Clear Sky Science · en
Engineering a cytochrome P450 enzyme as a peroxygenase for selective hydroxylation of steroids
Making Medicines from Tough-to-Tune Molecules
Steroid drugs—from anti-inflammatory pills to hormone therapies—are among the most widely used medicines in the world. Yet fine-tuning these molecules, by adding a single oxygen atom at exactly the right spot, is notoriously hard using traditional chemistry. This study shows how scientists re‑designed a natural enzyme so that it can precisely “polish” steroid molecules using simple hydrogen peroxide, potentially making greener, cheaper routes to important drugs.
Why Steroids Are So Hard to Customize
Steroids are built from a compact stack of carbon rings, with many nearly identical carbon–hydrogen bonds. Conventional chemical methods struggle to target just one of these bonds without hitting others, often requiring harsh reagents, high temperatures, and multiple protection steps that generate waste. Yet small changes—such as adding a single hydroxyl group (an –OH) at one specific position—can dramatically alter how a steroid behaves in the body. Enzymes from the cytochrome P450 family are nature’s specialists for this kind of precise C–H bond activation, but in their natural form they usually need expensive helper molecules and oxygen-handling machinery, which limits their direct use in manufacturing.
Discovering a Promising Enzyme Starting Point
Using genome mining, the researchers identified a self-sufficient P450 enzyme, called P450stri, from a soil bacterium. Unlike a classic workhorse enzyme named P450BM3, which prefers small fatty acids, P450stri naturally accepts bulky steroid molecules such as testosterone. Under its native conditions, powered by the cellular cofactor NADH, P450stri adds hydroxyl groups at three positions on testosterone, producing a mixture of products. This built‑in ability to recognize steroids made P450stri an attractive “starting scaffold” for engineering a more selective, industrially friendly catalyst.
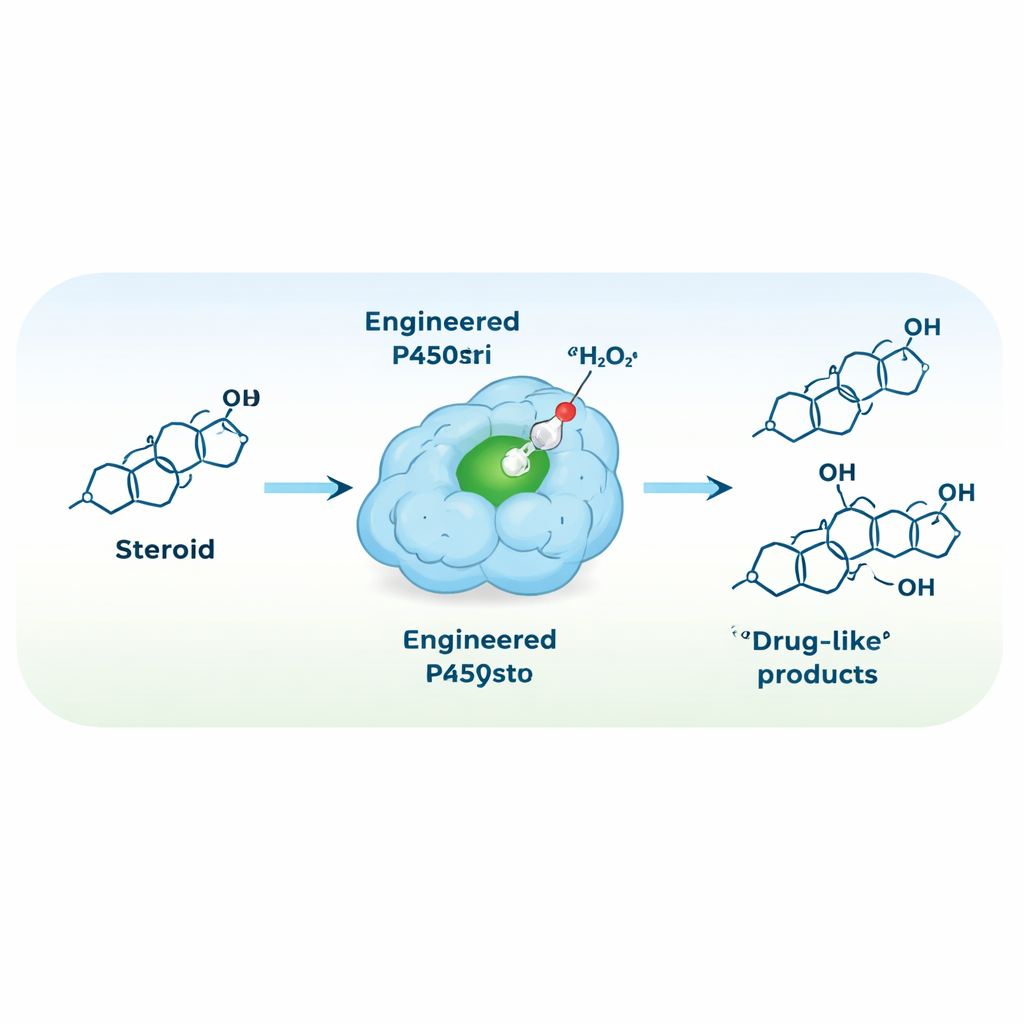
Rewiring the Enzyme to Run on Hydrogen Peroxide
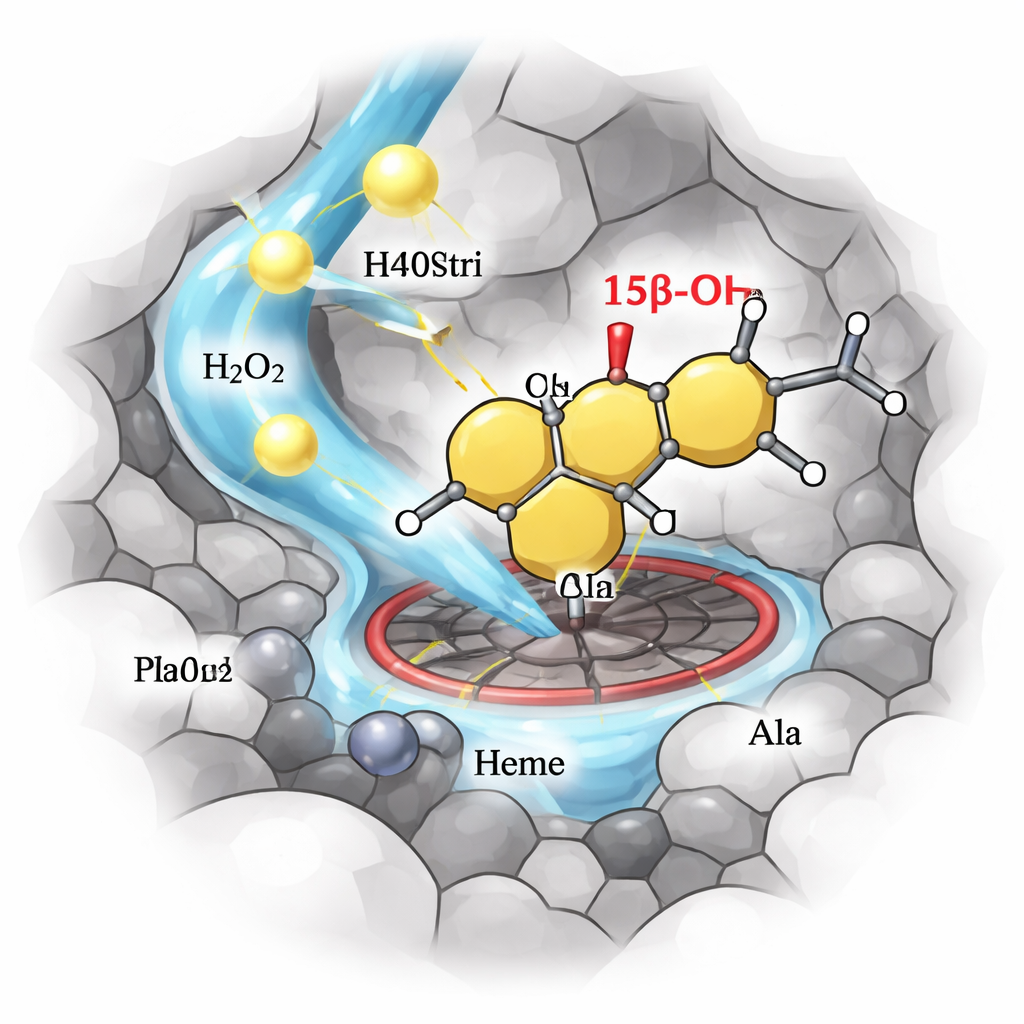
To simplify the reaction setup, the team set out to convert P450stri into a peroxygenase—an enzyme that uses hydrogen peroxide directly as the oxidant, removing the need for costly cofactors and auxiliary proteins. A single strategic mutation, replacing a bulky phenylalanine just above the heme center with a smaller alanine, created a variant called M1. This change opened space around the reactive iron center and surprisingly made the enzyme both more tolerant to high hydrogen peroxide levels and far more selective. In the new, peroxide-driven mode, M1 converted testosterone mainly into a single product, hydroxylated at the 15β position, with about 94% selectivity—up from 35% in the original enzyme.
A “Round Flask” Model for Smarter Enzyme Design
To push performance further, the authors introduced a design concept they call the “Round Flask” model. They treat the active site where chemistry happens as the rounded base and the narrow access tunnels as the neck and cap. The base determines which position on the steroid is modified, while the neck controls how easily the steroid and hydrogen peroxide reach the reactive center. Guided by computer simulations and tunnel-mapping software, they systematically adjusted a small set of amino acids lining these tunnels, tuning their size and hydrophobicity. This two-dimensional strategy yielded a standout variant, M4, which maintained high 15β selectivity while greatly boosting the reaction rate and tolerating hydrogen peroxide concentrations up to several hundred millimolar.
From Lab Bench to Scalable Steroid Upgrades
The engineered M4 enzyme was then tested in larger-scale reactions with testosterone and a related steroid used in performance-enhancing contexts. In simple aqueous mixtures at room temperature, driven only by hydrogen peroxide, M4 converted over 90% of the starting material and delivered isolated 15β-hydroxylated products in yields around 75–80%. These conditions are far milder and cleaner than typical chemical routes. Finally, the team showed that by transplanting the key mutations into several related P450 enzymes in the same evolutionary “branch,” they could reliably turn them into similarly selective and efficient peroxygenases, highlighting the generality of their design rules.
What This Means for Future Medicines
For a non-specialist, the bottom line is that the researchers have taught a natural enzyme to act like a highly precise, peroxide-powered “micro factory” for modifying steroid drugs. By combining a simple conceptual model with targeted mutations, they created an enzyme that can add an OH group at just the right spot, under gentle and scalable conditions, and then transferred this strategy to related proteins. This approach could streamline the production of existing steroids, open routes to new drug candidates, and serve as a blueprint for engineering other enzymes that cleanly activate stubborn C–H bonds in complex molecules.
Citation: Tang, T., Wang, R. & Chen, Y. Engineering a cytochrome P450 enzyme as a peroxygenase for selective hydroxylation of steroids. Nat Commun 17, 1996 (2026). https://doi.org/10.1038/s41467-026-69211-8
Keywords: steroid biocatalysis, cytochrome P450 engineering, peroxygenase, hydrogen peroxide catalysis, selective hydroxylation