Clear Sky Science · en
Heritable changes in chromatin contacts associated with transgenerational susceptibility to diet-induced insulin dysregulation and obesity
When Ancestral Diet Meets Modern Waistlines
Why do some people gain weight and develop blood sugar problems more easily than others, even when their own lifestyle looks healthy? This study explores an unsettling possibility: that exposure to certain industrial chemicals during pregnancy can quietly "prime" descendants, generations later, to respond badly to rich diets. Working in mice, the researchers uncover a way that this risk can be passed on without changing DNA letters, by reshaping how the genome folds inside cells.
A Chemical That Leaves a Long Shadow
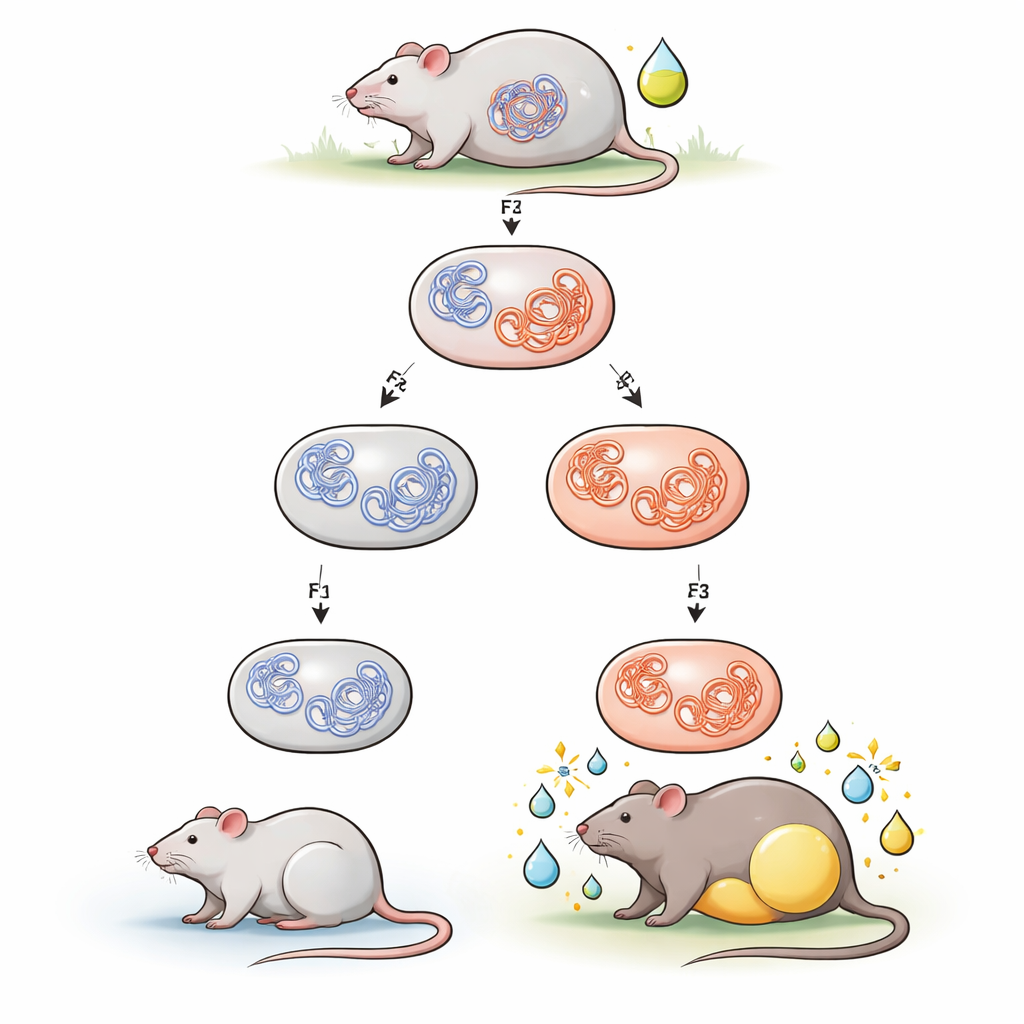
The team focused on tributyltin, a once-common antifouling and industrial compound known as an obesogen because it promotes fat storage. Pregnant mice were given low doses of tributyltin in their drinking water during gestation. Their offspring (the first generation) were exposed in the womb, their grandchildren (second generation) were exposed as developing germ cells inside those offspring, and their great-grandchildren (third generation) were never directly exposed. When young adult descendants were switched from standard chow to a higher-fat diet, males whose ancestors encountered tributyltin gained more body fat and weight than unexposed controls. Females, in contrast, showed little or no change. This male-specific tendency toward diet-induced obesity echoed earlier work from the same group.
Genome Folding as a Hidden Memory
To understand how this trait could be inherited without DNA mutations, the researchers examined primordial germ cells from embryos in the first three generations. These are the immature cells that eventually form sperm and eggs. Using a technique that maps which pieces of DNA touch each other in three-dimensional space, they looked for durable changes in how chromosomes fold. They found that, overall, the pattern of contacts was quite stable. However, in male germ cells, one small region on chromosome 19 stood out: a cluster of new, strengthened contacts appeared and persisted from the directly exposed embryos through two unexposed generations. This region contains the Ide gene, which encodes insulin-degrading enzyme, a key protein that normally helps clear insulin from the bloodstream.
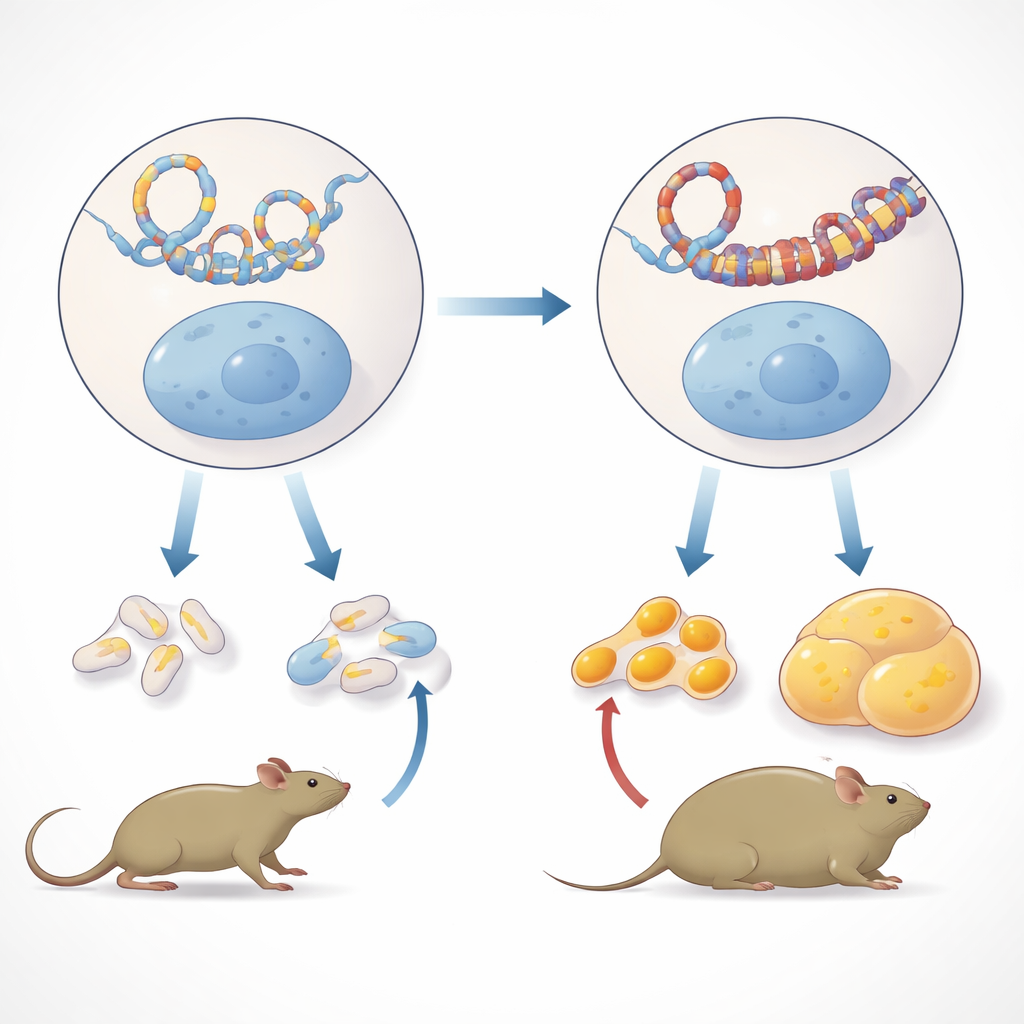
A Single Gene at the Heart of the Metabolic Shift
The team then asked whether these altered DNA contacts had consequences in ordinary body tissues. In the livers of adult great-grandsons of the exposed mice, they saw stronger binding of a genome-organizing protein called CTCF at several sites within the Ide region, consistent with the formation of a new chromatin loop. At the same time, Ide activity in male livers dropped, while nearby genes Hhex and Kif11 became more active. The pattern did not appear in female livers or in other male tissues such as muscle, fat pads, brain, or spleen, underscoring its tissue- and sex-specific nature. Importantly, the authors ruled out a simple structural duplication of this part of the chromosome as an explanation, showing that copy number of the Ide region did not differ between exposed and control lineages.
From Altered Insulin Clearance to Obesity
Because insulin-degrading enzyme helps remove insulin from circulation, reduced Ide activity should leave more insulin in the blood. That is exactly what the researchers saw. Even before the high-fat diet, male descendants of tributyltin-exposed dams had elevated insulin levels and higher scores on a standard index of insulin resistance, despite normal fasting blood sugar. After weeks on a richer diet, they developed both high blood sugar and high insulin, along with larger fat stores and elevated leptin, a hormone linked to body fat. Levels of C-peptide, which tracks how much insulin is being produced, rose similarly in exposed and control animals. The crucial difference was the ratio of C-peptide to insulin, which pointed to impaired breakdown of insulin rather than extra secretion—consistent with the observed drop in hepatic Ide.
What This Means for Inheritance and Health
Taken together, the findings outline a chain of events: ancestral exposure to an obesogenic chemical subtly reshapes how a specific region of the genome folds in male germ cells; this new 3D structure is recreated in the livers of male descendants, dialing down a single insulin-clearing gene; as a result, these animals carry chronically higher insulin levels and are primed to gain fat and develop blood sugar problems when challenged with a high-fat diet. For a lay reader, the key message is that what ancestors encounter in their environment can leave a physical "memory" in how DNA is packaged, not by changing genetic code but by changing its shape. That memory can quietly influence how later generations respond to modern diets, highlighting the importance of limiting harmful exposures long before health problems appear.
Citation: Chang, R.C., Egusquiza, R.J., Amato, A.A. et al. Heritable changes in chromatin contacts associated with transgenerational susceptibility to diet-induced insulin dysregulation and obesity. Nat Commun 17, 2662 (2026). https://doi.org/10.1038/s41467-026-69214-5
Keywords: transgenerational epigenetics, obesogens, insulin resistance, chromatin structure, environmental exposure