Clear Sky Science · en
Photocatalytic Stereoselective Editing of Alkynes to 3D Molecules via Hydrogen Atom Transfer-Mediated Dynamic Epimerization
Why Turning Flat Molecules into 3D Shapes Matters
Many modern medicines start from flat, sheet-like molecular pieces that are easy to make but not always ideal in the body. Drug hunters have learned that compact three-dimensional frameworks often behave better: they can fit more snugly into biological targets, move differently through the body, and sometimes cause fewer side effects. This article describes a new light-driven way to twist simple, flat molecular “wires” into intricate 3D cages in a single step, potentially speeding up the search for next-generation drugs and functional materials.
From Straight Lines to Tiny Cages
Most classic methods for building these cage-like structures, called bridged rings, take a scenic route: chemists first craft relatively flat ring systems and then stitch them together in a second step. That two-stage strategy works, but it is slow and limits which shapes can be reached. The authors instead start from very simple one-dimensional ingredients: linear alkynes (carbon–carbon triple bonds) and aldehydes (small carbon–oxygen units). Their goal is to jump directly from these straight chains to compact 3D frameworks in one operation, avoiding the usual detours and opening new “chemical space” that is valuable for drug design.
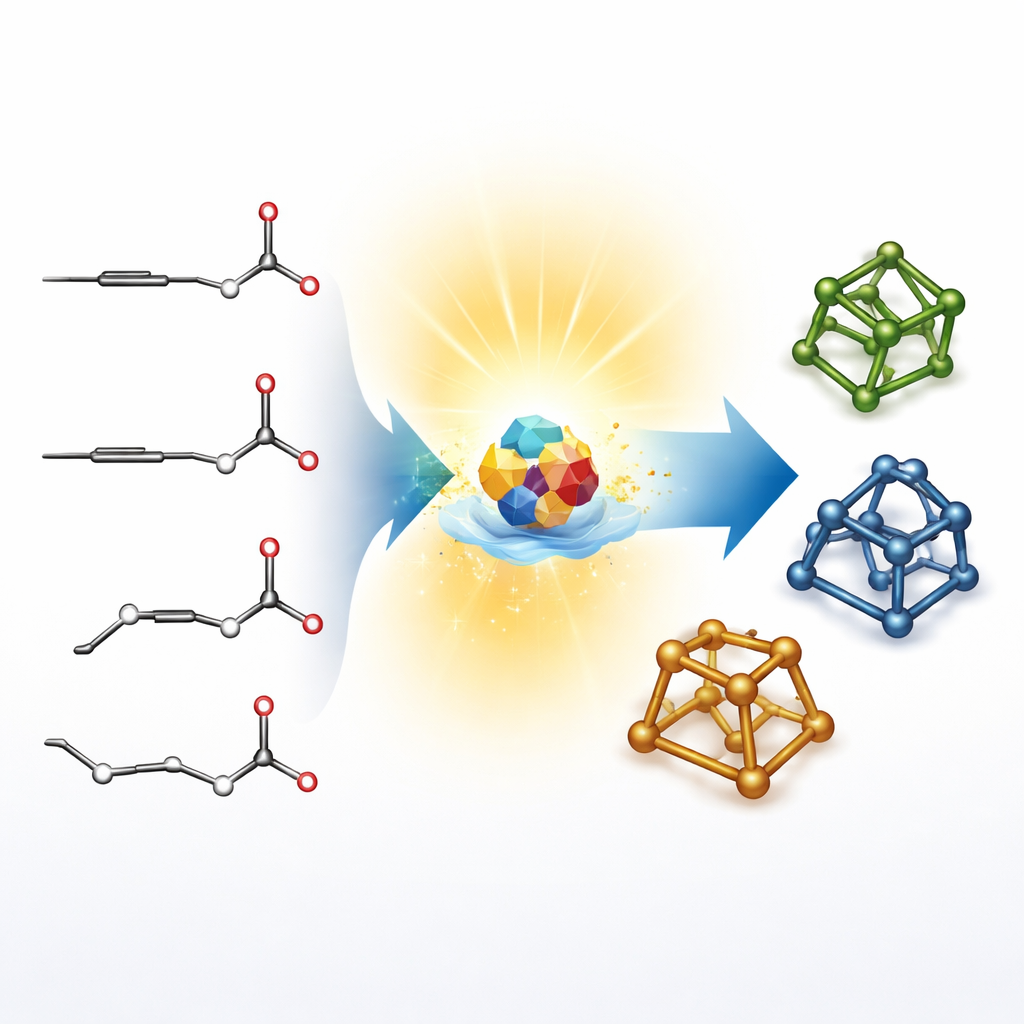
Letting Light Do the Heavy Lifting
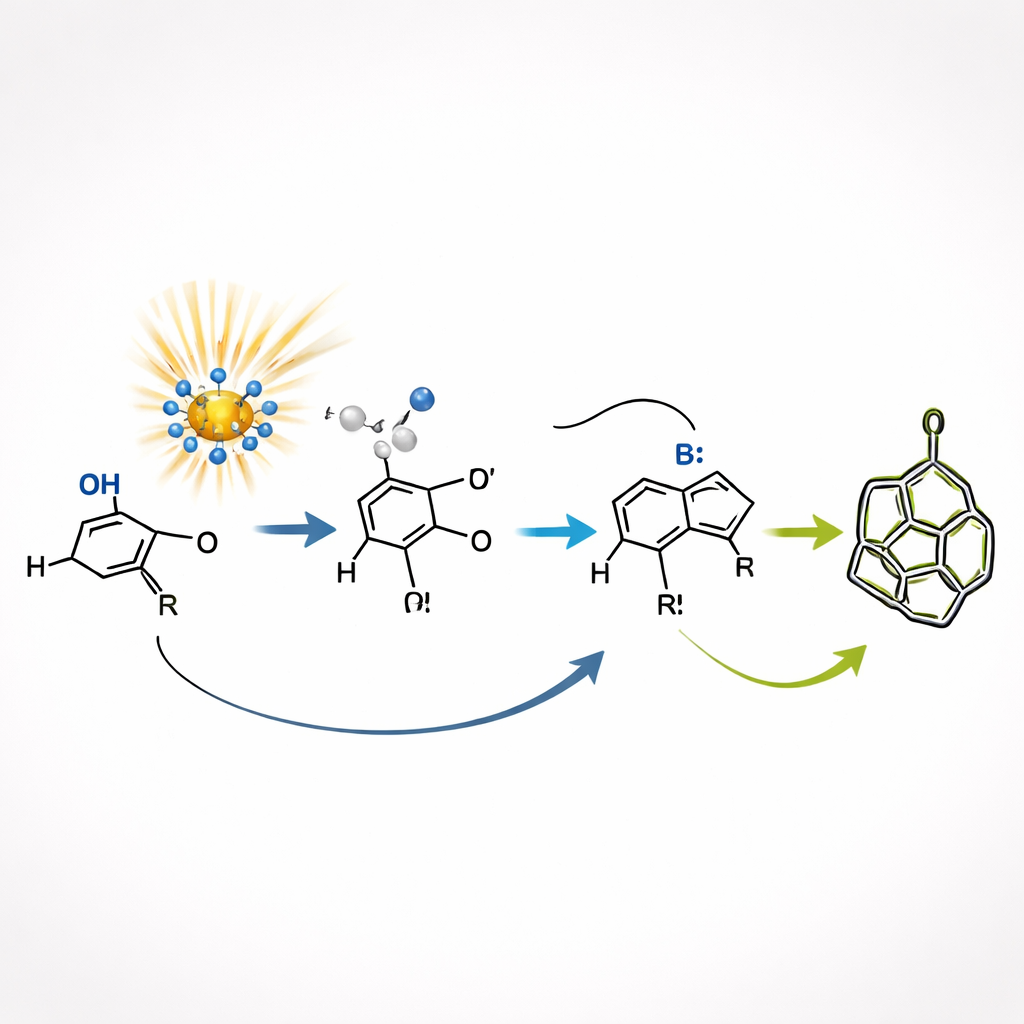
The team harnesses a special light-sensitive catalyst based on a polyoxometalate called decatungstate. Under violet light, this catalyst can grab a hydrogen atom from the aldehyde partner, briefly creating a highly reactive fragment. That fragment adds across the alkyne, folds back on itself, and closes into a five-membered ring. On its own, this first ring-forming step produces a messy mixture of 3D arrangements at key carbon atoms. The clever twist is that the same light-driven system can then reshuffle those arrangements in real time by repeatedly plucking and returning hydrogen atoms, allowing the mixture to drift toward the more stable shapes.
Self-Correcting 3D Editing
At the heart of the discovery is a kind of built-in self-correction, known as dynamic kinetic resolution. The initial ring intermediate exists as two closely related 3D forms, which differ only in how atoms are oriented in space. Light-activated hydrogen shuttling rapidly interconverts these forms, while a separate base-assisted closing step favors only one of them for final cage formation. As a result, the “preferred” intermediate is constantly drained away into a single, well-defined bicyclic product, and the less favored form is continuously converted into its partner. This interplay between reversible shape-flipping and selective trapping gives products with excellent control over orientation at multiple positions, even though the first step is quite unselective.
Building Diverse and Useful 3D Frameworks
The authors show that this light-driven approach works for a wide range of starting materials, decorating the resulting cages with many different chemical groups that chemists like to use as handles. They make two families of frameworks, bicyclo[2.2.1]heptanones and bicyclo[3.2.1]octanones, both of which are prized building blocks in medicinal chemistry. These structures appear in natural products, catalysts, and materials. The team further demonstrates that their 3D cages can be transformed into more elaborate compounds, including a key intermediate for an electroluminescent material and a rigidified version of a known brain-receptor blocker. In one case, swapping a flexible ring in the drug for a newly forged 3D cage even modestly improved its biological potency.
A Shortcut to Richer Molecular Shapes
In everyday terms, this work shows how to take simple, straight molecular “sticks” and, using light and a smart catalyst, fold them into precise 3D “sculptures” in a single move. The process does two jobs at once: it builds the underlying skeleton and fine-tunes the spatial arrangement of atoms, all under mild conditions. Because alkynes are common, inexpensive ingredients, this strategy could become a general shortcut for turning cheap raw materials into complex, drug-like architectures. As chemists push further into the world of three-dimensional molecules, tools like this may play a key role in crafting medicines and materials that work better by design.
Citation: Gu, Z., Zeng, T., Yuan, Z. et al. Photocatalytic Stereoselective Editing of Alkynes to 3D Molecules via Hydrogen Atom Transfer-Mediated Dynamic Epimerization. Nat Commun 17, 2518 (2026). https://doi.org/10.1038/s41467-026-69219-0
Keywords: photocatalysis, hydrogen atom transfer, 3D molecular scaffolds, dynamic kinetic resolution, medicinal chemistry