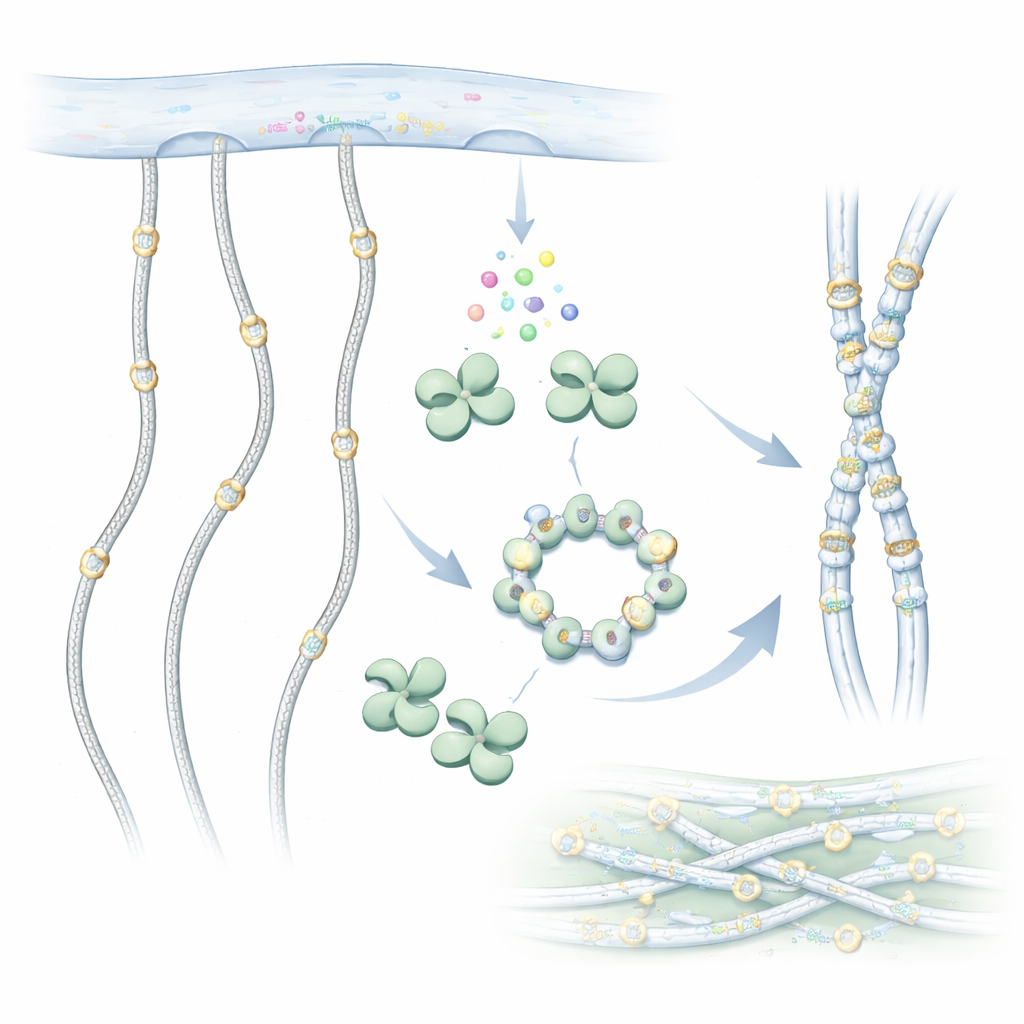
A closer look at collagen’s hidden makeover
Collagen molecules are long, rope-like chains that assemble into sturdy fibers outside cells. Before they leave the cell, however, they are chemically edited. One important edit is the addition of sugar groups to special building blocks called hydroxylysines along the collagen chain. GLT25D1 carries out the first step in this sugar tagging, transferring a galactose sugar from a donor molecule onto hydroxylysine. This sugar decoration is conserved from simple animals to humans and helps collagen fold correctly, interact with cells and resist wear and tear.
Revealing the shape of a collagen decorator
To understand how GLT25D1 works, the researchers used cryo–electron microscopy to image the human enzyme at near‑atomic resolution. They discovered that each GLT25D1 molecule has two similar lobes, both related to a common enzyme fold seen in sugar‑transfer proteins. These lobes pair up to form elongated dimers, and three dimers can further assemble into a hexamer shaped like a ring. In these assemblies, the true workhorses—the catalytic centers—sit far apart, a layout that may allow several sugar‑adding sites to operate along a stretched collagen chain at once.
The working parts: where sugar transfer really happens
The team solved structures of GLT25D1 bound to both its sugar donor and a short collagen‑like peptide containing hydroxylysine. This ternary complex showed that only the second lobe, called the C‑terminal domain, actually carries out the chemistry. There, the donor molecule nestles into a pocket stabilized by a metal ion, while the peptide lies in a narrow groove that enforces a specific local pattern: a hydroxylysine followed immediately by a tiny glycine. A single aspartate residue acts as a chemical base, activating the hydroxylysine’s hydroxyl group so it can attack the sugar and complete the transfer. Changes to any of these key amino acids drastically reduce or abolish activity, confirming their essential roles.
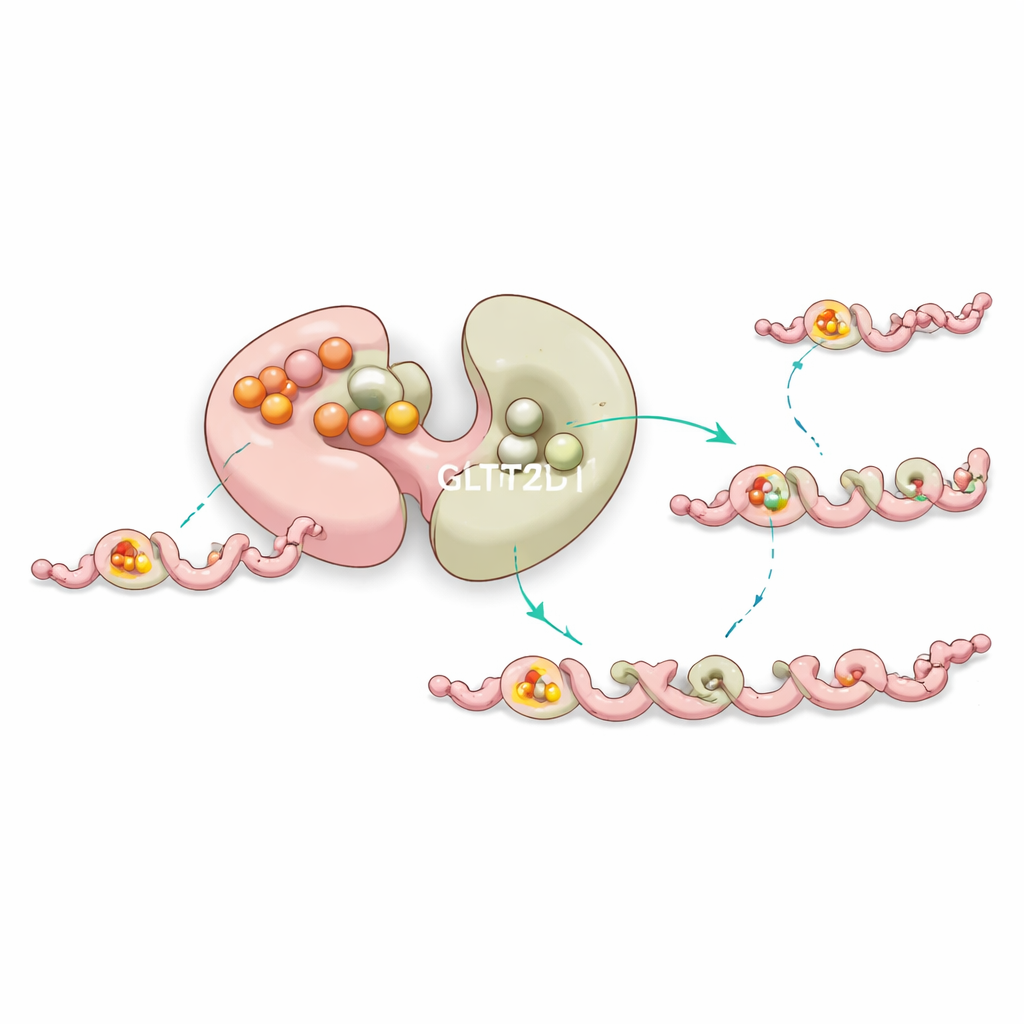
A built‑in stabilizer and long‑distance control
Curiously, the first lobe of GLT25D1 binds a sugar donor very tightly but does not perform the transfer. Instead, experiments and computer simulations suggest that this “silent” site helps stabilize the enzyme and subtly tunes the behavior of the active lobe through long‑range communication within the protein. Mutations near this non‑catalytic pocket often destabilize the enzyme or shift its efficiency, hinting that nature uses this extra binding site as a form of internal quality control to keep collagen modification running smoothly.
When the decorator fails: links to disease
By mapping patient‑derived mutations onto their structural model, the authors could explain how errors in GLT25D1 lead to human disease. Some mutations chop off the catalytic lobe entirely, others destabilize the protein core, and still others strike right at the sugar‑binding or collagen‑binding sites. These faults reduce or eliminate sugar addition to collagen and have been linked to small‑vessel brain disease, cognitive problems and muscle and skeletal defects. Cancer‑associated mutations also cluster in crucial regions, suggesting that altered collagen decoration may influence tumor growth and spread.
Why this matters for health and future treatments
By laying out a detailed three‑dimensional blueprint of GLT25D1 in action, this work explains how precise sugar tags are added to collagen and why this step is so important for tissue integrity. For non‑specialists, the key message is that tiny chemical changes on collagen can have outsized effects on blood vessels, bones and possibly cancer risk. The structural map of GLT25D1 now offers a guide for diagnosing harmful variants and for designing therapies—whether small molecules that stabilize faulty enzymes or gene‑based strategies—that could one day correct collagen sugar‑tagging defects at their source.
Citation: Sun, H., Zhang, M., Shi, Y. et al. Molecular basis of collagen galactosylation by GLT25D1. Nat Commun 17, 2426 (2026). https://doi.org/10.1038/s41467-026-69234-1
Keywords: collagen glycosylation, GLT25D1, extracellular matrix, vascular disease, cryo-EM structure