Clear Sky Science · en
Realistic atomic model for charge storage and charging dynamics of amorphous porous carbons
Why Tiny Cavities in Carbon Matter for Your Devices
From phones to electric cars, many modern gadgets rely on devices called supercapacitors to deliver bursts of power quickly and reliably. These devices often use a special form of carbon full of nanoscale holes, or pores, to store electrical charge. But because this carbon is disordered and its pores twist and branch in complex ways, scientists have struggled to picture exactly what happens inside. This study builds a realistic, atom-by-atom model of such porous carbon and shows how its tiniest pores play an outsized role in storing and moving charge.
Building a Digital Twin of Spongy Carbon
Real porous carbons look less like neatly drilled tunnels and more like tangled caves. Previous computer models simplified them into ideal slits or tubes, which miss much of this complexity. The authors combined several experimental techniques—X-ray scattering, gas adsorption measurements, and bulk density data—to reconstruct the real three-dimensional architecture of a commercial porous carbon. They first used small-angle X-ray scattering to infer how solid carbon and empty space are arranged at nanometer scales, then refined this information with a new statistical method that can capture multiple characteristic pore sizes seen in the data. This yielded a three-dimensional “matrix” describing where pore walls and voids should be located.
To turn this matrix into an atomically realistic solid, the team developed a hybrid reverse molecular dynamics scheme. They embedded individual carbon atoms into the 3D matrix and let them shuffle and rebond under realistic interatomic forces, while gently steering the structure so that the overall pore network remained faithful to the experimental template. The resulting digital carbon matched real samples in key properties: its surface area, how gases fill its pores, and even simulated electron microscope images all lined up closely with measurements. This agreement suggests that the model is not just a cartoon but a credible digital twin of an actual porous electrode.
How Ions Arrange Themselves to Store Charge
With this realistic carbon in hand, the researchers filled the pores in simulation with an ionic liquid—an electrically conducting salt that is molten at room temperature—and applied voltage, mimicking a working supercapacitor. They tracked how many positively and negatively charged ions entered or left the pores and how much charge accumulated on nearby carbon atoms. The predicted capacitance, a measure of how much charge can be stored per unit of voltage and mass, closely matched laboratory measurements using the same material and liquid. This success indicates that the model captures the crucial physics of how ions crowd into and out of nanopores when a device is charged.
A key insight emerged when the authors classified pores by their effective size using a geometric construction called Voronoi spheres. Pores narrower than about 0.7 nanometers—ultramicropores—behave very differently from slightly larger micropores. In ultramicropores, charging proceeds mainly by ion exchange: counter-ions rush in while like-charged ions are expelled, strongly changing the number and arrangement of ions. This process leads to larger induced charge on the carbon walls and thus higher local capacitance. In contrast, larger micropores act more like reservoirs: the total ion count hardly changes with voltage, and ions simply shift between the pore center and walls, storing less charge per surface area.
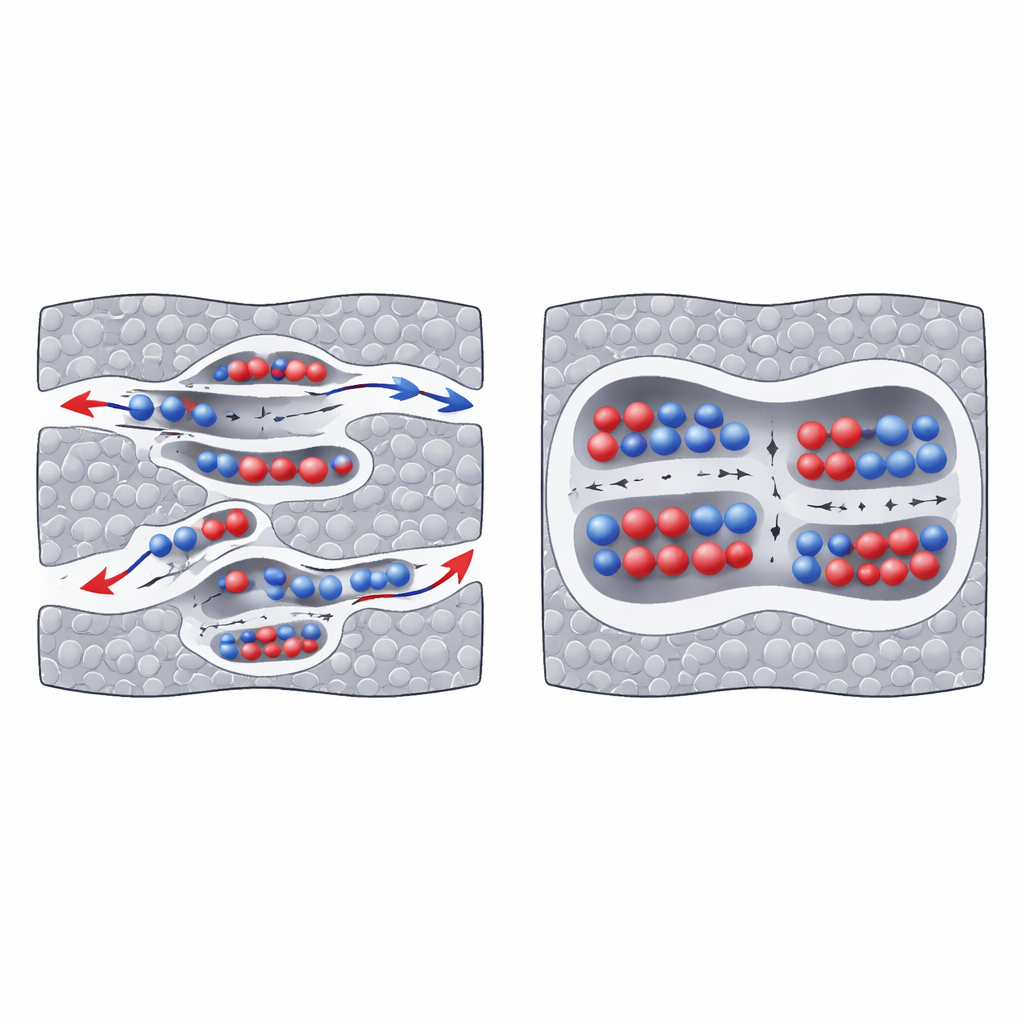
Why Some Tiny Pores Work Better Than Others
The story is not just about pore size but also about how pores connect. The authors distinguish “deep” ultramicropores, which are buried and connected to larger pores mainly at one end, from “facial” ultramicropores that open more directly onto bigger cavities. Deep ultramicropores show stronger ion exchange and higher induced charge than facial ones, especially in positively charged electrodes. Inside these deep regions, ion pairs are more effectively pulled apart, which enhances screening and charge storage but also slows down ion motion. Using a specially designed “fractal” electrical circuit model, the team extracted effective capacitances, conductivities, and charging times for each pore family. They found that ultramicropores dominate charge storage but charge much more slowly than their larger counterparts.
From Atoms to Full Devices
To link microscopic behavior to macroscopic performance, the researchers scaled their pore-level circuit up to represent whole carbon particles and then an entire electrode film, as used in real devices. This multi-scale impedance model reproduced experimental plots of how the device resists alternating currents over a range of frequencies—a stringent test of realism. The agreement indicates that atomistic simulations, if built on faithful structural models, can be used to predict and interpret the dynamic response of commercial supercapacitors, not just idealized systems.
What This Means for Future Energy Storage
By showing that the smallest and most tortuous pores both boost capacitance and slow down charging, this work clarifies a key trade-off in carbon-based energy storage. Designers aiming for fast, high-capacity supercapacitors must balance how many ultramicropores they create, how these pores connect to larger channels that supply ions, and how the overall network affects charge flow. Beyond supercapacitors, the same modeling framework can be applied to other technologies—such as catalysts, desalination membranes, or gas storage materials—where fluids move and react inside complex, disordered pore networks.
Citation: Peng, J., Wu, T., Zeng, L. et al. Realistic atomic model for charge storage and charging dynamics of amorphous porous carbons. Nat Commun 17, 2425 (2026). https://doi.org/10.1038/s41467-026-69231-4
Keywords: supercapacitors, porous carbon, ionic liquids, nanopores, energy storage modeling